FROM STIGMA TO SOLUTIONS: A COLLABORATIVE HEALTHCARE TEAM APPROACH TO ERECTILE DYSFUNCTION
Faculty:
L. Austin Fredrickson, MD, FACP
L. Austin Fredrickson is an Associate Professor of Internal Medicine at Northeast Ohio Medical University, where he serves as core faculty and teaches diagnostics, therapeutics, clinical skills, and health humanities. He is board-certified in general internal medicine and practices rural primary care.
Kristina (Tia) Neu, RN
Kristina (Tia) Neu is a licensed Registered Nurse and author currently developing in-service training for healthcare professionals. She is a National Board-Certified Health & Wellness and Lifestyle Medicine Coach. Her work experience includes several areas of the healthcare profession, such as psychiatric nursing, medical nursing, motivational health coaching, chronic case management, dental hygiene, cardiac technician, and surgical technician.
Liz Fredrickson, PharmD, BCPS
Liz Fredrickson, PharmD, BCPS, is an Associate Professor of Pharmacy Practice and Pharmaceutical Sciences at the Northeast Ohio Medical University (NEOMED) College of Pharmacy, where she is course director of the Parenteral Products and Basic Pharmaceutics Lab courses.
Pamela Sardo, PharmD, BS
Pamela Sardo, PharmD, BS, is a freelance medical writer and licensed pharmacist. She is the founder and principal at Sardo Solutions in Texas. Pam received her BS from the University of Connecticut and her PharmD from the University of Rhode Island. Pam’s career spans many years in retail, clinics, hospitals, long-term care, Veterans Affairs, and managed health care responsibilities across a broad range of therapeutic classes and disease states.
Abstract
Erectile dysfunction (ED) is the inability to maintain or achieve an erection satisfactory for intercourse. It is the most common cause of sexual dysfunction among men in the United States (US). An estimated 50% of men will experience some form of ED in their lifetime, making this a significant medical problem. Patients with ED require a patient-centered, holistic approach to care that involves treating underlying conditions and improving the quality and number of erections while avoiding medication side effects. Primary care providers are often the first to assess and initiate evaluation, making them critical in identifying underlying contributors such as diabetes, hypertension, or depression. Healthcare team members can also provide crucial counseling on the appropriate use of ED medications and help patients to mitigate side effects and avoid harmful drug interactions. This course will review the etiology and pathophysiology of ED and describe available treatment options in detail, including appropriate administration, mechanisms of action, side effects, and contraindications.
Accreditation Statements
In support of improving patient care, RxCe.com LLC is jointly accredited by the Accreditation CouncilTM for Continuing Medical Education (ACCME®), the Accreditation Council for Pharmacy Education (ACPE®), and the American Nurses Credentialing Center (ANCC®), to provide continuing education for the healthcare team.

This activity was planned by and for the healthcare team, and learners will receive 2 Interprofessional Continuing Education (IPCE) credits for learning and change.
Joint Universal Activity Number: The Joint Accreditation Universal Activity Numbers assigned to this activity are as follows:
Pharmacists: JA4008424-0000-26-030-H01-P
Pharmacy Technicians: JA4008424-0000-26-030-H01-T
Credits: 2 contact hour(s) (0.2 CEU(s)) of continuing education credit.
Credit Types:
IPCE Credits - 2 Credits
AAPA Category 1 Credit™️ - 2 Credits
AMA PRA Category 1 Credit™️ - 2 Credits
Pharmacy - 2 Credits
Type of Activity: Knowledge
Media: Computer-Based Training (i.e., online courses)
Estimated time to complete activity: 2 contact hour(s) (0.2 CEU(s)), including Activity Pre-Test, Post-Test, and Activity Evaluation.
Release Date: March 18, 2026 Expiration Date: March 18, 2029
Target Audience: This educational activity is for Physicians, Physician Assistants, Pharmacists, and Pharmacy Technicians
How to Earn Credit: From March 18, 2026, through March 18, 2029, participants must:
Read the “learning objectives” and “author and planning team disclosures;”
Take the “Educational Activity Pre-Test;”
Study the section entitled “Educational Activity;” and
Complete the Educational Activity Post-Test and Activity Evaluation. The Educational Activity Post-Test will be graded automatically. Following successful completion of the Educational Activity Post-Test with a score of 70% or higher, a statement of participation will be made available immediately. (No partial credit will be given.)
CME Credit: Credit for this course will be uploaded to CPE Monitor® for pharmacists. Physicians may receive AMA PRA Category 1 Credit™️ and use these credits toward Maintenance of Certification (MOC) requirements. Physician Assistants may earn AAPA Category 1 CME credit, reportable through PA Portfolio. All learners shall verify their individual licensing board’s specific requirements and eligibility criteria.
Statement of Need
Erectile dysfunction (ED) is highly prevalent, with up to half of men experiencing some degree of ED. Many men are uncomfortable initiating conversations about their sexual health. The reluctance to discuss ED causes many cases to go undiagnosed. This creates significant gaps in routine screening, risk factor modification, and identification of underlying conditions, such as cardiometabolic disease. Phosphodiesterase-5 inhibitors are first-line pharmacologic treatment; however, healthcare professionals are not uniformly aware of differences in onset, duration, food effects, contraindications, and adverse effects. Gaps also remain regarding second-line options, monitoring, and counseling, making an individualized approach to treatment and optimizing outcomes challenging. Erectile dysfunction management is often fragmented, when primary care, endocrinology, urology, and behavioral health should collaborate. This activity aims to address these gaps by covering epidemiology, comparing treatments, reinforcing monitoring, and clarifying the distinct roles of the healthcare team.
Learning Objectives: Upon completion of this educational activity, participants should be able to:
Describe the prevalence and underlying causes of erectile dysfunction
Compare and contrast phosphodiesterase-5 inhibitors
Recall indications, contraindications, side effect profiles, and monitoring parameters for erectile dysfunction treatments
Identify the roles of healthcare team members in the management of erectile dysfunction
Disclosures
The following individuals were involved in planning, developing, and/or authoring this activity: L. Austin Fredrickson, MD, FACP; Liz Fredrickson, PharmD, BCPS; Kristina (Tia) Neu, RN; and Pamela Sardo, PharmD, BS. None of the individuals involved in developing this activity has a conflict of interest or financial relationships related to the subject matter. There are no financial relationships or commercial or financial support relevant to this activity to report or disclose by RxCe.com or any of the individuals involved in the development of this activity.
© RxCe.com LLC 2026: All rights reserved. No reproduction of all or part of any content herein is allowed without the prior, written permission of RxCe.com LLC.
Educational Activity Pre-Test
Which of the following conditions is associated with a threefold increased risk of ED?
Asthma
Diabetes
Hypothyroidism
Gout
Which of the following PDE5 inhibitors is least affected by food intake?
