MANAGING BENIGN PROSTATIC HYPERPLASIA: INTERDISCIPLINARY STRATEGIES FOR IMPROVED OUTCOMES
Faculty:
L. Austin Fredrickson, MD, FACP
L. Austin Fredrickson is an Associate Professor of Internal Medicine at Northeast Ohio Medical University, where he serves as core faculty and teaches diagnostics, therapeutics, clinical skills, and health humanities. He is board-certified in general internal medicine and practices rural primary care.
Kristina (Tia) Neu, RN
Kristina (Tia) Neu is a licensed Registered Nurse and author currently developing in-service training for healthcare professionals. She is a National Board-Certified Health & Wellness and Lifestyle Medicine Coach. Her work experience includes several areas of the healthcare profession, such as psychiatric nursing, medical nursing, motivational health coaching, chronic case management, dental hygiene, cardiac technician, and surgical technician.
Liz Fredrickson, PharmD, BCPS
Liz Fredrickson, PharmD, BCPS, is an Associate Professor of Pharmacy Practice and Pharmaceutical Sciences at the Northeast Ohio Medical University (NEOMED) College of Pharmacy, where she is course director of the Parenteral Products and Basic Pharmaceutics Lab courses.
Pamela Sardo, PharmD, BS
Pamela Sardo, PharmD, BS, is a freelance medical writer and licensed pharmacist. She is the founder and principal at Sardo Solutions in Texas. Pam received her BS from the University of Connecticut and her PharmD from the University of Rhode Island. Pam’s career spans many years in retail, clinics, hospitals, long-term care, Veterans Affairs, and managed health care responsibilities across a broad range of therapeutic classes and disease states.
Abstract
Within the realm of men’s health conditions, benign prostatic hyperplasia (BPH) is a disorder with significant public health consequences. Benign prostatic hyperplasia is a histological diagnosis and refers to a benign growth of prostate tissue. It is a common cause of lower urinary tract symptoms (LUTS), which often progress in prevalence and severity as men age. Patients with BPH require a patient-centered, holistic approach to care that involves improving symptoms and quality of life, preventing disease progression, and minimizing medication side effects. All healthcare providers can play an important role in identifying patients who require BPH treatment, recommending referrals, and supporting the care team with BPH medication management. Further, primary care physicians and pharmacists can provide crucial counseling on the appropriate use of BPH medications and help patients to mitigate side effects and avoid harmful drug interactions.
Accreditation Statements
In support of improving patient care, RxCe.com LLC is jointly accredited by the Accreditation CouncilTM for Continuing Medical Education (ACCME®), the Accreditation Council for Pharmacy Education (ACPE®), and the American Nurses Credentialing Center (ANCC®), to provide continuing education for the healthcare team.

This activity was planned by and for the healthcare team, and learners will receive 2 Interprofessional Continuing Education (IPCE) credits for learning and change.
Joint Universal Activity Number: The Joint Accreditation Universal Activity Numbers assigned to this activity are as follows:
Pharmacists: JA4008424-0000-26-031-H01-P
Pharmacy Technicians: JA4008424-0000-26-031-H01-T
Credits: 2 contact hour(s) (0.2 CEU(s)) of continuing education credit.
Credit Types:
IPCE Credits - 2 Credits
AAPA Category 1 Credit™️ - 2 Credits
AMA PRA Category 1 Credit™️ - 2 Credits
Pharmacy - 2 Credits
Type of Activity: Knowledge
Media: Computer-Based Training (i.e., online courses)
Estimated time to complete activity: 2 contact hour(s) (0.2 CEU(s)), including Activity Pre-Test, Post-Test, and Activity Evaluation.
Release Date: March 18, 2026 Expiration Date: March 18, 2029
Target Audience: This educational activity is for Physicians, Physician Assistants, Pharmacists, and Pharmacy Technicians
How to Earn Credit: From March 18, 2026, through March 18, 2029, participants must:
Read the “learning objectives” and “author and planning team disclosures;”
Take the “Educational Activity Pre-Test;”
Study the section entitled “Educational Activity;” and
Complete the Educational Activity Post-Test and Activity Evaluation. The Educational Activity Post-Test will be graded automatically. Following successful completion of the Educational Activity Post-Test with a score of 70% or higher, a statement of participation will be made available immediately. (No partial credit will be given.)
CME Credit: Credit for this course will be uploaded to CPE Monitor® for pharmacists. Physicians may receive AMA PRA Category 1 Credit™️ and use these credits toward Maintenance of Certification (MOC) requirements. Physician Assistants may earn AAPA Category 1 CME credit, reportable through PA Portfolio. All learners shall verify their individual licensing board’s specific requirements and eligibility criteria.
Statement of Need
Benign prostatic hypertrophy is common in aging men and is a leading cause of lower urinary tract symptoms, impaired quality of life, and increased healthcare utilization. Many patients are underdiagnosed or sub-optimally managed. Healthcare professionals may be unfamiliar with the hormonal and other mechanisms driving benign prostatic tissue overgrowth, which limits their ability to link etiology and pathophysiology to individualized treatment plans. Evolving guideline-directed pharmacologic options require careful comparison of indications, contraindications, side-effect profiles, and monitoring requirements. Clearly defined collaborative roles among primary care providers, urologists, and other team members help optimize care. This activity aims to address these gaps to improve assessment, medication optimization, and timely referrals.
Learning Objectives: Upon completion of this educational activity, participants should be able to:
Describe the etiology of BPH
Compare and contrast treatment strategies for BPH
Recall indications, contraindications, side effect profiles, and monitoring parameters for pharmacologic therapies
Identify the roles of healthcare team members in the management of BPH
Disclosures
The following individuals were involved in developing this activity: L. Austin Fredrickson, MD, FACP; Liz Fredrickson, PharmD, BCPS; Kristina (Tia) Neu, RN; and Pamela Sardo, PharmD, BS. None of the individuals involved in developing this activity has a conflict of interest or financial relationships related to the subject matter. There are no financial relationships or commercial or financial support relevant to this activity to report or disclose by RxCe.com or any of the individuals involved in the development of this activity.
© RxCe.com LLC 2026: All rights reserved. No reproduction of all or part of any content herein is allowed without the prior, written permission of RxCe.com LLC.
Educational Activity Pre-Test
Which of the following hormones plays a key role in the development of BPH by stimulating epithelial proliferation in the prostate?
Testosterone
Estrogen
Dihydrotestosterone (DHT)
Progesterone
What role does estrogen play in the pathophysiology of BPH?
Directly stimulates epithelial proliferation
Blocks androgen receptors
Promotes stromal growth indirectly via androgen receptors
Suppresses DHT production
What monitoring parameter is essential before and during treatment with 5-alpha reductase inhibitors?
