FERTILITY MEDICATIONS: A PRIMER FOR THE PHARMACY SETTING
Faculty:
L. Austin Fredrickson, MD, FACP
L. Austin Fredrickson is an Associate Professor of Internal Medicine at Northeast Ohio Medical University, where he serves as core faculty and teaches diagnostics, therapeutics, clinical skills, and health humanities. He is board-certified in general internal medicine and practices rural primary care.
Liz Fredrickson, PharmD, BCPS
Liz Fredrickson, PharmD, BCPS, is an Associate Professor of Pharmacy Practice and Pharmaceutical Sciences at the Northeast Ohio Medical University (NEOMED) College of Pharmacy, where she is course director of the Parenteral Products and Basic Pharmaceutics Lab courses.
Pamela Sardo, PharmD, BS
Pamela Sardo, PharmD, BS, is a freelance medical writer and licensed pharmacist. She is the founder and principal at Sardo Solutions in Texas. Pam received her BS from the University of Connecticut and her PharmD from the University of Rhode Island. Pam’s career spans many years in retail, clinics, hospitals, long-term care, Veterans Affairs, and managed health care responsibilities across a broad range of therapeutic classes and disease states.
Abstract
Within the United States (US), an estimated 1 in 5 women will experience infertility. Infertility is a disease with health implications and important economic and social ramifications. Management of infertility is highly patient-specific, and factors including the underlying cause, patient age, comorbidities, and cost determine the treatment plan. A diagnosis of infertility can be devastating, and attempting to understand related procedures and medications can be overwhelming for patients. It is important for patients to be educated on the causes and risk factors associated with infertility, counseled on nonpharmacologic and pharmacologic medications, and offered resources to decrease the financial burden associated with infertility treatments. This course will provide an overview of the causes and processes involved in diagnosing infertility and discuss available infertility treatment options. Mechanisms of action, side effects profiles, benefits, and success rates will be discussed for various fertility medications.
Accreditation Statements
In support of improving patient care, RxCe.com LLC is jointly accredited by the Accreditation CouncilTM for Continuing Medical Education (ACCME®), the Accreditation Council for Pharmacy Education (ACPE®), and the American Nurses Credentialing Center (ANCC®), to provide continuing education for the healthcare team.
Joint Universal Activity Number: The Joint Accreditation Universal Activity Numbers assigned to this activity are as follows:
Pharmacy Technicians: JA4008424-0000-26-055-H01-T
Credits: 3 contact hour(s) (0.3 CEU(s)) of continuing education credit.
Credit Types:
Pharmacy - 3 Credits
Type of Activity: Application
Media: Computer-Based Training (i.e., online courses)
Estimated time to complete activity: 3 contact hour(s) (0.3 CEU(s)), including Course Test and course evaluation.
Release Date: April 25, 2026 Expiration Date: April 25, 2029
Target Audience: This educational activity is for Pharmacy Technicians
How to Earn Credit: From April 25, 2026, through April 25, 2029, participants must:
Read the “learning objectives” and “author and planning team disclosures;”
Take the “Educational Activity Pre-Test;”
Study the section entitled “Educational Activity;” and
Complete the Educational Activity Post-Test and Activity Evaluation. The Educational Activity Post-Test will be graded automatically. Following successful completion of the Educational Activity Post-Test with a score of 70% or higher, a statement of participation will be made available immediately. (No partial credit will be given.)
CME Credit: Credit for this course will be uploaded to CPE Monitor® for pharmacists. Physicians may receive AMA PRA Category 1 Credit™️ and use these credits toward Maintenance of Certification (MOC) requirements. Physician Assistants may earn AAPA Category 1 CME credit, reportable through PA Portfolio. All learners shall verify their individual licensing board’s specific requirements and eligibility criteria.
Statement of Need
Infertility can arise from female, male, or unexplained causes, including ovulatory dysfunction, tubal disease, endocrine disorders, diminished ovarian reserve, and conditions in males. Because evaluation and treatment are highly individualized, healthcare professionals must recognize common causes, explain the full range of treatment options, select age-relevant treatments and diagnosis-related processes, and consider comorbidities in shared decision-making. Variability remains in approaches to patients, and gaps remain in the assessment of ovulation induction and assisted reproductive technologies. Healthcare professionals are not uniformly aware of differences in efficacy, monitoring, side effects, and other risks, such as multiple gestation or ovarian hyperstimulation syndrome. This activity aims to bridge clinical gaps, provide practical guidance for comparing treatments, support patient-centered counseling that balances risks, benefits, and individualized preferences, and offer reminders of the opportunity for interprofessional collaboration in patient care.