Sildenafil
Avanafil
Vardenafil
All are equally affected
Which route of administration is most effective for alprostadil in treating erectile dysfunction?
Oral
Topical
Intraurethral
Intracavernosal
Educational Activity
From Stigma to Solutions: A Collaborative Healthcare Team Approach to Erectile Dysfunction
Introduction
Erectile dysfunction (ED) is the most common cause of sexual dysfunction among men in the United States. An estimated 50% of men will experience some form of ED in their lifetime, making this a significant medical problem. Despite the growing social awareness and acceptance of this disorder, many men remain hesitant to discuss issues related to ED with their partners and healthcare providers. This course will review the etiology and pathophysiology of ED and describe available treatment options in detail, including appropriate administration, mechanisms of action, side effects, and contraindications.
Erectile Dysfunction: An Overview
Erectile dysfunction is the inability to maintain or achieve an erection satisfactory for intercourse.1-5 Patients with ED require a patient-centered, holistic approach to care that involves treating underlying conditions and improving the quality and number of erections while avoiding medication side effects. Further, ED is too frequently not prioritized over other health issues. This is concerning from a public health perspective, given that ED often results from serious underlying conditions, including metabolic syndrome and cardiovascular disease.
Primary care providers are often the first to assess and initiate evaluation, making them critical in identifying underlying contributors such as diabetes, hypertension, or depression. Pharmacists also play an important role in identifying men who require ED treatment, making referrals, and helping the care team address medication-related causes of ED. Further, healthcare team members can collaborate and provide crucial counseling on the appropriate use of ED medications and help patients to mitigate side effects and avoid harmful drug interactions.
Etiology and Risk Factors
Erectile dysfunction can result from a number of causes, which may be vascular, neurologic, hormonal, and/or psychological in nature.1 In many cases, the etiology is multifactorial, and a thorough patient evaluation is essential to uncover contributing conditions. More than 80% of ED cases in older men are linked to diabetes, atherosclerosis, or medication use.1 Table 1 outlines selected causes of ED by category.1
Table 1
Selected Causes of Erectile Dysfunction1
| Etiology | Condition |
| Neurologic | Multiple sclerosis Stroke |
| Hormonal | Hypogonadism Thyroid disorders |
| Trauma | Spinal cord injuries |
| Metabolic | Diabetes mellitus |
| Psychologic | Depression Anxiety Post-traumatic stress disorder Substance use disorders |
| Other | Sleep apnea COPD CHF Urologic disorders Medications Glaucoma (correlated) |
The likelihood of developing ED increases with age, affecting approximately 52% of men between the ages of 40 and 70 and up to 80% of men older than 70.2,5 Importantly, this increased prevalence is not only due to aging, but also results from the development of chronic conditions and the use of medications that can impair erectile function.2 ED and metabolic syndrome share a well-established connection, and ED is increasingly recognized as an early warning sign of cardiovascular disease (CVD).1,3 Many of the same risk factors for CVD—including hypertension, smoking, and obesity—also contribute to ED.3 Notably, men with diabetes are nearly three times more likely to experience ED than those without the condition.4 Psychological comorbidities, including anxiety, depression, and post-traumatic stress disorder (PTSD), can also independently cause ED or exacerbate symptoms associated with other medical conditions.1,2
Medications contribute to approximately 25% of ED cases, and pharmacists can support care teams by identifying drugs that may trigger or worsen symptoms.2,4 To determine if a particular drug is a cause of ED, pharmacists should look for an association between the start of the medication and the onset of ED.1 A list of select medications associated with ED is detailed in Table 2.2
Table 2
Medications that Cause Erectile Dysfunction2
| Medication Class | Examples | Mechanism |
| Diuretics and antihypertensive agents | Hydrochlorothiazide Benazepril Chlorthalidone Propranolol | Reduce blood flow to the penis and may interfere with the nerve signals or hormone levels needed for an erection |
| Antidepressants | Fluoxetine Sertraline Citalopram Paroxetine | Selective serotonin reuptake inhibitors (SSRIs) may impair libido, delay orgasm, and inhibit nitric oxide synthesis, which is critical for erection |
| Antihistamines | Diphenhydramine Meclizine Promethazine | Anticholinergic effects can impair nerve signaling and reduce libido, contributing to erectile dysfunction |
| NSAIDs | Naproxen Indomethacin | May cause ED by affecting prostaglandin synthesis and endothelial function, especially with chronic use |
| H2-receptor antagonists | Cimetidine Famotidine | May increase prolactin levels and interfere with androgen metabolism, leading to decreased testosterone and libido |
Antidepressants are one of the most notorious classes known to cause ED, in particular the selective serotonin reuptake inhibitors (SSRIs) citalopram, fluoxetine, paroxetine, and sertraline.2,4 Illicit drug use, tobacco, and the use of alcohol can worsen ED.4 Use of antipsychotics and finasteride can also result in erectile dysfunction.6,7
Pathophysiology of Erectile Dysfunction
A clear understanding of penile erection physiology is essential for evaluating ED and selecting appropriate treatment modalities. Penile erection is a neurovascular event that requires a coordinated interaction between the central nervous system (CNS), peripheral nervous system (PNS), and the vascular and smooth muscle tissues of the penis (Figure 1).1,8,9 The penis is comprised of the corpora cavernosa and the corpus spongiosum, which play a role in the erectile response.1,9 (See Figure 1.) For an erection to occur, the smooth muscle of the corpora cavernosa must relax, allowing blood to flow into the penis.1,9 As arterial inflow increases, intracavernosal pressure rises, and venous outflow is compressed, helping to maintain the erection.1,9
Figure 11,9
Anatomy of the Male Reproductive System

To achieve an erection, the CNS must coordinate sensory information from visual, auditory, tactile, and imaginative sources.9 The PNS then releases nitric oxide from parasympathetic nerve terminals (Figure 2).1,9,10 Nitric oxide activates the enzyme soluble guanylate cyclase (sGC), which catalyzes the conversion of GTP to cyclic guanosine monophosphate (cGMP).1,9 The rise in cGMP leads to smooth muscle relaxation in the corpus cavernosum, allowing further arterial blood inflow.9 cGMP is eventually degraded by phosphodiesterase-5 (PDE-5), which is the target of many pharmacologic treatments for ED.9 Additional support comes from vasoactive prostaglandins, including PGE1 and PGF2α, which also promote smooth muscle relaxation.1,9