Blood glucose
Prostate-specific antigen (PSA)
Serum creatinine
Liver enzymes
Educational Activity
Managing Benign Prostatic Hyperplasia: Interdisciplinary Strategies for Improved Outcomes
Introduction
Benign prostatic hyperplasia (BPH) is most prevalent in men as they age. Effective management of BPH requires a patient-centered, multidisciplinary approach that prioritizes symptom relief, quality-of-life improvement, prevention of complications, and minimization of treatment-related adverse effects. Healthcare professionals should familiarize themselves with BPH management guidelines and follow them. This continuing education activity will review the etiology and pathophysiology of BPH and examine current treatment options, including their mechanisms of action, side-effect profiles, and contraindications. It will also emphasize the collaborative roles of the interdisciplinary care team in delivering high-quality, evidence-based care for patients with BPH.
Overview of Benign Prostatic Hyperplasia
Benign prostatic hyperplasia (BPH) is one of the most prevalent and burdensome conditions affecting men’s health, with significant clinical and public health implications.1,2 Confirmation of prostate growth or inflammation is done histologically, i.e., by performing a biopsy (microscopic examination of prostate tissue).3
Histologically, BPH is characterized by the nonmalignant proliferation of prostatic stromal and epithelial tissue, leading to progressive enlargement of the prostate gland.1,2 This growth often results in lower urinary tract symptoms (LUTS), such as urinary frequency, urgency, nocturia, and decreased urinary stream, which can substantially diminish the patient’s quality of life.1,2 Beyond its clinical burden, BPH also contributes to significant healthcare resource utilization, including outpatient visits, medication use, surgical interventions, and hospitalizations.2
Effective management of BPH requires a patient-centered, multidisciplinary approach that prioritizes symptom relief, quality-of-life improvement, prevention of complications, and minimization of treatment-related adverse effects. The American Urological Association (AUA) provides evidence-based guidelines to support clinicians in the diagnosis and treatment of BPH and LUTS, most recently updated in 2021 and reaffirmed with a 2023 amendment.1,4 These updates reinforce the importance of shared decision-making and highlight the need for cautious use of combination therapy in older adults due to concerns about polypharmacy.¹ Achieving optimal outcomes depends on the coordinated efforts of primary care providers, urologists, pharmacists, and other healthcare professionals, each of whom plays a vital role in the evaluation, management, and ongoing support of patients with BPH. A holistic assessment and individualized treatment plan, informed by guideline-based care and tailored to patient needs, are essential components of effective, long-term management.
Epidemiology
Benign prostatic hyperplasia is among the most frequently diagnosed urologic disorders in older men and one of the most common causes of LUTS.1,2 Histologic evidence of BPH is rare in men under age 40; however, the prevalence increases substantially with age, starting at age 40-45 years, affecting approximately 50% of men in their 50s, 60% by age 60, and 80% by age 80.1,2
Although histologic BPH does not always result in symptoms, about 25% of men will develop moderate-to-severe LUTS by age 55, and more than 50% by age 75.5 Globally, the burden of BPH continues to rise in parallel with aging populations. In the United States alone, BPH accounts for millions of outpatient visits annually and generates substantial healthcare expenditures, including medication costs, diagnostic procedures, and surgical interventions.6 Despite its high prevalence, many men do not seek care until symptoms significantly interfere with daily activities, highlighting the need for greater awareness and proactive screening in primary care and urology settings.2
Pathophysiology
The underlying pathophysiology of BPH involves several interrelated mechanisms, including the intraprostatic accumulation of dihydrotestosterone (DHT), increased activity of type 2 5-alpha reductase, and heightened alpha-adrenergic tone within the prostatic stroma and bladder neck.2 These factors contribute to static and dynamic components of urethral compression, ultimately leading to LUTS and bladder outlet obstruction in affected individuals.2 To better understand how these changes occur, it is essential first to examine the anatomy and hormonal function of the prostate gland.
The prostate is a small, chestnut-shaped gland situated just below the bladder and in front of the rectum. 2,7,8 (See Figure 1.) It surrounds the urethra—the tube that carries urine from the bladder out of the body.2
Figure 1
Normal versus Enlarged Prostate7
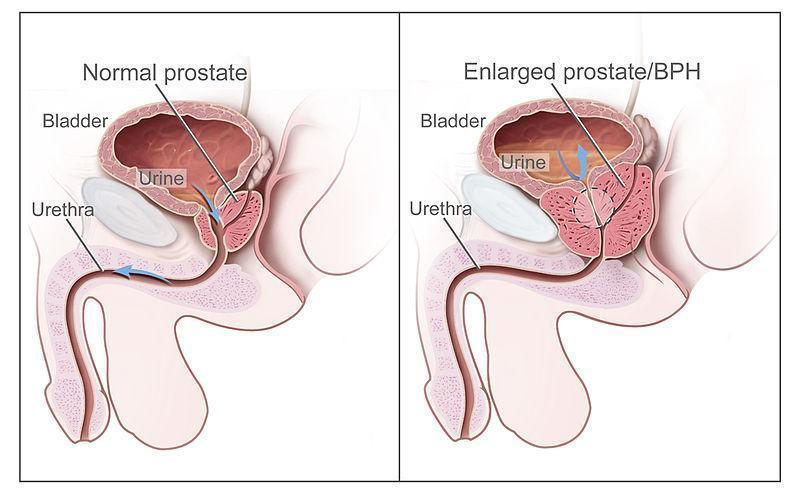
The prostate’s primary role is to produce a fluid that constitutes approximately 20–40% of the ejaculate, contributing to sperm motility and fertility.2 Testosterone, the primary male sex hormone, is converted within the prostate by the enzyme 5-alpha reductase into dihydrotestosterone (DHT). This more potent androgen plays a pivotal role in the development of BPH.2 Prostatic epithelial and stromal cells express androgen receptors (ARs), and DHT binding stimulates epithelial proliferation, while estrogens indirectly promote stromal growth via estrogen-sensitive AR pathways.2 This hormonal signaling drives gradual glandular enlargement over time.2
In adult men, a healthy prostate typically weighs between 15 and 20 grams (about one ounce).2 The prostate experiences two primary growth phases: a rapid one during puberty and a second, slower phase beginning around age 25.2 In men with BPH, this second phase progresses abnormally, leading to prostatic enlargement.2 Even modest inward (periurethral) growth can constrict the urethra and disrupt urinary flow, as the urethra’s diameter is quite narrow.2 This constriction can cause progressive bladder outlet obstruction, and as the bladder struggles to empty, it compensates by hypertrophying the detrusor muscle.2 Over time, bladder contractility weakens, leading to residual urine retention.2 This dysfunction results in characteristic LUTS, including increased frequency, urgency, nocturia, hesitancy, and a weak urinary stream.2 Many patients describe a persistent feeling of incomplete emptying and frequent urges to void.
Etiology
Benign prostatic hyperplasia is associated with non-modifiable and modifiable risk factors.6 While age and genetics are primary non-modifiable contributors, growing evidence highlights the importance of lifestyle and metabolic health in BPH development and progression.6
Poor diet, sedentary behavior, obesity, and insulin resistance are all linked to greater prostate volume and LUTS severity.6 In one study, men with metabolic syndrome were 80% more likely to experience LUTS compared to those without any metabolic syndrome components.9 Obesity appears to correlate directly with prostate enlargement, potentially due to hormonal dysregulation and increased systemic inflammation.6 Conversely, physical activity may be protective. Men who engage in moderate-to-vigorous exercise have been shown to have a 25% reduced risk of developing BPH or of requiring surgical intervention compared to sedentary individuals.10 Regular exercise can also improve insulin sensitivity, reduce inflammation, and modulate hormone levels, collectively mitigating BPH risk.10 Non-modifiable and modifiable BPH risks are detailed in Table 1 below.