Learning Objectives: Upon completion of this educational activity, participants should be able to:
Recall causes of infertility
Describe available treatment options for infertility
Discuss indications, contraindications, side effect profiles, and monitoring parameters for fertility medications
Disclosures
The following individuals were involved in planning, developing, and/or authoring this activity: L. Austin Fredrickson, MD, FACP; Liz Fredrickson, PharmD, BCPS; and Pamela Sardo, PharmD, BS. None of the individuals involved in developing this activity has a conflict of interest or financial relationships related to the subject matter. There are no financial relationships or commercial or financial support relevant to this activity to report or disclose by RxCe.com or any of the individuals involved in the development of this activity.
© RxCe.com LLC 2026: All rights reserved. No reproduction of all or part of any content herein is allowed without the prior, written permission of RxCe.com LLC.
Educational Activity Pre-Test
Which of the following is true regarding clomiphene citrate?
The brand name of clomiphene citrate is Femara
The starting dose of clomiphene citrate is 10 mg daily for three days
Side effects of clomiphene citrate include hot flashes, gastrointestinal distress, and bloating
The use of clomiphene citrate is associated with a low incidence of twin pregnancies
Which of the following medications is recommended first-line for the treatment of infertility associated with PCOS?
Clomiphene citrate
Letrozole
Menotropins
Follitropin alpha
Which of the following gonadotropins is a mixture of purified FSH and LH in a 1:1 ratio?
Menopur
Gonal-f
Ovidrel
Follistim AQ
Educational Activity
Fertility Medications: A Primer for the Pharmacy Setting
Introduction
Approximately 20% of women in the United States will experience infertility. A diagnosis of infertility can be devastating, and attempting to understand related procedures and medications can feel overwhelming for patients. It is important that patients are educated on the causes and risk factors associated with infertility, counseled on nonpharmacologic and pharmacologic medications, and offered resources to decrease the financial burden associated with infertility treatments. This course will provide an overview of the causes and processes involved in diagnosing infertility and discuss available infertility treatment options. Mechanisms of action, side effects profiles, benefits, and failure rates will be discussed for various fertility medications.
Definition and Prevalence of Infertility
Within the United States, an estimated 1 in 5 women are infertile.1 Defined as failure to achieve a pregnancy after 12 months of regular, unprotected sexual intercourse, infertility is a disease with not only health implications but also important economic and social ramifications as well.2 Management of infertility is highly patient-specific, and factors including the underlying cause, patient age, comorbidities, and cost will determine the treatment plan.2 The terms defined in Table 1 may be used when discussing infertility (Table 1).
Table 1
Definition of Terms
| Term | Definition |
|---|---|
| Fecundity | Capacity to have a live birth |
| Fecundability | The probability of achieving a pregnancy in a single menstrual cycle with adequate sperm exposure and no contraception that results in a live birth |
| Fertility | The ability to have a clinical pregnancy |
| Time to pregnancy | The length of time, usually measured in months, that it takes a couple to conceive |
Etiology and Risk Factors
An estimated 13% of reproductive-age women in the US seek out infertility treatment each year.3 Current guidelines recommend couples consider infertility treatments after 12 months of regular, sexual intercourse with no resulting pregnancy.4 However, this decision should be patient-specific, and older women and those with relevant medical histories or physical findings may require an earlier evaluation.4 It is recommended that women 35 years or older seek evaluation by the six-month mark, and women older than 40 can do so immediately.4 Patients with known oligomenorrhea (fewer than 6-8 menstrual periods per year), amenorrhea (one or more missing menstrual periods), or uterine or tubal disease should also be evaluated prior to the 12-month mark.3
For most couples, infertility is the result of either an abnormality in physiology or an underlying disease. However, 15% of infertile couples will be given a diagnosis of “unexplained infertility”, with no discernible cause identified.3 Infertility can be subdivided into primary infertility (no previous pregnancies achieved) and secondary infertility (a minimum of one prior pregnancy has been achieved).5 Causes of infertility may be categorized as female, male, or unexplained.2 Table 2 details these common etiologies.2,6
Table 2
Common Causes of Infertility2
| Cause | Percent of Cases | Types |
|---|---|---|
| Female | 30-40% |
|
| Male | 40-50% |
|
Ovulation disorders account for many cases of infertility and are categorized into three groups by the World Health Organization (Table 3).7 Women with Group 1 disorders will present with amenorrhea and have low levels of gonadotropins and estrogen.7 Causes of Group 1 disorders include eating disorders and excessive exercise.7 Group 2 disorders result from dysfunction within the hypothalamic-pituitary-ovarian axis with related conditions, including polycystic ovarian syndrome (PCOS) and hyperprolactinemic amenorrhea.6 Polycystic ovarian syndrome is the most common cause of anovulatory infertility.2 Group 3 disorders are due to ovarian failure, in which hypergonadotropic, hypoestrogenic anovulation is seen.6 Ovarian reserve decreases naturally as women age, especially after age 35. However, many other factors can hasten this process, including prior chemotherapy or radiation, ovarian surgery, and a family history of premature menopause.2