Figure 21,9
Nitric Oxide Pathway
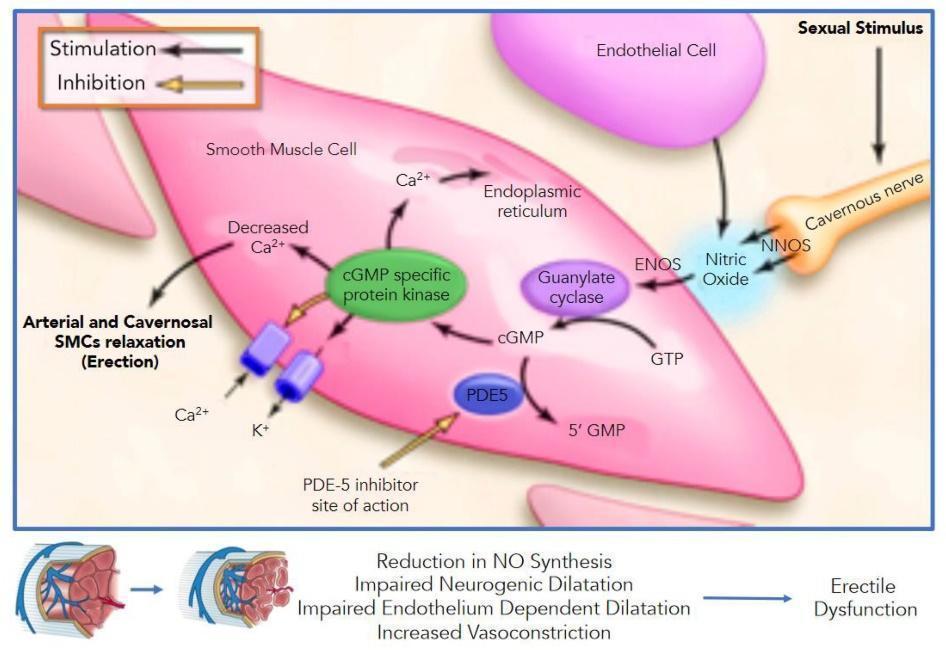
Erectile dysfunction typically results from one or more of the following mechanistic failure:1
Failure to initiate – often psychogenic, neurogenic, or hormonal in origin
Failure to fill – due to impaired arterial inflow (e.g., atherosclerosis)
Failure to store – related to veno-occlusive dysfunction, where blood cannot be retained in the corpora cavernosa
The most common underlying mechanism is endothelial dysfunction, which leads to reduced nitric oxide synthesis or activity.1 This impairs smooth muscle relaxation and blood flow, ultimately resulting in ED.1
Clinical Pearl Because endothelial dysfunction is a key driver of ED, its presence should prompt consideration of cardiovascular risk assessment and preventive strategies.10 |
Diagnosing Erectile Dysfunction
The diagnosis and management of ED require a coordinated, interprofessional approach. Primary care providers (PCPs) are often the first to assess and initiate evaluation, making them critical in identifying underlying contributors such as diabetes, hypertension, or depression. Cardiologists play a particularly vital role, especially when considering pharmacologic therapy, as ED can be an early warning sign of cardiovascular disease. Before prescribing agents such as phosphodiesterase type 5 (PDE5) inhibitors, it is essential to determine if the patient has undergone a recent cardiac evaluation, particularly for older adults or those with a known cardiac history. The cardiologist may be best positioned to assess whether sexual activity and ED pharmacotherapy are safe for the patient. Care team members can help identify medication-induced ED, recommend safer alternatives, counsel patients on proper medication use, and support adherence. Additional specialists, including endocrinologists (for hormonal imbalances), urologists (for treatment-resistant cases), and mental health professionals (for psychogenic factors), also contribute meaningfully to a patient-centered, comprehensive ED management plan. Collaboration across disciplines ensures that ED is not treated in isolation but rather as part of a broader strategy to improve physical, emotional, and sexual well-being. Clear communication among team members—and with the patient—is vital for coordinated care, reducing stigma, and ensuring personalized, evidence-based management.
The diagnosis of ED is primarily clinical and should begin with a comprehensive medical, sexual, and psychosocial history.11 According to the American Urological Association (AUA) guidelines, clinicians are encouraged to proactively initiate this conversation, as many patients may feel embarrassed or reluctant to bring up concerns related to sexual function.11 Additionally, a full review of the patient’s medication list—including prescription, over-the-counter, and recreational substances—is essential, as many drugs are known contributors to ED.11
To quantify ED severity and assess treatment response over time, clinicians should incorporate validated psychometric tools.11 Among these, the most widely used tools include the International Index of Erectile Function (IIEF), its abbreviated version IIEF-5 (also known as the Sexual Health Inventory for Men or SHIM), and the Erection Hardness Scale (EHS).12–15 The full IIEF evaluates five domains: erectile function, orgasmic function, sexual desire, intercourse satisfaction, and overall satisfaction.12 The IIEF-5 focuses specifically on erectile function and is often used in both clinical and research settings.12 The EHS, a single-item scale, allows patients to rate their erection hardness on a scale of 1 (penis is larger but not hard) to 4 (completely hard and fully rigid).15 These tools provide a practical and evidence-based method to define ED severity and monitor progression or improvement with treatment.12
A comprehensive physical examination should include a genital exam, blood pressure measurement, and assessment of body mass index (BMI). Clinicians should evaluate for signs of hypogonadism, such as low libido, fatigue, or decreased muscle mass, as well as indicators of vascular disease, given their strong associations with ED. Because ED is often multifactorial, it is also essential to screen for psychosocial contributors, including depression, anxiety, and substance use, all of which can exacerbate or underlie sexual dysfunction. Laboratory and diagnostic testing should be guided by clinical suspicion. Table 3 below outlines standard evaluations and their purposes.16
Table 3
Evaluation of Causes of Erectile Dysfunction
| Test | Purpose |
| Fasting glucose or hemoglobin A1C | Evaluate glycemic control and screen for diabetes |
| Lipid panel | Assess cardiovascular risk factors |
| Thyroid-stimulating hormone (TSH) | Screen for thyroid dysfunction |
| Morning serum total testosterone | Recommended when hypogonadism is suspected (e.g., low libido, fatigue) |
Advanced diagnostic evaluations are typically reserved for patients with unclear etiology or those who do not respond to first-line therapy.15 Routine imaging is not generally recommended, as most modalities have limited diagnostic value.15 For instance, penile duplex Doppler ultrasound may aid in identifying vasculogenic ED but is constrained by non-standardized cutoff values for arterial dysfunction and low predictive utility for diagnosing corporal veno-occlusive dysfunction (CVOD).15
Experimental imaging approaches such as penile MRI and penile shear wave elastography are being studied but have not yet been validated for routine clinical use.15 Nocturnal penile tumescence and rigidity testing (NPTR), although historically limited by cost and technical complexity, is regaining attention due to the emergence of wireless, home-based devices.15 These innovations allow for convenient, less invasive overnight monitoring, potentially improving differentiation between psychogenic and organic causes of ED.15
Wearable health technologies are also gaining traction among patients interested in tracking their sexual function.15 These devices measure physiological parameters in real-time.15 One example is the Tech Ring, which functions as both a penoscrotal constriction ring and a sexual health monitor.15 It uses embedded sensors to track erection characteristics during sexual activity and at night; however, the constriction ring may affect the natural erectile process, potentially influencing the accuracy of its measurements.15 The ADAM sensor is another ring-like device that measures nocturnal penile tumescence and may become a valuable tool for remote monitoring.15 While these technologies are not yet a replacement for clinical evaluation, they may serve as adjuncts that enhance patient engagement and support ongoing assessment, particularly in telehealth settings.