Table 1
Risk Factors for BPH6
| Non-modifiable | Modifiable |
Age Genetics | Testosterone Dihydrotestosterone Estrogen Metabolic syndrome Obesity Diabetes Diet Physical activity Inflammation |
Diagnosis and Evaluation
Men with BPH often present either symptomatically with LUTS or asymptomatically with BPH discovered incidentally during digital rectal examination or imaging.2 Lower urinary tract symptoms associated with BPH are generally categorized as either obstructive (e.g., hesitancy, straining, weak stream) or irritative, such as frequency, nocturia, and/or urgency (Table 2).2 The presence, severity, and type of symptoms help clinicians assess the degree and impact of prostate-related obstruction and guide management.
Table 2
Lower Urinary Tract Symptoms2
| Obstructive Symptoms | Irritative Symptoms |
Urine hesitancy Straining Weak flow Prolonged voiding Partial or complete urinary retention Urinary incontinence | Urinary frequency Nocturia Dysuria Decreased void volume |
A thorough evaluation is essential prior to diagnosing BPH. This includes obtaining a detailed medical and medication history, performing a physical examination (including a digital rectal exam or DRE), and ruling out other causes of LUTS.1,2 Notably, prostate size on examination does not always correlate with symptom severity, as inward growth around the urethra can cause obstruction even if the prostate feels only mildly enlarged on DRE.1,2,11 To quantify the severity of symptoms, clinicians frequently use the American Urological Association Symptom Score (AUASS) (Questionnaire 1) or its international counterpart, the International Prostate Symptom Score (IPSS) (Questionnaire 2).12,13 The IPSS includes an additional quality-of-life question assessing how bothersome the symptoms are from the patient’s perspective.1 Both tools assign numeric values to symptoms such as urinary frequency, nocturia, weak stream, intermittency, and urgency.1,13 Patients with scores of 0–7 are considered to have mild symptoms, 8–19 moderate, and 20–35 severe.¹ Voiding diaries, which track frequency and volume of urination over several days, can also help objectively evaluate LUTS severity.2
Questionnaire 1
AUA Symptom Score1
| Symptom score point scale | ||
| Use the following point scale to answer the questions. | ||
| ||
| ||
| ||
| ||
| ||
| ||
| Symptom score questions | ||
| ||
| ||
| ||
| ||
| ||
| ||
| ||
| ||
| ||
| ||
| ||
| ||
| ||
| Score and severity of the problem | ||
| Score | Severity | |
| 0 to 7 | Mild | |
| 8 to 19 | Moderate | |
| 20 to 35 | Severe | |
Questionnaire 2
IPSS Additional Quality of Life Question to AUA Score1
| Quality of Life Additional Question |
| If you were to spend the rest of your life with your urinary condition just the way it is now, how would you feel about that? |
|
|
|
|
|
|
|
Differential Diagnosis
Because LUTS are not specific to benign prostatic hyperplasia (BPH), a thorough differential diagnosis is essential. A focused history and physical exam should be accompanied by urinalysis to rule out urinary tract infections, hematuria, or crystalluria suggestive of urolithiasis.2 Prostate-specific antigen (PSA) testing may aid in evaluating prostate cancer risk, though elevated levels can also result from benign prostatic enlargement, inflammation, trauma, or infection.14
Objective assessments such as uroflowmetry and post-void residual (PVR) measurement provide valuable information. A peak urinary flow rate below 10–12 mL/sec may indicate bladder outlet obstruction or detrusor underactivity, while a PVR above 25–50 mL suggests impaired bladder emptying due to obstruction or weak detrusor contraction.2,15,16 Notably, not all patients with obstructive symptoms have prostatic enlargement; increased alpha-adrenergic tone in the bladder neck and prostatic stroma can cause functional obstruction, exacerbated by stress or sympathetic overactivity.2
Several conditions can mimic or coexist with BPH, including urethral stricture, neurogenic bladder, and detrusor overactivity.11 Systemic diseases, such as diabetes mellitus, may cause polyuria and frequency from glucosuria, while nocturia may reflect heart failure or excessive fluid intake before bed.17 Hematuria, although sometimes caused by fragile prostatic vessels, warrants evaluation for malignancy or stones if it is gross or persistent.2,11 Ultimately, distinguishing BPH from other etiologies of LUTS requires a comprehensive and individualized evaluation to guide appropriate management and referral when necessary.11
Medications that Exacerbate BPH
Clinicians should be vigilant in identifying medications that may worsen symptoms of BPH, as certain agents can contribute to urinary retention or exacerbate LUTS. For example, alpha-adrenergic agonists such as pseudoephedrine and phenylephrine increase smooth muscle tone in the prostate and bladder neck, leading to urethral constriction.2 Beta-adrenergic agonists like terbutaline may relax the detrusor muscle, impairing bladder emptying.2 Additionally, anticholinergics, antihistamines, phenothiazines, and tricyclic antidepressants can reduce detrusor contractility, further increasing the risk of urinary retention in patients with compromised urinary flow.2
Polyuria may also occur as a side effect of certain medications unrelated to bladder function. For example, sodium-glucose cotransporter-2 (SGLT2) inhibitors such as canagliflozin and dapagliflozin promote glucosuria as part of their glucose-lowering effect, leading to increased urinary frequency.11 Recognizing these pharmacologic contributors is critical when evaluating LUTS and developing a safe and effective management plan for patients with BPH.
Table 3
Medications that Exacerbate BPH
| Medication/Class | Therapeutic Category | Reason for Exacerbating BPH |
|---|---|---|
| Pseudoephedrine | Alpha-adrenergic agonist | Stimulates alpha receptors in the prostate, causing smooth muscle contraction |
| Phenylephrine | Alpha-adrenergic agonist | Increases smooth muscle tone in the prostate and bladder neck, worsening urinary obstruction |
| Terbutaline | Beta-adrenergic agonist | Relaxes the bladder detrusor muscle, impairing bladder emptying |
| Anticholinergic agents | Various (e.g., bladder antispasmodics, antihistamines) | Decrease detrusor contractility, leading to urinary retention |
| First-generation antihistamines (e.g., diphenhydramine) | H1 receptor antagonists | Anticholinergic effects reduce bladder contractility |
| Phenothiazines | Antipsychotics | Anticholinergic properties reduce detrusor function |
| Tricyclic antidepressants (e.g., amitriptyline) | Antidepressants with anticholinergic effects | Impair bladder emptying due to anticholinergic activity |
| Canagliflozin | SGLT2 inhibitor (antidiabetic) | Increases urinary frequency by promoting glycosuria, potentially worsening LUTS |
| Dapagliflozin | SGLT2 inhibitor (antidiabetic) | Causes polyuria through osmotic diuresis, potentially aggravating urinary symptoms |
Management of BPH
The AUA guidelines provide evidence-based recommendations for the management of BPH-related LUTS, though they intentionally do not prescribe a rigid sequence of treatments.1 Instead, treatment plans should be individualized based on factors such as symptom severity (obstructive vs irritative), prostate size, risk of disease progression, patient comorbidities, medication side effects, and patient preferences.1 The primary goals of BPH management are to reduce bothersome LUTS, prevent disease progression, and avoid complications or surgery.2 A reduction of at least 3 points in the AUA Symptom Score is generally considered a clinically meaningful improvement.1,2 In addition to symptom relief, consideration should be given to preserving bladder function and preventing acute urinary retention. Use of the IPSS is recommended for symptom assessment and treatment monitoring, although the strength of supporting evidence is limited.1 Initial management should include shared decision-making that incorporates education, lifestyle counseling, pharmacologic options, and a discussion of potential surgical referral when appropriate.