Table 3
World Health Organization Ovulation Disorders6
| Group | Cause | Disorders | Percent of Ovulation Disorders |
|---|---|---|---|
Group 1 (Hypogonadotropic hypogonal anovulation) | Hypothalamic pituitary failure | Hypothalamic amenorrhea Hypogonadotropic hypogonadism | 10% |
Group 2 (Normo-gonadotropic, normo-estrogenic anovulation) | Hypothalamic- pituitary-ovarian axis dysfunction | Polycystic ovarian syndrome (PCOS) and hyperprolactinemic amenorrhea | 85% |
Group 3 (Hyper-gonadotropic, hypoestrogenic anovulation) | Ovarian failure | — | 5% |
Tubal infertility occurs when the fallopian tubes are blocked or are unable to pick up an oocyte due to issues such as pelvic adhesions.3 These cases account for anywhere from 11 to 67% of infertility diagnoses.3 Tubal infertility can result from pelvic inflammatory disease, salpingitis isthmica nodosum, endometriosis, and prior surgery.2
Infertility can occur in women of all ages, but age does play a role. This may be due to many factors, including an increased risk of disorders affecting fertility.4 Women younger than 30 years old have an 85% chance of conceiving within one year.8 By the age of thirty, this decreases to approximately 75%.8 At age 35, the chance is 66%, and at age 40, it is 44%.8 Age is also a major consideration in determining the success of infertility treatments. As patients age, the success rates for treatments generally decrease.3
Evaluation and Diagnosis
Evaluating a patient for infertility and determining a diagnosis involves an understanding of both the female reproductive system and the menstrual cycle. These are described in detail below.
Female Reproductive System
The female reproductive system consists of the vagina, ovaries, uterus, cervix, and fallopian tubes.9 The uterus is a pelvic organ that serves many functions, including as a site of implantation for a blastocyst and later providing support for a developing fetus.10 It also has a membrane lining called the endometrium, which is shed during the menstrual period.10 The ovaries are the main organs of the female reproductive system and contain thousands of follicles.10 A woman is born with all the oocytes she will have during her lifetime (between 1 and 2 million); by the age of menopause, this number is approximately 1000.4 The ovaries connect to the uterus via the fallopian tubes, which carry a fertilized egg cell (zygote) to the uterus for implantation.10 The vagina is the entryway for the sperm, and the cervix is a passageway into and out of the uterus that allows for sperm entry.10 For the female reproductive system to function normally, hormonal signals from the hypothalamus, pituitary gland, and ovaries must integrate to promote follicle development and ovulation.11
Menstrual Cycle
An understanding of the menstrual cycle can assist pharmacists in understanding the causes of infertility. The menstrual cycle begins with menarche (the occurrence of a first menstrual cycle), which occurs between the ages of 10 and 16, and continues until menopause is reached, typically around age 50.11 During a cycle, various hormones interact to stimulate ovulation. The menstrual cycle is divided into three phases: the follicular phase, the ovulatory phase, and the luteal phase.11 The length of a menstrual cycle is the number of days from the first day of bleeding to the onset of menstruation in the next cycle.12 The average length of a menstrual cycle is 28 days, but this can vary widely (a common range is 21 to 40 days) and be impacted by numerous factors, including body weight, nutritional status, and medical conditions.11 The figure below visually depicts the menstrual cycle, and the three phases are described in more detail below.13
The Menstrual Cycle13
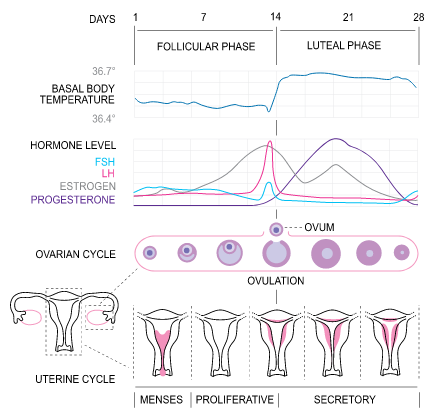
The Follicular (Proliferative) Phase
The first day of menses (bleeding) is considered Day 1 of the menstrual cycle and commences the follicular, or proliferative, phase.11,12 In a standard, 28-day cycle, the follicular phase will continue until day 14.11,12 The range of normal menstrual cycle lengths is due to variations in the length of the follicular phase.12,13 At the onset of the follicular phase, levels of FSH increase, which aids in recruiting follicles to grow and develop.11 A follicle is a small, fluid-filled sac that contains an immature egg.14 One of these follicles will become the dominant follicle and release the oocyte (egg cell) via rupturing.11,12 This dominant follicle can prevent the remaining follicles from developing by increasing levels of two hormones: estradiol and inhibin.11 These hormones act on the hypothalamus to prevent the release of gonadotropin-releasing hormone (GnRH) and on the pituitary gland to prevent the release of FSH in a negative feedback manner.11 The more estradiol a woman’s body produces, the less GnRH and FSH will be released. As the dominant follicle grows, it produces three other hormones: estradiol, progesterone, and androgen.11 Estradiol stops menses from the previous cycle, which thickens the uterine lining.11 At the end of the follicular phase, the dominant follicle is ready for ovulation.