Evidence-Based Management of Erectile Dysfunction
AUA Reaffirmation
As of February 2026, the AUA has not released a formal update to the 2018 Erectile Dysfunction: AUA Guideline; however, it has reaffirmed and clarified existing recommendations through continued educational dissemination.17 They continue to emphasize a patient-centered approach, recommending shared decision-making as a critical component of ED management.15,17 Clinicians are encouraged to present all appropriate treatment options—ranging from lifestyle modifications to oral pharmacotherapy, injections, vacuum devices, and surgical interventions—without adhering to a stepwise progression from least to most invasive.17 This flexibility allows treatment to be tailored based on patient preference, clinical appropriateness, and informed consent.
The AUA also highlights the importance of addressing psychological contributors to ED.17 Mental health referrals and behavioral counseling are particularly valuable for patients experiencing performance anxiety, depression, or relationship stress, all of which can contribute to or exacerbate ED.17 The guidelines support integration of mental health professionals and sex therapists into the care team when appropriate.17 Overall, the 2023 reaffirmation underscores a multidisciplinary, individualized approach that recognizes ED as a complex condition influenced by physical, psychological, and lifestyle factors.17
Goals of Treatment
The primary goals of treating ED are threefold: 1) to enhance the quality and reliability of erections to a degree satisfactory to the patient and their partner(s), 2) to improve the patient’s overall physical health, and 3) to optimize quality of life and psychosocial well-being.¹ Achieving these goals requires a patient-centered, evidence-based, and individualized approach. Clinicians must establish realistic expectations with patients through shared decision-making, ensuring that therapeutic goals, potential limitations, and the range of treatment options are well understood.1 This process improves adherence, satisfaction, and long-term outcomes. Treatment should also be tolerable, effective, and affordable, and tailored to the patient’s values, lifestyle, and comorbidities.
Treating Psychogenic ED
In addition to well-characterized organic causes, psychogenic ED remains a critical but frequently overlooked contributor.11 For patients whose ED is primarily psychological in origin—or where psychological comorbidities such as anxiety or depression are contributing—management should involve addressing underlying mental health concerns.11 This may include psychological counseling, cognitive behavioral therapy (CBT), sex therapy, and, when appropriate, pharmacologic treatment for underlying mood disorders.11 Although a detailed review of psychogenic ED strategies is beyond the scope of this paper, clinicians should remain vigilant and refer patients to mental health professionals when needed.
Lifestyle Interventions as a Foundation of ED Management
Implementation of lifestyle changes is critical in both preventing and managing ED.11,15 According to the AUA guidelines, these interventions should be encouraged before initiating pharmacotherapy, as they support general vascular and endocrine health.
Recommended lifestyle modifications include the following:11,15,17
Maintaining a healthy weight
Engaging in regular aerobic and resistance exercise
Adopting a well-balanced, heart-healthy diet (e.g., Mediterranean diet)
Managing stress and mental health
Quitting tobacco use
Reducing excessive alcohol consumption
Improving sleep quality and duration
These changes positively influence endothelial function, nitric oxide availability, testosterone levels, and psychological well-being—all of which are integral to erectile function.11 Furthermore, smoking cessation can reduce ED risk significantly. Current smokers face a >50% increased risk of ED, and even former smokers retain a ~20% elevated risk.4 In patients with hyperlipidemia or elevated cardiovascular risk, statin therapy may offer dual benefits by improving both cardiovascular outcomes and International Index of Erectile Function (IIEF-5) scores.18
When lifestyle optimization alone does not produce sufficient improvement, pharmacologic treatments can be initiated. While the mainstays of ED treatment have generally remained the same, patients have several new management strategies as well (Table 4). Primary treatment options include phosphodiesterase type 5 inhibitors (PDE5is) and prostaglandin E1 analogs. These medications are discussed in more detail in the next section. In general, an ideal pharmacologic treatment is safe, efficacious, fast-acting, and cost-effective.² Importantly, treatment plans should be reassessed regularly in collaboration with the patient and involve a diverse, interprofessional healthcare team to optimize outcomes.
Table 4
Current and Emerging ED Treatment Options19
| Treatment Category | Subcategory | Examples |
|---|---|---|
| Current Treatments | Oral Therapy | - Phosphodiesterase type 5 inhibitors: - Sildenafil - Tadalafil - Vardenafil - Avanafil |
| Mechanical/Physical Treatment | - Vacuum erection device - Low-intensity extracorporeal shockwave therapy | |
| Local Vasoactive Agents | - Intracavernosal injection: - Papaverine - Prostaglandin E1 - Phentolamine - Vasoactive intestinal peptide - Intraurethral prostaglandin E1 | |
| Emerging Treatments | Novel and Emerging Oral Therapies | - Rho-kinase inhibitors - Melanocortin receptor agonists - Maxi-K+ channel activators - Guanylate cyclase activators - Guanylate cyclase stimulators - Nitric Oxide donors |
| Emerging Local Treatments | - Stem cells - Platelet-rich plasma - Botulinum neurotoxin - Glyceryl trinitrate-based gel | |
| Emerging Physical Treatments | - Low-intensity radiofrequency |
Phosphodiesterase-5 Inhibitors
Mr. R is a 58-year-old man with a history of hypertension and type 2 diabetes mellitus, well-controlled on lisinopril and metformin. He presents to his primary care provider with concerns about difficulty achieving and maintaining an erection sufficient for sexual activity over the past 6 months. He reports that the issue has gradually worsened and is now affecting his relationship with his partner. He is not on any medications known to interfere with sexual function. His morning testosterone level is within normal range, and he is not currently experiencing depressive symptoms. He expresses interest in treatment options but is unsure where to begin. He is sexually active, has no contraindications to sexual activity, and is not taking nitrate medications.