Watchful Waiting
Watchful waiting is recommended as an initial strategy for men with mild or non-bothersome symptoms (IPSS score ≤7).1,6 This approach involves routine monitoring every 6 to 12 months to assess symptom progression and quality of life.2 At follow-up visits, clinicians should review the patient’s symptom scores (e.g., IPSS), evaluate for any new complications, and reinforce symptom management strategies.1 Even though no medications are prescribed at this stage, patients should be counseled on lifestyle modifications that may prevent symptom worsening, including the following:2
Reducing caffeine and alcohol intake
Avoiding evening fluid consumption
Practice double voiding or scheduled voiding
Avoiding constipation
Identifying and discontinuing medications that exacerbate BPH (e.g., antihistamines, decongestants)
Pharmacologic Treatment
Guideline Summary
The "Trial of Medical Therapy Algorithm" outlines a stepwise approach to managing LUTS associated with BPH.1,2 Treatment typically begins with an alpha-blocker, unless the patient also has erectile dysfunction (ED), in which case a phosphodiesterase type 5 inhibitor (PDE5i) may be considered as initial therapy.1,2 If the patient experiences a lack of or incomplete response to alpha-blocker therapy or is unable to tolerate it, the next steps depend on prostate size and symptom severity.1,2 For patients with prostates larger than 30 cc, the addition of a 5-alpha reductase inhibitor (5ARI) should be considered.1,2 In cases of persistent or incomplete symptom control, a trial of a PDE5i may be appropriate. If symptoms still do not improve, surgical options should be discussed using a dedicated surgical management algorithm. Additionally, patients with mixed symptoms of overactive bladder (OAB) and bladder outlet obstruction (BOO) should be managed in accordance with OAB guidelines, including potential use of anticholinergic or beta-3 agonist therapy.1,2
The "Basic Management of LUTS in Men" algorithm provides a structured approach for evaluating and treating bothersome LUTS.1,2,4 Initial management involves obtaining a medical history, conducting a physical exam, administering a symptom score such as the International Prostate Symptom Score (IPSS), and performing urinalysis.1,2,4 If equipment is available, post-void residual (PVR) and uroflowmetry are recommended. Regardless of symptoms, a PVR ≥300 cc should prompt evaluation by a specialist. Once reversible factors such as caffeine intake and fluid timing are addressed, standard treatment is initiated based on the Trial of Medical Therapy Algorithm.1,2,4
If symptoms persist or worsen, further evaluation is warranted. This may include optional tests such as repeat PVR, uroflowmetry, ultrasound, or cystoscopy, especially if these were not performed earlier.1,2,4 Depending on symptom patterns, the approach may differ: for nocturia-predominant cases, a frequency-volume chart and medication trial are advised; for overactive bladder (OAB)-predominant symptoms, follow the OAB guideline; and for mixed symptoms involving OAB and bladder outlet obstruction (BOO), refer to OAB guidelines and storage symptom strategies.1,2,4 If symptoms are mainly obstructive or unresponsive to medication, surgical management should be considered. In all cases, treatment efficacy should be monitored, and if resolution is not achieved, alternative diagnoses such as sleep disorders should be explored.1,2,4
Pharmacologic therapies are indicated for patients with moderate-severe symptoms and those who perceive symptoms to be bothersome.1,2 For these patients, watchful waiting has been associated with untoward outcomes, including increased PVR urine volumes and voiding symptoms.18,19 There are many pharmacologic agents available for treating BPH, including alpha1 adrenergic antagonists, 5 alpha-reductase inhibitors, phosphodiesterase inhibitors, anticholinergic agents, and mirabegron. These will be described in detail below.
Alpha-1 Adrenergic Antagonists
The AUA guidelines suggest clinicians offer alpha-1 adrenergic antagonists as a treatment option to patients with bothersome, moderate-to-severe LUTS/BPH.1 The FDA-approved alpha-1 adrenergic antagonists, hereafter referred to as “alpha-blockers,” include alfuzosin, doxazosin, tamsulosin, terazosin, and silodosin.20-26 Alpha-blockers can be remembered as they end in the suffix “osin.”20-26 Alpha-blockers used in the management of BPH are listed in Table 4.