Ovulatory Phase
Ovulation occurs 14 days before menses, so it would occur around day 14 during an average 28-day cycle.11 The ovulatory phase is the time during which the oocyte ruptures from the follicle.11 When critical levels of estradiol are reached, the pituitary gland releases high levels of a hormone called luteinizing hormone (LH).11,12 This is known as the “LH surge”.11,12 The rapid rise in LH causes the final maturation of the follicle and release of the oocyte.12 Ovulation will generally occur sometime between 24 and 36 hours after estradiol peaks and 10 to 12 hours after LH peaks.11,12 Once ovulation has occurred, the oocyte is able to travel via the fallopian tube for fertilization and then to the uterus for implantation of the embryo if fertilization was successful.11,12
Luteal Phase
The final phase of the menstrual cycle is the luteal or secretory phase, which follows ovulation and lasts until the next menstrual cycle begins.11,12 A typical luteal phase lasts 14 days.12 The dominant hormone in this phase is progesterone, which acts in a negative feedback manner to decrease LH and FSH levels.12 During the luteal phase, the dominant follicle becomes the corpus luteum.12 Progesterone produced by the corpus luteum is key in maintaining the endometrial lining to support the implanted embryo and maintain a pregnancy.11 If pregnancy does occur, a hormone called human chorionic gonadotropin (HCG) will sustain the corpus luteum and stimulate continued production of estrogen and progesterone until the placenta takes over.11,12 Progesterone levels decrease if pregnancy does not occur as a result of failed fertilization or implantation.11,12 This rapid drop in progesterone causes the endometrial lining to shed, and a new menstrual cycle then begins.11,12
Fertility Evaluation
Fertility evaluations should involve both partners, given the broad range of causes and high prevalence of unexplained infertility.2 Initial evaluations include a medical history, laboratory testing, and physical exam.2 Research has shown that an important part of fertility evaluations is the evaluation of sexual function and experience.14 Arizona Sexual Experience Scale (ASEX) is an acceptable initial assessment for all patients.15 The International Index of Erectile Function (IIEF) is a preferred scale for men, and the Female Sexual Function Index (FSFI) is a good scale for women.14,16,17
Additionally, medical histories can uncover potential risk factors for infertility, such as alcohol and tobacco use, and intense exercise.2 Clinicians should also determine how well a couple understands ovulation and if they are using correct methods to detect ovulation and are timing intercourse appropriately.2 The female partner should undergo a physical assessment, including body weight, blood pressure, and thyroid, breast, and pelvic examinations.2 Ultrasound can be used to detect uterine abnormalities and evaluate ovarian reserve via counting the number of antral follicles.2 Fallopian tube patency can be determined via a hysterosalpingogram (HSG), which is completed by injecting a radiopaque contrast into the uterus and imaging the flow through the fallopian tubes.2 Obtaining an FSH level and estradiol level on day 2 or 3 of the menstrual cycle can assist with assessing ovarian reserve.2 Serum anti-mullerian hormone (AMH) levels can be used in combination with ultrasound findings and the patient’s age to identify diminished ovarian reserve.2 Other lab work to consider in women with irregular menstrual cycles includes TSH, prolactin, and androgens.2
Treatment of Infertility
Nonpharmacologic Management
Managing infertility from a nonpharmacologic perspective involves reducing risk factors that may contribute to infertility. Prior to initiating infertility treatment, it is important to manage identified underlying causes, including excess body weight, lack of exercise, poor diet, and medication use.6 Including a dietician and/or behavioral nutritionist specialist in the care of patients is important. Many electronic medical record systems have screening tools that are important to use during initial intake appointments. Determining which questionnaires are most relevant is a critical process that will enable medical staff to help patients access essential resources.