Pause and Ponder What are possible treatment options for Mr. R? What are the possible side effects? What are the patient counseling points? |
Phosphodiesterase-5 inhibitors remain the cornerstone of pharmacologic therapy for ED, given their efficacy and safety profiles.11,17 Sildenafil was initially studied for coronary artery disease but was found to enhance erectile function during trials.20 This discovery led to the approval of sildenafil (Viagra®) for ED treatment in 1998, followed by the introduction of tadalafil (Cialis®), vardenafil (Levitra® and Staxyn®), and avanafil (Stendra®) over the subsequent 14 years.20 Sildenafil became available in generic form in 2017, increasing accessibility and flexibility in dosing.20
Mechanism of Action and Pharmacokinetics
Phosphodiesterase-5 inhibitors do not directly induce erections; instead, they enhance the natural erectile response to sexual stimulation by preventing the breakdown of cyclic guanosine monophosphate (cGMP).20 Upon sexual stimulation, nitric oxide is released in the corpus cavernosum, activating guanylate cyclase and increasing cGMP levels.20 PDE5Is block the PDE5 enzyme responsible for cGMP degradation, promoting smooth muscle relaxation, vasodilation, and increased penile blood flow.20 These agents are ineffective without sexual stimulation and may be less effective in men with severe ED, such as those with post-radical prostatectomy or significant vascular damage.20
Pharmacokinetic profiles vary significantly across agents. For example, avanafil has a rapid onset of action (15-30 minutes). In contrast, tadalafil has a long duration of action (up to 36 hours) and is suitable for both on-demand and once-daily dosing.21-24 Tadalafil and avanafil are less affected by food intake, whereas high-fat meals can delay the onset of action of sildenafil and vardenafil.23,24 Table 5 lists the pharmacokinetic properties of PDE5 inhibitors.
Table 5
Pharmacokinetic Properties of PDE5 Inhibitors21-24
Generic (Brand) | Onset | Duration | Metabolism | Effect of food intake |
| Sildenafil | 30-60 mins | Up to 12 hours | Hepatic via CYP3A4 and CYP2C9 | High-fat meals decrease efficacy |
| Avanafil | 15-30 min | Up to 6 hours | Hepatic via CYP3A4 and CYP2C9 | Not affected |
| Tadalafil | 1-2 hours | Up to 36 hours | Hepatic via CYP3A4 | Not affected |
| Vardenafil | 30-60 mins | Up to 10 hours | Hepatic via CYP3A4 and CYP2C9 | High-fat meals decrease efficacy |
Dosing and Optimization
Initial and maximum doses vary among agents and are summarized in Tables 6 and 7.21-24 Table 8 includes examples of considerations for concomitant use with medications affecting the cytochrome P 450 system. In general, dose titration should be individualized based on efficacy and tolerability.11 Using the lowest effective dose minimizes side effects. Doses should be titrated up or down based on patient response and side effects until an optimal dose is reached.11 Dose reductions or medication weaning can be considered in men with ED of psychogenic origin once related issues have been resolved.11 Generic versions of sildenafil are also available in 20mg tablets (historically used for pulmonary hypertension dosing), but using these for ED may not only be more affordable but also allow for greater flexibility when titrating.11
Table 6
Dosing of Phosphodiesterase-5 Inhibitors21-24
| Generic (Brand) | Initial Dose | Max Dose | Dose Reduction |
| Sildenafil | 50 mg once daily as needed 1 hour before sexual activity | 100 mg | 25 mg |
| Avanafil | 100 mg 15 minutes before sexual activity | 200 mg | 50 mg 30 minutes before sexual activity |
| Tadalafil | 10 mg 30 minutes or more before sexual activity | 20 mg | 5 mg |
| Tadalafil | 2.5 mg daily | 5 mg | --- |
| Vardenafil | 10 mg 1 hour before sexual activity | 10 mg per day | --- |
| Vardenafil | 10 mg 1 hour before sexual activity | 20 mg per day | 5 mg |
Table 7
PDE5i Drug Dosing with CYP3A4 Inhibitors21-24
| Agent | Modified Dose |
| Sildenafil | 25 mg |
| Vardenafil | 2.5 mg every 24 hours |
| Vardenafil (Staxyn) | Avoid use |
| Tadalafil (on demand) | 10 mg every 72 hours |
| Tadalafil (daily) | 2.5 mg |
| Avanafil | Avoid use with potent inhibitors. Limit dose to 50 mg every 24 hours with moderate inhibitors. |
Adverse Effects
A significant benefit of PDE5is is their mild side-effect profiles.21-24 Common adverse effects include dyspepsia, headache, flushing, back pain, myalgia, and dizziness.21-24 While rates of dyspepsia and dizziness are similar among these medications, sildenafil and vardenafil are associated with higher rates of headache and flushing.11 Use of daily dosing of tadalafil is associated with lower rates of adverse effects compared to on-demand use.11 Men who are post-radical prostatectomy and post-radiation therapy may experience higher rates of adverse effects than those in the general ED population.11
More serious adverse effects of PDE5is include prolonged erections (erections lasting longer than 4 hours), priapism (prolonged, painful erections), and nonarteritic anterior ischemic optic neuropathy (NAION).21-24 NAION is a rare condition in which vision loss in one eye occurs suddenly. Risk factors include men older than 50, small optic discs with a low cup-to-disc ratio, and Caucasian ethnicity.21 The absolute risk is small (3 cases per 100,000 men 50 years or older), but patients should be counseled on how to recognize NAION.11,18 Sildenafil is also associated with other notable visual side effects, including blue-tinged vision, increased light sensitivity, and blurred vision. These have been reported in 3–11% of patients taking standard doses (25–100 mg) and in up to 100% of patients at doses exceeding 600 mg.25 These symptoms are typically mild and transient, though isolated reports describe persistence for up to 21 days.25 While changes in color perception—especially in the blue-green spectrum—have been observed, studies in both healthy individuals and those with erectile dysfunction or age-related macular degeneration generally show no significant or lasting impairments in overall visual function.25
It is also important for the healthcare team to counsel patients on the drug-food interactions associated with PDE5is. Most notably, men prescribed regular use of nitrates should not use PDE5is, as this may result in large drops in blood pressure.21-24 If sublingual nitroglycerin is used for angina, PDE5i use should be avoided for 24 hours and longer for tadalafil.21-24 Other longer-acting nitrate agents (isosorbide mononitrate/dinitrate, etc.) should similarly be avoided.18-24
Additionally, patients taking sildenafil should avoid administration with high-fat foods as this can decrease absorption.21 The concurrent use of grapefruit juice should also be avoided.21 Vardenafil (Levitra) should be avoided with concurrent use of Type 1A or Type 3 antiarrhythmics and is not recommended if patients have congenital prolonged QT intervals.24
Patient Monitoring and Counseling
Clinicians can assess the efficacy of PDE5i therapy after a trial of several weeks using a single agent.11 The degree of satisfaction with any agent is patient-specific, so patients must be highly involved in shared decision-making. Importantly, treatment failures have been noted due to unrealistic expectations, relationship dynamics, and performance anxiety, and these reasons should be explored if patients are not seeing positive effects.26 If patients still do not see improvement in the quality and quantity of erections, the dose can be increased (up to the maximum tolerated dose), or the patient can be switched to a different drug.11 Other treatment modalities may also be explored.9 General monitoring parameters for PDE5is include monitoring blood pressure and heart rate in those concurrently using antihypertensive agents and monitoring for pulmonary edema.21-24
Healthcare Professional Patient Counseling Tips:
Ask if they are receiving prescription or over-the-counter products from an online company.
Do not use more than prescribed.
Store at room temperature or as instructed.
These treatments are not FDA-approved for use by females.