Table 4
Alpha-Blocker Dosing20-25
Generic (Brand) | Dosing |
| Alfuzosin (Uroxatral) | 10 mg once daily |
| Doxazosin (Cardura) | Immediate-release: 1 mg once daily. Titrate by doubling the dose up to 8 mg/day |
| Extended-release: 4 mg once daily in the morning. Titrate up to 8 mg once daily | |
| Silodosin (Rapaflo) | 8 mg once daily with a meal |
Tamsulosin (Flomax) | 0.4 mg once daily, 30 minutes before the next meal. Can increase to 0.8 mg once daily |
| Terazosin | 1 mg once daily at bedtime. Titrate slowly every few weeks to a usual dose of 10 mg once daily. The max dose is 20 mg once daily. |
Mechanism of Action and Dosing
Alpha-blockers facilitate smooth muscle tone by activating the alpha-1-adrenoreceptors in the sympathetic nervous system. 20-26 These medications block the alpha-1 adrenoreceptors in the prostate, prostatic urethra, and bladder neck.20-26 When these receptors are blocked, smooth muscle in the bladder and prostate relaxes, improving urinary flow rate and BPH symptoms.20-26 The choice of an alpha-blocker will depend on the patient’s age, comorbidities, and the side-effect profile of the selected agent.26 All alpha-blocking agents are comparable in terms of efficacy and begin to work a few hours or even days after administration.26
Administration
Administration varies among the alpha-blockers. Alfuzosin, tamsulosin, and silodosin should be taken within or right after the same meal each day.20,23,25 These capsules should never be crushed, chewed, or opened.20,23,25 Silodosin can be mixed with applesauce, and doxazosin and terazosin can be taken with or without food.20,23,25 After taking any alpha-blocker, patients should be cautioned against standing or moving suddenly, given the risk of orthostatic hypotension.20,23,25
Side Effects and Warnings
Common side effects of alpha-blocker therapy include asthenia (abnormal lack of energy), dizziness, fatigue, lassitude, orthostatic hypotension, palpitations, and peripheral edema.20-26 Patients may also experience nausea, impotence, drowsiness, and vertigo.20-26 The development of ejaculatory disorders is not uncommon.20-26
Contraindications to alpha-blocker therapy include hypersensitivity to these agents.20-26 Additionally, prostatic carcinoma should be ruled out prior to beginning treatment with an alpha-blocker to avoid masking the symptoms of progressive prostate cancer that need evaluation and management.20-26 Clinicians should also be aware of the many precautions associated with alpha-blocker use. This class of medications may cause CNS depression, impairing physical and mental abilities, and patients should be cautioned not to operate machinery or perform tasks that require mental alertness.20-26 Another considerable risk is the development of floppy iris syndrome. This may occur in patients who undergo cataract surgery and are currently taking or have previously taken alpha-blockers, and it involves poor pupil dilation and sudden pupil constriction.27 It is recommended that patients stop alpha blockers prior to surgery and then resume treatment once the surgery is complete.27
Alpha-blockers can also cause orthostatic hypotension or syncope to occur.28 This is especially common following the first dose and within the first few days of therapy. Increasing the dose of an alpha-blocker and taking an alpha-blocker concurrently with other antihypertensive drugs may also result in orthostatic hypotension. These effects are more common with doxazosin and terazosin than with alfuzosin, tamsulosin, or silodosin.28 Finally, these agents have the potential to cause priapism (prolonged, painful erection), though the risk is rare.28
Alpha-blocker Efficacy
Alpha-blockers have similar efficacy in treating BPH or LUTS, and numerous randomized controlled trials and systematic reviews support their use for these indications.18,19 Within these studies, no patient group has been shown to benefit more from alpha-blockers than any other.1,18,19 If the use of an alpha-blocker does not improve a patient’s symptoms, it is unlikely another agent will, given their similarities.1 However, it is prudent to change alpha blockers based on side effects.1 Tamsulosin, alfuzosin, and silodosin are less likely to cause orthostatic hypotension, whereas tamsulosin and silodosin are more likely to cause ejaculatory disorders.1
Patient Monitoring
An evaluation score (AUA or IPSS) should be taken at baseline and then 4-12 weeks after the start of treatment.20-26 Additionally, both objective and subjective signs of BPH and LUTS should be monitored throughout therapy. If alpha-blocker therapy is discontinued, it should be tapered with close monitoring of blood pressure.20-26
5-alpha Reductase Inhibitors
5-alpha reductase inhibitors (5ARIs) include dutasteride (Avodart®) and finasteride (Proscar®).29-31 The AUA guidelines recommend 5-ARI monotherapy as a treatment option in patients with LUTS/BPH with prostatic enlargement.1 Use of dutasteride and finasteride is reserved for patients with prostate volumes >30 mL, PSA >1.5 ng/dL, or palpable prostate enlargement on an exam.1 These agents can also be used alone or in combination with alpha-blockers to prevent the progression of LUTS or BPH and to reduce the risk of urinary retention and the need for future surgery.1
Mechanism of Action and Dosing
Dutasteride is an analog of testosterone and competitively and selectively inhibits 5AR types 1 and 2, blocking the conversion of testosterone to dihydrotestosterone and decreasing serum dihydrotestosterone levels.29 It is dosed at 0.5 mg once daily alone or in combination with an alpha-1-adrenergic antagonist.29 Patients should be counseled to swallow capsules whole.29
Finasteride competitively inhibits type 2 5-alpha reductase, thereby blocking the conversion of testosterone to dihydrotestosterone and decreasing dihydrotestosterone levels.31 It is dosed at 5 mg once daily, either alone or in combination with an alpha-1 adrenergic antagonist.31 Finasteride needs to be taken for at least 6-12 months to see symptom improvement.31 In general, the larger a patient’s prostate gland, the larger the reduction in prostate volume will be seen with 5ARI use.31
5-alpha reductase inhibitors differ in their pharmacokinetic properties. Finasteride has a half-life of between 3 and 16 hours, and dutasteride’s half-life is 5 weeks.29,31 Because of this, the adverse effects of dutasteride can persist even after the patient stops taking the medication.29 Additionally, 5ARIs have slow onsets of action, and patients should be counseled to expect slow improvement in symptoms. Both medications can be administered with or without food.29,31
Side Effects and Warnings
The frequency of adverse events with dutasteride therapy is highest in the first few months of therapy and tends to decrease after six months.29 Side effects include decreased libido, gynecomastia, increased testosterone levels, and increased thyroid-stimulating hormone levels.29 Impotence and ejaculation disorders are also possible.29
Healthcare providers should also recognize that because type II 5α-reductase inhibitors inhibit the conversion of testosterone to 5α-dihydrotestosterone (DHT), finasteride and dutasteride can cause abnormalities of the external genitalia of a male fetus.29,31 Caution is advised for females who handle these medications when pregnant or of childbearing potential to avoid broken tablets.29,31 Males taking these medications are urged to use condoms if the female partner is pregnant or of childbearing potential.29,31
Dutasteride should be used cautiously in patients who have diminished urinary flow and those with hepatic impairment.29 Interactions with CYP3A4 inhibitors (such as ketoconazole and erythromycin) can occur, and these should be used cautiously, if at all, when patients are taking dutasteride.29 Blood donation should also be avoided when patients are taking dutasteride to avoid transfusion to a pregnant recipient.29
Finasteride may cause various forms of sexual dysfunction, including impotence, decreased libido, ejaculatory disorders, and sexual disorders.27 These side effects may continue even after therapy has been discontinued, and sexual dysfunction side effects can result in anxiety, depression, and suicidal ideation.31 These effects are time and dose-related, and older patients and those taking finasteride for greater than 1 year are at the highest risk.31 Although rare, this post-finasteride syndrome should be counseled when initiating patients on this drug.31 Other possible adverse reactions include orthostatic hypotension, breast tenderness, decreased ejaculate volume, and high-grade prostate cancer.31 The AUA guidelines give a moderate recommendation to inform patients of the risks of sexual side effects and the low risk of prostate cancer before prescribing a 5ARI.1
Efficacy
A direct comparison of finasteride to dutasteride use was evaluated in one study.32 Men were randomized to either therapy for 12 months, and no differences in prostate volume, AUA-SI, and Qmax were noted.32 Alone, finasteride and dutasteride have demonstrated significant improvements in symptom scores compared to placebo.1,32
Monitoring