A 2017 review sought to assess the effect of physical activity on ovulation.18 Cohort studies in this review found that the risk of anovulation increased among those who engaged in extremely heavy exercise (> 60 minutes per day), whereas exercising between 30 and 60 minutes per day reduced the risk of anovulatory infertility.18 In patients with PCOS, exercising, even without changes in diet, can assist with the resumption of ovulation.18
Patients should also focus on consuming a healthy diet—one low in saturated fat, high in fiber, and low in animal protein.6 Other recommendations include avoiding alcohol, tobacco, and recreational drug use.6 A review of the effects of diet on infertility suggests that the use of antioxidant supplements is not likely to make a difference in terms of improving fertility, but this is limited by a lack of data to make this determination.19 Use of folic acid in conjunction with assisted reproductive technology may lead to increased fertility rates and live births.19 Higher doses of folic acid with additional vitamin B12 may also be of value.19 Literature assessing vitamin D use has focused only on women with levels close to normal, so while adequate supplementation does not appear to play a role in fertility, the effects of severe deficiency cannot be ruled out.19 While few studies have investigated the effects of dairy foods, these likely do not have an important effect on fertility.19 In general, consuming a “healthy” diet will likely assist in improving fertility and live birth rates in those undergoing ART.
Treatment Termination Conversations
White women in households with incomes of >$100,000 are the primary subject for fertility treatments.20 Many of these women are female physicians.21 Research is showing that when insured women stop fertility treatments, it is the result of the psychological burden that it creates.22 The conversation of fertility treatment termination is critical. Many nurses struggle with patients who look to them for advice on when to terminate treatments.23 In a small qualitative study, researchers found that patients felt the possibility of the “technologically mediated pregnancy” had over their mental, financial, or physical well-being.24
Medical staff helping with fertility clinics, awareness of the importance of termination conversations is crucial.25 In 2011, it was estimated that the average cost of medications only was $1,182, rising to $61,000 for successful IVF outcomes in the United States.26 Each year, the cost continues to increase. Helping patients and partners navigate their thresholds is essential.
Routine Psychosocial Care
Fertility treatment has been found to take a mental as well as physical toll on the patients involved. An important part of treatment is also providing the mental support that patients need during this time. Providing and/or encouraging infertile couples to seek counseling and psychological interventions has been found to increase marital and sexual satisfaction.21 The nurse’s role in identifying psychosocial care is essential. Administering routine psychosocial assessments helps the entire team address issues more holistically.
One study found that military veterans with a history of sexual assault were twice as likely to report infertility.27 When providing fertility treatment, an initial screening for sexual assault is important to help guide medical staff in their interactions with patients.28 Fertility treatments, administration, and procedures can bring up traumatic reactions.28
Assisted Reproductive Technology
Within the United States, the use of assisted reproductive technology (ART) has risen significantly over the past few decades, with 182,111 procedures completed in 2015.29 Assisted reproductive technology encompasses several procedures that involve handling embryos or oocytes and sperm in vitro.29 Each protocol may vary and is specific to the fertility clinic, including intrauterine insemination (IUI) and in vitro fertilization (IVF).6 For IUI, sperm is prepared and placed into the uterus directly.6 IUI procedures are often done in conjunction with a medication regimen that stimulates ovulation prior to the procedure.6 Intrauterine insemination can assist not only in cases of unexplained infertility, but also for couples with male factor infertility, those who cannot engage in vaginal intercourse, or those in same-sex relationships.6 In vitro fertilization is a procedure in which oocytes are fertilized in a laboratory and then transferred directly to the uterus, bypassing the fallopian tubes.2 In vitro fertilization provides couples with tubal ligation infertility the best chance of success.2 These procedures are more involved (and typically more expensive) than IUI. The following steps are included within a typical ART cycle:30
Medications are started to stimulate the growth of multiple ovarian follicles, while other drugs are given to suppress the natural menstrual cycle and down-regulate the pituitary gland
Ovarian-stimulating drugs are started
Follicle growth is monitored
Ovulation-triggering medication is given to cause egg maturation once follicles have reached an appropriate size
Eggs are collected, and sperm is retrieved (if needed)
Fertilization via IVF or ICSI is completed
Laboratory procedures are done for embryo culture
Embryos are placed in the uterus
The luteal phase is supported via progesterone, estrogen, and/or hCG
Laws and Regulations of Assisted Reproductive Technology (ART)
In the United States, ART is regulated for safety at both the federal and state levels. Fertility Clinic Success Rate and Certification Act (Public Law 102-493) 1992- Mandates that all ART clinics report success rate data to the federal government.31 ART is an expensive emotional, physical, and monetary investment. Patients should be encouraged to know a fertility clinic's success rate. That unbiased information is collected and provided by the Centers for Disease Control and Prevention.32
Pharmacologic Therapies
Infertility treatments involve ovulation induction. Physiologically, only one follicle typically becomes dominant during the menstrual cycle, but ovulation induction seeks to develop and mature many follicles.2 Use of these medications is commonly paired with either IUI or IVF.2 Patients may also have the option of having timed intercourse.2 Oral and parenteral medications are available for use. Medications used for infertility are summarized in Table 4.