PDE5i Efficacy
The PDE5is have been studied extensively, with research including men in the general ED population and those with comorbidities such as diabetes, benign prostatic hyperplasia, and post-spinal cord injury.11 In general, these agents are considered equally efficacious.11 Within the literature, the IIEF is commonly used to evaluate efficacy, and the magnitude of changes from pre-treatment to post-treatment scores is similar among all agents.11 There is notably limited data comparing these agents to one another. A 2013 review of 118 trials found PDE5is were superior to placebo in improving ED.27 Avanafil was less effective compared to tadalafil and vardenafil for question 1 on the Global Assessment Questionnaire.27 Within the erectile function domain of the IIEF, tadalafil was found to be more effective than vardenafil.27 No major differences were noted for the agents in terms of safety.27 Within this review, tadalafil was found to be the most effective PDE5i.27 Comparisons within this review were determined to be of low-grade evidence due to poor original reporting and indirect comparisons.27
In a 2017 meta-analysis, tadalafil and sildenafil were directly compared for the treatment of ED. Sixteen trials included in the analysis found that tadalafil and sildenafil were similarly efficacious and had the same rate of side effects.28 Tadalafil was associated with lower rates of flushing, but higher rates of back pain and myalgia compared to sildenafil.28 Tadalafil improved psychological outcomes over sildenafil, and patients also preferred tadalafil over sildenafil.28 There were no differences in adherence rates between the two medications.28 This analysis suggests tadalafil may be a preferred choice for patients, but the choice should be patient-driven.
Emerging and Nontraditional Benefits
PDE5Is may confer additional systemic benefits beyond ED treatment.15 Evidence suggests potential renoprotective effects in patients with chronic kidney disease, improvements in heart failure symptoms through reduced myocardial stress, and even possible roles in slowing cognitive decline.15 Emerging studies also suggest immunomodulatory and anticancer properties, though larger trials are needed to confirm these findings.15
Prostaglandin E1
Alprostadil, a synthetic analog of prostaglandin E1, is a well-established second-line treatment for ED. It is marketed under several brand names, including Caverject, Caverject Impulse, Edex, and Muse.¹ Alprostadil is particularly useful for men who are contraindicated for phosphodiesterase-5 inhibitors (PDE5Is), do not tolerate them well, or prefer non-oral treatment options.² While approximately 60% of men respond to PDE5Is, efficacy may decline over time, making alprostadil a valuable alternative.²
Mechanism of Action and Dosing
Alprostadil, a synthetic analog of prostaglandin E1, is a well-established second-line treatment for erectile dysfunction ED.29 Marketed under brand names such as Caverject, Caverject Impulse, Edex, and Muse, alprostadil is particularly useful for men who are contraindicated for phosphodiesterase-5 inhibitors (PDE5Is), do not tolerate them well, or prefer a non-oral medication route.29 While around 60% of men respond to PDE5Is, this responsiveness may diminish as ED progresses, making alprostadil a valuable therapeutic option.29
Alprostadil induces erections by directly stimulating adenylate cyclase, increasing intracellular cyclic AMP (cAMP), and relaxing smooth muscle in the corpus cavernosum.29 This leads to enhanced arterial inflow and venous occlusion, facilitating penile rigidity. Alprostadil is administered either via intracavernosal injection (ICI) or intraurethral application (IU), and its efficacy varies depending on the delivery route, the underlying cause of ED, and patient-specific factors.29
Intracavernosal Administration
Intracavernosal injection of alprostadil requires initial dose titration in a healthcare setting.29 If the initial dose does not induce an erection, a second dose can be administered within one hour; however, if still ineffective, treatment should be paused for 24 hours before the next attempt. The goal is to identify the lowest effective dose, as higher doses are associated with increased discomfort and risk of adverse effects.29 Initial dosing typically starts at 2.5 mcg for vasculogenic or mixed etiologies and 1.25 mcg for neurogenic causes.29 Dose titration involves incremental increases every 24 hours until an optimal response is achieved.29 Once an effective dose is established, patients may self-administer injections up to three times per week, with at least 24 hours between doses.29
Administration technique is critical for safety and efficacy. For Caverject, Caverject Impulse, and Edex, the injection should be made into the dorsolateral aspect of the proximal third of the penis using a 27–30 gauge, ½-inch needle, avoiding visible veins and alternating injection sites. Post-injection compression with a sterile swab for five minutes helps minimize bleeding and hematoma formation.29 Caverject Impulse is supplied as a prefilled dual-chamber syringe, which simplifies the injection process, while Edex requires manual reconstitution. Injection should occur over 5–10 seconds to improve comfort.29
Intraurethral alprostadil, marketed as Muse, involves inserting a medicated pellet directly into the urethra using a specialized applicator.29 Initial dosing ranges from 125 to 250 mcg and may be adjusted up or down to achieve a satisfactory erection.29 For best results, administration should occur immediately after urination, which lubricates the urethra and enhances drug absorption.29 The penis should be gently stretched to full length, and the applicator stem inserted into the urethra up to the collar.25 The button is then pressed to release the medication, and the device is left in place for five seconds before removal.29 After administration, the penis should be held upright and gently rolled between the hands for 10 seconds to facilitate dispersion. Patients are encouraged to walk or stand for approximately 10 minutes after administration to enhance penile blood flow further.29
Table 8
Alprostadil Intracavernosal Dosing29
| Etiology | Initial Dose | Titration | Maintenance Dose |
| Vasculogenic, psychogenic, mixed etiology | 2.5 mcg | Partial response: give another 2.5 mcg dose. Can be titrated in increments of 5-10 mcg 24 hours apart until an adequate response is reached No response: give second dose of 7.5 mcg within 1 hour | Once an appropriate dose is reached, the patient can self-administer injections no more than 3 times/week, with 24 hours minimally in between doses |
| Neurogenic | 1.25 mcg | Partial response: give another 1.25 mcg dose within 1 hour. Can be titrated to another dose of 5 mcg 24 hours later. Repeat as needed until the optimal dose is reached |
Adverse Reactions
Alprostadil’s adverse effect profile depends on the route of administration.29 Intracavernosal injections may cause penile pain, injection site hematoma or bleeding, dizziness, headache, and, in rare cases (approximately 0.4% of users), priapism.29 This treatment is contraindicated in men with conditions that increase their risk of complications, such as sickle cell anemia, multiple myeloma, severe coagulopathy, Peyronie’s disease, or poor manual dexterity.29 Intraurethral administration commonly results in local discomfort such as penile or urethral burning, testicular or perineal pain, dizziness, headache, and tachycardia.29 Although generally better tolerated, intraurethral alprostadil tends to be less effective than its injectable counterpart.29
Patient Monitoring
Monitoring during alprostadil therapy should include assessment of blood pressure, heart rate, respiratory status, temperature, the duration and quality of erections, and any penile pain or evidence of fibrosis or infection.29 No dose adjustments are required for patients with renal or hepatic impairment.29 However, clinicians should use the lowest effective dose in older adults to minimize the risk of systemic side effects.29
Alprostadil Efficacy
The efficacy of alprostadil varies depending on the formulation and patient population. Intraurethral alprostadil achieves successful intercourse in 29% to 78% of cases, while intracavernosal formulations yield success rates ranging from 53% to 100%, with higher rates in men with vasculogenic ED. Placebo-controlled trials confirm the superiority of intraurethral alprostadil over placebo; however, comparative studies consistently show greater efficacy for intracavernosal use.11 Studies have further confirmed alprostadil ICI as one of the most effective second-line treatments, particularly for patients with neurogenic or diabetic ED.30 Moreover, emerging evidence supports combination therapy, such as pairing alprostadil with PDE5 inhibitors or vacuum erection devices, for patients unresponsive to monotherapy. For men with hypogonadism, adding testosterone replacement therapy may further enhance response.11
Testosterone
By age 70, approximately 30% of men have testosterone levels below the normal threshold of 300 ng/dL, and men with low testosterone are significantly more likely to experience ED compared to those with normal levels.31 The AUA recommends against prescribing testosterone therapy for men with ED who have normal testosterone levels, and the utility of routine testosterone monitoring remains a subject of clinical debate.11 However, several randomized controlled trials have demonstrated that men with confirmed hypogonadism (total testosterone <300 ng/dL) may experience an improvement in erectile function with testosterone therapy.31,32 Observational studies also support a positive correlation between testosterone levels and erectile function.31
Recent evidence continues to support the role of testosterone therapy in improving sexual function among men with symptomatic hypogonadism. A 2022 meta-analysis reinforced the benefit of testosterone therapy for ED in men with confirmed low serum testosterone, especially when used in combination with PDE5 inhibitors.32 The review also highlighted improvements in libido, overall sexual satisfaction, and mood in many patients undergoing testosterone replacement therapy.32 However, long-term cardiovascular and prostate safety data remain areas of ongoing investigation.