A baseline PSA level should be obtained before initiating therapy with a 5-alpha-reductase inhibitor, as these agents reduce PSA levels by approximately 50% and may mask rising PSA levels associated with prostate cancer.30,31 A repeat PSA should be obtained at least 6 months after initiation to establish a new baseline for ongoing prostate cancer screening and interpretation while the patient is on 5-AR therapy.30,31 PSAs should then be monitored regularly (typically annually) in therapy. The IPSS, urinalysis, and objective and subjective signs of BPH and LUTS should be observed.30,31
Muscarinic Receptor Antagonists
Muscarinic receptor antagonists decrease smooth muscle cell contractions in the bladder by inhibiting the muscarinic receptors.33,34 Agents in this category include tolterodine (Detrol® and Detrol LA®) and fesoteridine (Toviaz®).33-35 Tolterodine is dosed between 1-2 mg twice daily, Detrol LA at 4 mg daily, and fesoteridine between 4-8 mg daily.33,34 Xerostomia (dry mouth) is a common side effect of these medications.33,34 Muscarinic receptor antagonists are recommended for patients who have BPH with OAB symptoms.36
Phosphodiesterase-5 Inhibitor (PDE-5)
The AUA guidelines now recommend tadalafil, a PDE-5 inhibitor, for patients with LUTS/BPH, without consideration of comorbid erectile dysfunction.1 This is a newer recommendation within these guidelines. Tadalafil likely mediates a reduction in smooth muscle and endothelial cell proliferation, decreases nerve activity, and increases smooth muscle relaxation of the prostate and bladder.1 It is dosed at 5 mg once daily.37 If a patient is also taking finasteride, the duration of tadalafil treatment should be limited to 26 weeks.37
Side effects of tadalafil include potential hearing loss (with tinnitus and dizziness) that can occur within 12-24 hours of taking the drug.37 Hypotension may also occur, with reductions in blood pressure of up to 7 mmHg systolic and 5 mmHg diastolic seen.37 This is more likely when tadalafil is used with concurrent antihypertensives, alcohol, and in patients who are fluid-depleted or have autonomic dysfunction.37 Priapism has been reported with tadalafil use, though this is rare.37 It typically occurs within 24 hours of use.37
Mirabegron
Mirabegron (Myrbetriq®) is a beta-3 agonist.38 It activates the beta-3 adrenergic receptors in the bladder, which causes relaxation of the detrusor smooth muscle during the urine storage phase.38 This leads to an increase in bladder capacity.38 Mirabegron is approved for the treatment of overactive bladder symptoms and offers patients a treatment option with a lower rate of anticholinergic side effects.38,39 Mirabegron may be useful for treating OAB in men with BPH who are taking tamsulosin.38,39 A 2020 meta-analysis of a randomized controlled trial of 1217 BPH patients assessed the efficacy and safety of using mirabegron to treat OAB in men taking tamsulosin therapy.39 Mirabegron was found to decrease the mean number of micturitions per day significantly, the number of urgency episodes per day, the total OAB Symptom score, and the mean volume voided.39 Overall, it was found to be both safe and efficacious in this patient cohort, though it may increase the risk of increased post-void residual urine volume.39 For the treatment of OAB, mirabegron is dosed at 25 mg once daily.38 It may be increased to 50 mg once daily after 4-8 weeks if needed based on response and tolerability.38
Combination Therapy
The AUA guidelines provide different recommendations for combination therapy. First, 5-ARIs can be combined with alpha-blockers as a treatment option for patients with LUTS that are associated with prostatic enlargement, judged by prostate volume > 30 mL on imaging, a PSA >1.5 ng/dL, or palpable prostate enlargement.1 Second, anticholinergic medications can be used in combination with an alpha-blocker for patients with moderate-to-severe predominant storage LUTS.1 Beta-3 agonists can be combined with an alpha-blocker for patients with moderate-to-severe predominant storage LUTS.1 The guidelines note that low-dose tadalafil should not be combined with alpha-blockers for the treatment of LUTS/BPH because this combination offers no advantage in the improvement of symptoms versus the use of either medication alone.1
A combination product of dutasteride and tamsulosin is also available.40 Dosing for dutasteride-tamsulosin is 0.5 mg/0.4 mg once daily.40 This combination may help increase adherence versus taking both medications separately and has shown considerable efficacy as compared to monotherapy of either agent in improving symptoms and decreasing the progression of BPH.41 Adverse events mimic those seen in monotherapy and may be more pronounced.40,41
Nutraceuticals
Some clinical trial data have shown that various plant-based and herbal medications demonstrate efficacy in treating LUTS. However, current AUA guidelines do not offer extensive guidance on the use of nutraceutical and herbal products.1 Agents such as saw palmetto, stinging nettle, zinc, selenium, and others have been studied and demonstrated efficacy to an extent.1,42,43 Notably, these trials suffer from various limitations, including single-center studies, short durations, and poor methodology.42,43 Two double-blind, placebo-controlled studies evaluated the use of saw palmetto in terms of symptoms, bother, quality of life, flowrate recordings, serum PSA, and other measurable parameters.44,45 Both trials found no benefit of saw palmetto over placebo.44,45
Surgical Interventions and Emerging Therapies
If patients are experiencing serious complications because of BPH, surgical corrections should be offered.1,2 Drug therapy in these cases will only delay the worsening of BPH complications.2 Per AUA guidelines, surgery is recommended for patients with renal insufficiency secondary to BPH, refractory urinary retention secondary to BPH, recurrent urinary tract infections (UTIs), recurrent bladder stones or gross hematuria due to BPH, and/or with LUTS/BPH refractory to or unwilling to use other therapies.1 While evaluation for the presence of bladder outlet obstruction can be considered, clinicians should not perform surgery solely due to the presence of an asymptomatic bladder diverticulum.1
In recent years, the management of BPH has expanded to include a growing array of minimally invasive surgical treatments (MISTs), offering effective symptom relief with reduced recovery times and a lower risk of sexual side effects compared to traditional transurethral resection of the prostate (TURP).2,46 Options such as transurethral water vapor therapy (Rezūm), prostatic urethral lift (UroLift), temporary implantable nitinol device (iTind), prostate artery embolization (PAE), and the newer Optilume BPH balloon dilation system have shown promising outcomes in selected patient populations. These procedures vary in mechanism, invasiveness, and suitability based on prostate size, anatomy (e.g., presence of a median lobe), and patient preferences regarding catheter use, anesthesia, and preservation of ejaculatory function. While longer-term data are still emerging for some modalities, especially newer options like Optilume, clinical trials have demonstrated improvements in symptom scores and urinary flow rates, with certain MISTs also showing superiority in preserving sexual function.2,46 Careful patient selection and provider experience remain critical to optimizing outcomes and minimizing complications, making shared decision-making an essential part of BPH treatment planning.
Interprofessional Considerations
Effective management of BPH relies on an interprofessional approach that draws on the expertise of primary care providers, urologists, pharmacists, nurses, and other allied health professionals. Primary care clinicians often will conduct initial assessments and identify patients with LUTS.27,47 They play a key role in identifying risk factors, routinely monitoring patients, and determining when to refer patients for specialty care.
Urologists play a crucial role in confirming diagnoses, conducting advanced diagnostic evaluations, and providing surgical or procedural interventions when necessary. They typically manage more complex or refractory cases and perform procedures when needed. Urologists and PCPs are responsible for diagnosing BPH and determining its severity through a medical history, physical examination (including digital rectal exam), and diagnostic tests such as urinalysis, PSA blood test, cystoscopy, and ultrasound. Urologists should also collaborate with other specialists and the PCP. For example, if a patient is taking an alpha-blocker and has a history of cataracts or a planned cataract surgery, the urologist should discuss this with the ophthalmologist and PCP to determine if the patient should stop alpha blockers before surgery and then resume treatment once the surgery is complete.27
Pharmacists optimize medication therapy for patients with BPH, ensuring appropriate drug selection, dosing, monitoring for side effects, and drug interactions. Additionally, they are ideally positioned to counsel patients on how to take their medications correctly, potential adverse effects, and the importance of adherence to the prescribed regimen.