Table 4
Medications Used to Treat Infertility6
| Medication(s) | Indications | Dosing | Storage conditions | Adverse reactions and contra- indications |
|---|---|---|---|---|
| Clomiphene citrate (Clomid) | Treatment of ovulatory disorders | 50-150 mg once daily by mouth for 5 days (starting on days 3, 4, or 5 of the cycle) | Store at room temperature Avoid excessive exposure to heat, light, and humidity | Ovary enlargement, hot flashes, headache, abdominal distension, vomiting, and breast discomfort. Potential visual changes and OHSS Contraindicated in thyroid and adrenal dysfunction |
| Letrozole (Femara) | Ovulation stimulation in anovulatory women with PCOS | 2.5-7.5 mg once daily by mouth for 5 days starting on day 3, 4, or 5 of the cycle | Store at room temperature | Flushing, edema, headache, dizziness, fatigue, night sweats, weight gain, nausea |
| Follitropin alpha (Gonal-f; Follitropin beta (Follistim AQ) | Ovulation induction and multifollicular development during ART | Dose is individualized based on therapeutic response. Doses can vary from 37.5 to 450 IU daily | Follitropin alpha: refrigerate or store vials at room temperature; after reconstitution, store them at room temperature or in a refrigerator for up to 28 days Follitropin beta should be refrigerated before dispensing and stored in a refrigerator or at room temperature for up to 28 days | Headache, ovarian cysts, abdominal pain, nausea, injection site reaction, potential OHSS |
| Menotropin (Menopur) | Ovulation induction and multifollicular development during ART | Doses vary. | Refrigerate or store at room temperature | Headache, OHSS, abdominal pain, vomiting, diarrhea |
Leuprolide (Lupron- Depot) | Ovulation trigger (off-label use) | Dosing protocols vary. | Refrigerate | Pain at the injection site; mood changes, headache, skin rash |
| Cetrorelix (Cetrotide) and Ganirelix (Fyremadel) | Adjunct to controlled ovarian stimulation | Varying dosing protocols | Refrigerate and protect from light | Headache, OHSS, nausea, pelvic pain, injection site reaction |
Clomiphene
Clomiphene citrate (CC), brand name Clomid, is a selective, nonsteroidal estrogen receptor modulator (SERM).33 It has FDA approval for the treatment of ovulatory dysfunction in women with infertility.6 SERMs are a class of medications that have tissue-specificity and can function as either estrogen receptor agonists or antagonists depending on the tissue.6,33 Clomiphene citrate exerts its effects at the hypothalamus, where it binds estrogen receptors (ER) longer than estrogen does.33 By doing so, clomiphene interferes with receptor recycling and inhibits normal estrogenic negative feedback, increasing GnRH secretion and release of FSH and LH.33 This leads to ovarian follicle development and ovulation.
Ovulation is expected 5 to 10 days following a course of CC, and because of this, it is generally taken around the fifth day of a menstrual cycle.33 The starting dose of clomiphene is 50 mg once daily for five days.33 The dose can be increased to 100 mg once daily if ovulation does not occur at the 50 mg dose.33 While doses up to 250 mg once daily have been used, doses greater than 100 mg daily have not been shown to increase pregnancy rates, and this is the recommended maximum dose.33 It is recommended to discontinue the use of CC if ovulation does not occur after three courses of treatment or if ovulation occurs three times, but no pregnancy is achieved.33 No dose adjustments are needed for impaired renal function, but use is contraindicated in patients who have a history of liver disease.33
Clomiphene is associated with many side effects, including ovarian enlargement, hot flashes, gastrointestinal distress, bloating, and breast discomfort.33 A serious potential side effect is ovarian hyperstimulation syndrome (OHSS), which is characterized by severe abdominal pain, severe nausea and vomiting, excessive weight gain, and shortness of breath. Another serious side effect is visual changes.33 These are rare and more likely to occur with higher doses and longer durations of therapy, and in some cases, the changes will be irreversible.33 If OHSS or visual changes occur, CC should be discontinued, and the patient should be seen as soon as possible for evaluation.33 Contraindications include hypersensitivity to clomiphene, uncontrolled thyroid or adrenal dysfunction, pregnancy, and organic intracranial lesions.33
In clinical trials, CC pregnancies occurred in approximately 30% of patients, with an incidence of multiple pregnancies (twins, triplets, or more) of approximately 8%.6 Up to 15% of patients may be clomiphene resistant and not respond to treatment.34
Letrozole