Despite this, testosterone is not considered effective monotherapy for ED.11 According to the AUA guidelines, men with low testosterone who are initiating PDE5 inhibitor (PDE5i) therapy should be informed that combination treatment with testosterone may offer improved outcomes.11 Indeed, clinical trials have shown that men who received testosterone replacement and a PDE5i reported higher erectile function scores than those treated with either therapy alone.11 This is particularly relevant for men with hypogonadism who are partial responders to PDE5 inhibitors.11
Advances in Non-Oral and Regenerative Therapies for Erectile Dysfunction
While oral phosphodiesterase type 5 inhibitors (PDE5Is) remain the first-line treatment for erectile dysfunction ED, a variety of alternative and emerging options are available for patients who are unresponsive or unsuitable for these agents.15 Intracavernosal injections (ICIs) of vasoactive agents, such as alprostadil, have long been used as effective second-line therapies, with high success rates but notable side effects including penile pain, priapism, and fibrosis.15 Intraurethral formulations of alprostadil (cream and pellet) offer a less invasive route but tend to be less efficacious. Combination injectables such as Bimix and Trimix—though used off-label—may be better tolerated due to their reduced alprostadil content.15 Invicorp, a phentolamine and vasoactive intestinal peptide formulation, has demonstrated efficacy in clinical trials abroad. In select patients, botulinum toxin A (BoNT-A) has shown promise in improving erectile function when other treatments have failed, but larger trials are needed before routine use can be recommended.15
Regenerative therapies aim to restore erectile function through tissue repair and vascular remodeling.15 Low-intensity shockwave therapy (SWT) has gained popularity due to its safety profile and noninvasiveness, though inconsistent trial quality and modest effect sizes limit its widespread endorsement; currently, only the European Association of Urology endorses SWT for mild ED.15 Other investigational modalities include platelet-rich plasma (PRP) and stem cell therapies, which have shown encouraging preliminary results but remain limited by heterogeneous protocols, small patient populations, and a lack of standardized outcome measures. These therapies are not yet recommended for routine use outside of clinical trials.15 Similarly, newer pharmacologic agents—such as Rho-kinase inhibitors, guanylate cyclase activators, and central-acting melanocortin receptor agonists—target distinct pathways implicated in ED pathophysiology but have not been widely adopted due to limited human data and safety concerns.15
When non-invasive and pharmacologic interventions fail, penile prosthesis implantation remains a definitive treatment option.15 Satisfaction rates are high when patients are properly counseled, and ongoing innovations in device design aim to improve functionality and ease of use.15 While investigational technologies like magnetically activated or remote-controlled implants show potential, further clinical validation is needed.15 Taken together, these advancements reflect a growing emphasis on individualized care, particularly for men with complex, refractory, or multifactorial ED.15
Summary
Erectile dysfunction is the inability to maintain or achieve an erection satisfactory for intercourse. A multidisciplinary, individualized approach that recognizes ED as a complex condition influenced by physical, psychological, and lifestyle factors should be used. The guidelines support integration of mental health professionals and sex therapists into the care team when appropriate. This is a patient-centered, holistic approach to care that treats underlying conditions, improves the quality and frequency of erections, and avoids medication side effects. Shared decision-making and setting realistic expectations are critical components of ED management.
References
McVary KT. Sexual Dysfunction. In: Loscalzo J, Fauci A, Kasper D, Hauser S, Longo D, Jameson J, eds. Harrison’s Principles of Internal Medicine. 21st ed. McGraw-Hill; 2022.
Lee M, Sharifi R. Erectile Dysfunction. In: DiPiro JT, Yee GC, Posey L, Haines ST, Nolin TD, Ellingrod V, eds. Pharmacotherapy: A Pathophysiologic Approach. 11th ed. McGraw-Hill; 2020.
Sánchez E, Pastuszak AW, Khera M. Erectile dysfunction, metabolic syndrome, and cardiovascular risks: facts and controversies. Transl Androl Urol. 2017;6(1):28-36. doi:10.21037/tau.2016.10.01
Rew K, Heidelbaugh J. Erectile dysfunction. Am Fam Physician. 2016;94(10):820-827.
Snoga J, Williams B, Courtney L. Erectile dysfunction overview and treatment considerations in special populations. US Pharm. 2022;47(6):18-22.
Uzedy. Prescribing information. Teva Pharmaceuticals USA, Inc. Updated October 9, 2025. Accessed March 17, 2026. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=734eb776-4be0-4808-834b-0d8b0f9e021e
Proscar. Prescribing information. Organon LLC. December 23, 2024. Accessed March 17, 2026. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=6f988153-fc74-4ca4-b29a-111f750c4a4b
Male reproductive system. WikiCommons. Published 2023. Accessed June 1, 2025. https://en.wikipedia.org/wiki/Male_reproductive_system.