Nurses and care coordinators further support patient outcomes by facilitating education, follow-up, and care navigation. In complex cases, collaboration with endocrinologists, cardiologists, or geriatricians may be warranted to manage comorbidities that influence treatment choices or outcomes. Interprofessional communication is crucial for ensuring continuity of care, preventing medication-related complications, and aligning treatment plans with patients' preferences and goals. A team-based model fosters more holistic and personalized care, promotes early intervention, and ultimately improves quality of life for patients living with BPH.
Summary
Benign prostatic hyperplasia is a histological diagnosis and refers to the benign growth of prostate tissue. It is a common cause of LUTS, which often progresses in prevalence and severity as men age. Benign prostatic hyperplasia is associated with significant healthcare costs. The interdisciplinary team’s role in caring for patients with BPH is essential. Clinicians must evaluate the patient, consider a differential diagnosis, and provide a treatment plan that yields the best possible outcome. Treatment approaches should be individualized based on a patient’s symptoms (whether obstructive or overactive) and prostate size, the potential side effects of medication therapy, and the patient’s goals. The use of the IPSS is recommended to monitor the patient’s therapeutic progress, but there is insufficient evidence to support this recommendation.
References
Lerner LB, McVary KT, Barry MJ, et al. Management of lower urinary tract symptoms attributed to benign prostatic hyperplasia: AUA guideline part I, initial work‑up and medical management. J Urol. 2021;206(3):806–817.
Lee M, Sharifi R. Benign Prostatic Hyperplasia. In: DiPiro JT, Yee GC, Posey L, Haines ST, Nolin TD, Ellingrod V, eds. Pharmacotherapy: A Pathophysiologic Approach. 12th ed. New York, NY: McGraw‑Hill; 2023.
Nickel JC. Inflammation and benign prostatic hyperplasia. Urol Clin North Am. 2008;35(1):109-vii. doi:10.1016/j.ucl.2007.09.012
Sandhu JS, Bixler BR, Dahm P, et al. Management of Lower Urinary Tract Symptoms Attributed to Benign Prostatic Hyperplasia (BPH): AUA Guideline Amendment 2023. J Urol. 2024;211(1):11-19. doi:10.1097/JU.0000000000003698
Chung SD, Tzeng YM, Lin HC, Huang CY. Healthcare utilization and costs in patients with benign prostatic hyperplasia: a population‑based study. Asian J Androl. 2016;18(6):942–945. doi:10.4103/1008‑682X.167718
Roehrborn C. Benign prostatic hyperplasia and lower urinary tract symptom guidelines. Can Urol Assoc J. 2012;6(5 Suppl 2):S130–S132. doi:10.5489/cuaj.12231
Kobayashi S, Tang R, Shapiro E, et al. Characterization and localization of prostatic α₁‑adrenoceptors using radioligand receptor binding on slide‑mounted tissue sections. J Urol. 1993;150(6):2002–2006.
Patel ND, Parsons JK. Epidemiology and etiology of benign prostatic hyperplasia and bladder outlet obstruction. Indian J Urol. 2014;30(2):170–176. doi:10.4103/0970‑1591.126900
Wikimedia Commons. Benign Prostatic Hyperplasia image. Published 2005. Accessed April 15, 2025.
Roehrborn CG. Benign prostatic hyperplasia: an overview. Rev Urol. 2005;7(Suppl 9):S3–S14.
Meigs JB, Mohr B, Barry MJ, Collins MM, McKinlay JB. Risk factors for clinical benign prostatic hyperplasia in a community-based population of healthy aging men. J Clin Epidemiol. 2001;54(9):935-944. doi:10.1016/s0895-4356(01)00351-1
Miernik A, Gratzke C. Current Treatment for Benign Prostatic Hyperplasia. Dtsch Arztebl Int. 2020;117(49):843-854. doi:10.3238/arztebl.2020.0843
Unnikrishnan R, Almassi N, Fareed K. Benign prostatic hyperplasia: Evaluation and medical management in primary care. Cleve Clin J Med. 2017;84(1):53-64. doi:10.3949/ccjm.84a.16008
National Institutes of Health. National Cancer Institute. Prostate-Specific Antigen (PSA) Test. NIH-NCI Cancer.gov. January 31, 2025. Accessed August 10, 2025. https://www.cancer.gov/types/prostate/psa-fact-sheet
Welliver C, Feinstein L, Ward JB, et al. Trends in Lower Urinary Tract Symptoms Associated with Benign Prostatic Hyperplasia, 2004 to 2013: the Urologic Diseases in America Project. J Urol. 2020;203(1):171-178. doi:10.1097/JU.0000000000000499
Hollingsworth JM, Wilt TJ. Lower urinary tract symptoms in men. BMJ. 2014;349:g4474. Published 2014 Aug 14. doi:10.1136/bmj.g4474
Sarma AV, Parsons JK, McVary K, Wei JT. Diabetes and benign prostatic hyperplasia/lower urinary tract symptoms--what do we know?. J Urol. 2009;182(6 Suppl):S32-S37. doi:10.1016/j.juro.2009.07.088
Edwards JL. Diagnosis and management of benign prostatic hyperplasia. Am Fam Physician. 2008;77(10):1403-1410.
Lepor H. Evaluating men with benign prostatic hyperplasia. Rev Urol. 2004;6(Suppl 1):S8–S15.