Letrozole, brand name Femara, is a third-generation, nonsteroidal, highly selective aromatase inhibitor (AI).34,35 It was first used to induce ovulation in animals in 1993.34 The aromatase enzyme catalyzes the conversion of androstenedione to estrone and of testosterone to estradiol.34 Letrozole reversibly binds to and inhibits the aromatase enzyme in a competitive manner (more than 99%), thereby reducing plasma estrogen levels.34,35 This decrease in estrogen levels increases FSH secretion, which stimulates ovulation.35 Letrozole may also act peripherally by blocking the conversion of androgen substrates to estrogen, thereby increasing follicular sensitivity through upregulation of the FSH receptor gene.34 Letrozole reaches steady-state levels between 2 and 6 weeks and is rapidly and well-absorbed (mean bioavailability is 99.9%).34,35 It undergoes hepatic metabolism via CYP3A4 and CYP2A6.35 It can be taken with or without food.35
Use of letrozole is off-label for infertility, but it is common for females with ovulatory disorders, normal or elevated estrogen concentrations, and irregular ovulation.35 It is recommended first-line for patients with PCOS, given the results of the Polycystic Ovary 2 Trial, which found that letrozole resulted in higher live births than clomiphene (27.5% vs. 19.1%).36 These results were echoed by a 2018 Cochrane review that included 42 RCTS.37 Within this review, live birth rates were higher with letrozole, and there was no difference between rates of OHSS, miscarriage, and multiple pregnancies.37 Letrozole has also demonstrated effectiveness in preparing the endometrium for thawed-embryo transfers (FET) and can be used in women who ovulate to better their chances of pregnancy.34,35 Diagnoses that may benefit include endometriosis, pelvic factors, and advanced maternal age.34
Letrozole protocols mirror those used for clomiphene.34 For use as an ovulation induction agent, letrozole is started at 2.5 mg once daily for 5 days, typically on days 3, 4, or 5 of the menstrual cycle or following a progestin-induced bleed.35 Typical follicle sizes at this time range from 6 to 8 mm, and these follicles have high levels of androgen receptors and increased androgen levels, encouraging FSH receptor induction.35 The dose may be increased to 5 mg daily for five days if ovulation does not occur with use of the 2.5 mg dose.35 The maximum recommended dose is 7.5 mg/day. It is not clear how often letrozole can be used, but one study dosed letrozole for a maximum of five cycles.34
The adverse effects of letrozole were demonstrated in its use for post-menopausal women with breast cancer who were on the treatment much longer than those using it for ovulation induction.34 However, women using letrozole for infertility may still experience headache, flushing, diaphoresis, night sweats, and hot flashes.35 OHSS may also occur.35 Three significant letrozole side effects include decreased bone mineral density, ischemic cardiovascular events, and musculoskeletal effects, but these are not as likely with the short-term use associated with ovulation induction.34,35 Letrozole is safe from a teratogenic perspective, but additional research is needed to determine the long-term effects on children.34
Letrozole use can be paired with timed intercourse or IUI. The use of letrozole is associated with pregnancy rates of 23% and an incidence of multiple pregnancies of 13%.6 Studies have not found a significant difference in many side effects when comparing letrozole and clomiphene, though letrozole may cause more fatigue and dizziness, while clomiphene may cause more hot flashes.6 Compared to clomiphene, letrozole tends to promote the development of only one follicle, as it does not inhibit the negative feedback of estrogen.6 Also, compared to clomiphene, letrozole has a short half-life and therefore may not cause the adverse effects on cervical mucus and endometrium that clomiphene does.35
Patients in WHO Group 2 anovulation (described above in Table 3) who use letrozole may have higher ovulation rates, pregnancy rates, and live birth rates, but no differences in multiple pregnancies and miscarriage rates.36,37 Additional studies are needed to determine the role of letrozole in unexplained infertility.37,38
Gonadotropins
Patients with a condition called hypogonadotropic hypogonadism have little or no response to pituitary gonadotropin release.2 Because of this, clomiphene and letrozole are of little use.3 Instead, exogenous gonadotropins can be used to stimulate ovarian follicles.3 Women who benefit from gonadotropins include those with PCOS who have not ovulated or conceived despite weight loss or use of clomiphene or letrozole therapy; those with hypothalamic anovulatory hypopituitarism disorders; and those with hypothalamic amenorrhea.3 A 2019 Cochrane review found that gonadotropins resulted in a greater percentage of live births than clomiphene after six cycles for those with PCOS, but also a higher rate of multiple pregnancies.39