Muneer A, Kalsi J, Ralph D. Erectile dysfunction part I: pathophysiology and risk factors. Urology News. 2014. Accessed March 17, 2026. https://www.urologynews.uk.com/features/synopsis/post/erectile-dysfunction-part-i-pathophysiology-and-risk-factors
Sangiorgi G, Cereda A, Benedetto D, et al. Anatomy, pathophysiology, molecular mechanisms, and clinical management of erectile dysfunction in patients affected by coronary artery disease: a review. Biomedicines. 2021;9(4):432. doi:10.3390/biomedicines9040432
Burnett AL, Nehra A, Breau RH, et al. Erectile dysfunction: AUA guideline. J Urol. 2018;200(3):633-641. doi:10.1016/j.juro.2018.05.004
Vallejo-Medina P, Saffon JP, Álvarez-Muelas A. Colombian clinical validation of the International Index of Erectile Function (IIEF-5). Sex Med. 2022;10(1):100461. doi:10.1016/j.esxm.2021.100461
Roberson DW, Kosko DA. Men living with HIV and experiencing sexual dysfunction: an analysis of treatment options. J Assoc Nurses AIDS Care. 2013;24(1 Suppl):S135-S145. doi:10.1016/j.jana.2012.08.010
Sexual Health Inventory for Men (SHIM) questionnaire. The British Association of Urological Surgeons. Accessed March 17, 2026. https://www.baus.org.uk/_userfiles/pages/files/Patients/Leaflets/SHIM.pdf
Capogrosso P, Albersen M, Burnett AL, et al. Erectile Dysfunction: Update on Clinical Management. Eur Urol. Published online May 19, 2025. doi:10.1016/j.eururo.2025.05.004
Yafi FA, Jenkins L, Albersen M, et al. Erectile dysfunction. Nat Rev Dis Primers. 2016;2:16003. Published 2016 Feb 4. doi:10.1038/nrdp.2016.3
Kim ED. Erectile Dysfunction Guidelines. American Urological Association. Medscape. Updated: Aug 08, 2024. Accessed March 17, 2026. https://emedicine.medscape.com/article/444220-guidelines#:~:text=Men%20presenting%20with%20symptoms%20of,;%20Evidence%20Level:%20Grade%20C)
Kostis JB, Dobrzynski JM. The effect of statins on erectile dysfunction: a meta-analysis of randomized trials. J Sex Med. 2014;11(7):1626-1635. doi:10.1111/jsm.12509
Kim S, Cho MC, Cho SY, Chung H, Rajasekaran MR. Novel Emerging Therapies for Erectile Dysfunction. World J Mens Health. 2021;39(1):48-64. doi:10.5534/wjmh.200007
was .16 Murphy M, Demers J, Ostroff J. Oral PDE5 inhibitors for erectile dysfunction. US Pharm. 2018;43(6):29-33.
Sildenafil (Viagra) [package insert]. Pfizer; revised May 2025. Accessed June 1, 2025. https://labeling.pfizer.com/ShowLabeling.aspx?id=597
Avanafil (STENDRA) [package insert]. Vivus, Inc.; revised April 2025. June 1, 2025. https://www.stendra.com/PrescribingInformation.pdf
Tadalafil (Cialis) [package insert]. Eli Lilly and Company; revised March 2025. June 1, 2025. https://uspl.lilly.com/cialis/cialis.html
Vardenafil (Levitra, Staxyn) [package insert]. Bayer; revised February 2025. June 1, 2025. https://labeling.bayer.com/levitra/levitra.html
Ausó E, Gómez-Vicente V, Esquiva G. Visual Side Effects Linked to Sildenafil Consumption: An Update. Biomedicines. 2021;9(3):291. Published 2021 Mar 12. doi:10.3390/biomedicines9030291
Park NC, Kim TN, Park HJ. Treatment Strategy for Non-Responders to PDE5 Inhibitors. World J Mens Health. 2013;31(1):31-35. doi:10.5534/wjmh.2013.31.1.31
Yuan J, Zhang R, Yang Z, et al. Comparative effectiveness and safety of oral phosphodiesterase type 5 inhibitors for erectile dysfunction: a systematic review and network meta-analysis. Eur Urol. 2013;63(5):902-912. doi:10.1016/j.eururo.2013.01.012
Gong B, Ma M, Xie W, et al. Direct comparison of tadalafil with sildenafil for the treatment of erectile dysfunction: a systematic review and meta-analysis. Int Urol Nephrol. 2017;49(10):1731-1740. doi:10.1007/s11255-017-1644-5
Alprostadil (Caverject, Muse) [package insert]. Endo Pharmaceuticals; revised January 2025. Accessed June 1, 2025. https://www.endo.com/shared-content/labeling/caverject.html
Taşkapu H, Sönmez MG, Kılınç MT, et al. The effect of intracavernosal alprostadil use in erectile dysfunction second‑line treatment on sexual functions. New J Urol. 2022;17(1):1–5. doi:10.33719/yud.2022;17‑1‑940774
Rizk PJ, Kohn TP, Pastuszak AW, Khera M. Testosterone therapy improves erectile function and libido in hypogonadal men. Curr Opin Urol. 2017;27(6):511-515. doi:10.1097/MOU.0000000000000442
Corona G, Rastrelli G, Maggi M. The role of testosterone in male sexual function. J Sex Med. 2022;19(7):S144–S159. https://doi.org/10.1016/j.jsxm.2022.01.011
DISCLAIMER
The information provided in this course is general in nature, and it is designed solely to provide participants with continuing education credit(s). This course and materials are not meant to substitute for the independent, professional judgment of any participant regarding that participant’s professional practice, including but not limited to patient assessment, diagnosis, treatment, and/or health management. Medical and pharmacy practices, rules, and laws vary from state to state, and this course does not cover the laws of each state; therefore, participants must consult the laws of their state as they relate to their professional practice.
Healthcare professionals must consult their employer, healthcare facility, hospital, or other organization for guidelines, protocols, and procedures to follow. The information provided in this course does not replace those guidelines, protocols, and procedures, but is for academic purposes only, and this course’s limited purpose is for the completion of continuing education credits.
Participants are advised and acknowledge that information related to medications, their administration, dosing, contraindications, adverse reactions, interactions, warnings, precautions, or accepted uses is constantly changing. Any person taking this course understands that such a person must make an independent review of medication information before any patient assessment, diagnosis, treatment and/or health management. Any discussion of off-label use of any medication, device, or procedure is informational only, and such uses are not endorsed hereby.
Nothing contained in this course represents the opinions, views, judgments, or conclusions of RxCe.com LLC. RxCe.com LLC is not liable or responsible to any person for any inaccuracy, error, or omission with respect to this course or course material.
© RxCe.com LLC 2026: All rights reserved. No reproduction of all or part of any content herein is allowed without the prior, written permission of RxCe.com LLC.
RxCe.com
© RxCe.com LLC 2025: All rights reserved.