UROXATRAL- alfuzosin hcl tablet, extended release. Prescribing Information. Advanz Pharma (US) Corp. December 5, 2024. Accessed March 18, 2026. https://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=9fc7f119-f36b-44cf-945d-940160f3afe3
DOXAZOSIN- doxazosin mesylate tablet. Prescribing Information. Aurobindo Pharma Limited. Updated September 6, 2024. Accessed March 18, 2026. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=93f2d724-5a54-4721-b04a-e46bc764607b
CARDURA- doxazosin mesylate tablet CARDURA- doxazosin tablet. Prescribing Information. ROERIG. January 20, 2022. Accessed March 18, 2026. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=eefc47bc-c10f-49bb-8ef7-5999f798ab39
TAMSULOSIN HYDROCHLORIDE capsule. Prescribing Information. Aurobindo Pharma Limited. November 17, 2023. Accessed March 18, 2026. https://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=8cb27f78-2d5d-4640-b7b5-ed06973c8133
TERAZOSIN- terazosin hydrochloride capsule. Prescribing Information. Jubilant Cadista Pharmaceuticals Inc. July 10, 2025. Accessed March 18, 2026. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=43e92495-2cf2-46dd-a80c-f0c3c88c7b73
SILODOSIN capsule. Prescribing Information. Lupin Pharmaceuticals, Inc. Updated October 7, 2025. Accessed March 18, 2026. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=2300db48-477f-4357-8de9-f723737071b4
Lokeshwar SD, Harper BT, Webb E, et al. Epidemiology and treatment modalities for the management of benign prostatic hyperplasia. Transl Androl Urol. 2019;8(5):529-539. doi:10.21037/tau.2019.10.01
Zaman F, Bach C, Junaid I, et al. The floppy iris syndrome - what urologists and ophthalmologists need to know. Curr Urol. 2012;6(1):1-7. doi:10.1159/000338861
Yu ZJ, Yan HL, Xu FH, et al. Efficacy and Side Effects of Drugs Commonly Used for the Treatment of Lower Urinary Tract Symptoms Associated With Benign Prostatic Hyperplasia. Front Pharmacol. 2020;11:658. Published 2020 May 8. doi:10.3389/fphar.2020.00658
AVODART- dutasteride capsule, liquid filled. Waylis Therapeutics LLC. March 12, 2025. Accessed March 18, 2026. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=960797b3-09b1-4d6a-8ece-8b5d918e93e4
PROSCAR- finasteride tablet, film coated. Prescribing Information. Organonon, LLC. December 23, 2024. Accessed March 18, 2026. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=6f988153-fc74-4ca4-b29a-111f750c4a4b
FINASTERIDE- finasteride tablet, film coated. Prescribing Information. Camber Pharmaceuticals, Inc. January 24, 2025. Accessed March 18, 2026. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=cc9709ce-2d29-48bd-b46b-3f72114f2527
Nickel JC, Gilling P, Tammela TL, Morrill B, Wilson TH, Rittmaster RS. Comparison of dutasteride and finasteride for treating benign prostatic hyperplasia: the Enlarged Prostate International Comparator Study (EPICS). BJU Int. 2011;108(3):388-394. doi:10.1111/j.1464-410X.2011.10195.x
DETROL- tolterodine tartrate tablet, film coated. Prescribing Information. Viatris Specialty LLC. Updated February 15, 2023. Accessed March 18, 2026. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=41eb00d2-64e3-4ec9-8732-4bfc81b04180
DETROL LA- tolterodine tartrate capsule, extended release. Prescribing Information. Viatris Specialty LLC. March 15, 2023. Accessed March 18, 2026. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=d7274947-4f88-47ee-a39f-bd5ac46fcf8d
TOVIAZ- fesoterodine fumarate tablet, film coated, extended release. Prescribing Information. Pfizer Laboratories Div Pfizer Inc. December 12, 2024. Accessed March 18, 2026. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=5be745f0-8ae7-4c3c-9962-37d6263326f1
Cameron AP, Chung DE, Dielubanza EJ, et al. The AUA/SUFU Guideline on the Diagnosis and Treatment of Idiopathic Overactive Bladder. J Urol. 2024;212(1):11-20. doi:10.1097/JU.0000000000003985
CIALIS- tadalafil tablet, film coated. Prescribing Information. Eli Lilly and Company. Updated December 30, 2025. Accessed March 18, 2026. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=bcd8f8ab-81a2-4891-83db-24a0b0e25895
MIRABEGRON tablet, extended release. Prescribing Information. Zydus Lifesciences Limited. October 9, 2024. Accessed March 18, 2026. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=461b2b94-205a-4860-9cb4-d30267ada9df
Su S, Lin J, Liang L, Liu L, Chen Z, Gao Y. The efficacy and safety of mirabegron on overactive bladder induced by benign prostatic hyperplasia in men receiving tamsulosin therapy: A systematic review and meta-analysis. Medicine (Baltimore). 2020;99(4):e18802. doi:10.1097/MD.0000000000018802
JALYN- dutasteride and tamsulosin hydrochloride capsule. Prescribing Information. Waylis Therapeutics. Updated March 9, 2026. Accessed March 18, 2026. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=aab9a2d4-5328-40f3-986f-e7753a928bc0
Wu Y, Davidian MH, DeSimone II EM. Guidelines for the Treatment of Benign Prostatic Hyperplasia. US Pharm. 2016;41(8):36-40.
Stewart KL, Lephart ED. Overview of BPH: Symptom Relief with Dietary Polyphenols, Vitamins and Phytochemicals by Nutraceutical Supplements with Implications to the Prostate Microbiome. Int J Mol Sci. 2023;24(6):5486. Published 2023 Mar 13. doi:10.3390/ijms24065486
Antoniou V, Gauhar V, Modi S, Somani BK. Role of Phytotherapy in the Management of BPH: A Summary of the Literature. J Clin Med. 2023;12(5):1899. Published 2023 Feb 28. doi:10.3390/jcm12051899
Bent S, Kane C, Shinohara K, et al. Saw palmetto for benign prostatic hyperplasia. N Engl J Med. 2006;354(6):557-566. doi:10.1056/NEJMoa053085.
Barry MJ, Meleth S, Lee JY, et al. Effect of increasing doses of saw palmetto extract on lower urinary tract symptoms: a randomized trial. JAMA. 2011;306(12):1344-1351. doi:10.1001/jama.2011.1364
Chiu PK, Siu BWH, De Nunzio C. Selecting minimally invasive surgical treatments for benign prostatic hyperplasia: A clinical consultation guide. Eur Urol Focus. 2025 May 9;S2405-4569(25)00101-4. doi:10.1016/j.euf.2025.04.025
Shea LA, Goldwire MA. Optimizing Urology Care With Clinical Pharmacists. US Pharm. 2025;50(6):HS7-HS12.
DISCLAIMER
The information provided in this course is general in nature, and it is designed solely to provide participants with continuing education credit(s). This course and materials are not meant to substitute for the independent, professional judgment of any participant regarding that participant’s professional practice, including but not limited to patient assessment, diagnosis, treatment, and/or health management. Medical and pharmacy practices, rules, and laws vary from state to state, and this course does not cover the laws of each state; therefore, participants must consult the laws of their state as they relate to their professional practice.
Healthcare professionals must consult their employer, healthcare facility, hospital, or other organization for guidelines, protocols, and procedures to follow. The information provided in this course does not replace those guidelines, protocols, and procedures, but is for academic purposes only, and this course’s limited purpose is for the completion of continuing education credits.
Participants are advised and acknowledge that information related to medications, their administration, dosing, contraindications, adverse reactions, interactions, warnings, precautions, or accepted uses is constantly changing. Any person taking this course understands that such a person must make an independent review of medication information before any patient assessment, diagnosis, treatment and/or health management. Any discussion of off-label use of any medication, device, or procedure is informational only, and such uses are not endorsed hereby.
Nothing contained in this course represents the opinions, views, judgments, or conclusions of RxCe.com LLC. RxCe.com LLC is not liable or responsible to any person for any inaccuracy, error, or omission with respect to this course or course material.
© RxCe.com LLC 2026: All rights reserved. No reproduction of all or part of any content herein is allowed without the prior, written permission of RxCe.com LLC.
RxCe.com
© RxCe.com LLC 2025: All rights reserved.