Gonadotropins are injectable medications used as ovarian stimulators.6 They enhance ovulation function by providing FSH and LH activity, leading to the recruitment and growth of many follicles.6 Menotropins (brand name Menopur®) are one category of gonadotropins.40 These are a purified combination of FSH and LH in a 1:1 ratio that has been extracted from the urine of postmenopausal women.40 Menopur is available as a powder for reconstitution and contains 75 units of menotropins, which represents 75 units of FSH and LH activity.40 The half-life of Menopur is 11-13 hours, and it peaks within 18 hours following a single subcutaneous dose.40 Menopur is dosed as 225 units once daily beginning on cycle day 2 or 3.40 The dose can be adjusted after 5 days based on ultrasound monitoring of ovarian response and/or measurement of serum estradiol levels.40 It is recommended not to make additional adjustments more frequently than once every 2 days or by >150 units.40 The maximum daily dose is 450 units.40
Follitropin alpha (Gonal-f) is another type of gonadotropin used for ovarian stimulation.41 For use in ovulation induction, it is dosed subcutaneously as 75 units for 14 days in an initial cycle, with doses increased by 37.5 units after 14 days, based on ovarian response.41 The dose can continue to be adjusted to a maximum of 300 units daily.41 Gonal-f is continued until serum estradiol levels and/or follicle growth indicate an appropriate ovarian response.41 It is generally recommended that therapy does not exceed 35 days.41 For use in multi-follicular development during ART, Gonal-f is dosed around day 2 or 3 of the cycle at 150 units daily until appropriate follicular development is attained.41
Follitropin beta, brand name Follistim AQ, can be used for ovulation induction in anovulatory patients.42 It is available as a cartridge that contains a prefilled solution and is administered via a pen injector device.42 Dosing for Follistim AQ cartridge is done via a stepwise approach, with an initial dose of 50 units per day for the first 7 days.42 The dose is then increased by 25 or 50 units weekly until follicular growth or estradiol levels demonstrate an adequate ovarian response.42 The dose can be adjusted to prevent the development of too many follicles, which would result in cancellation of the cycle.26 The maximum recommended dose is 250 units.42
Another gonadotropin used is recombinant chorionic gonadotropin.43 This medication, brand name Ovidrel, causes ovulation to occur following the use of other fertility medications.43 Ovidrel is injected subcutaneously at a dose of 250 mcg 1 day after the final dose of a follicle-stimulating agent.43
Gonadotropin therapy is monitored with transvaginal ultrasound and by measuring levels of FSH, LH, and estradiol.40-42 Depending on the protocol, an ovulation trigger is given once the target number of follicles is developed.6 Cycles may be canceled if follicles do not reach the target size (>15 mm) or an inadequate number develops.6 Use of gonadotropins is associated with injection site reactions.40-42 These medications also put patients at a much higher risk of multiple gestations compared to clomiphene and letrozole.40-42 Other side effects include headache, abdominal pain, nausea, and OHSS.40-42
GnRH Agonists and Antagonists
Some women will undergo controlled ovarian hyperstimulation (COH) to develop many follicles that can be retrieved and fertilized.6 These agents are used along with medications that cause ovulation induction.6 Leuprolide (Lupron-Depot) is a GnRH agonist.44 By inhibiting gonadotropin secretion, leuprolide increases levels of LH and FSH.44 Leuprolide has a half-life of about 3 hours and has a bioavailability of 94%.44 It is administered subcutaneously, and typical starting doses are 0.5 to 1 mg daily.44 Common side effects include pain at the injection site, emotional lability, headache, mood changes, weight gain, and skin rash.44
Cetrorelix (Cetrotide) and ganirelix (Fyremadel) are GnRH antagonists. These agents compete with GnRH for binding to pituitary receptors, thereby delaying the LH surge and preventing ovulation until the follicles reach the appropriate size.45,46 Cetrorelix and ganirelix are used as adjuncts for controlled ovarian stimulation.6,45,46 They are given subcutaneously, and dosing protocols vary based on the patient.45,46 Patients can self-administer the medications; as a result, self-administration instructions, administration practice, and disposal of needles need to be provided to patients. Rotating the injection site can help reduce local irritation.45,46
Precautions for Potential Anaphylaxis First administration of the medications needs to be with a physician present and anaphylaxis crash cart present. Monitor a patient for allergic reaction for 30 minutes after first administration of medication.47 |
|---|
Possible side effects include headache, OHSS, nausea, and injection site reactions.45,46
Determining a Management Strategy
Once a patient has been evaluated, a treatment recommendation will be made based on results and findings. Success rates vary and will depend on many factors, including the female partner’s age, ovarian reserve, history of smoking, BMI, and race.2 Table 5 summarizes recommendations based on the underlying cause of infertility.2
Table 5
Management Recommendations
| Condition | Recommendation |
|---|---|
| Ovulatory dysfunction that is the result of endocrine disorders (hypothyroidism) | Treat conditions prior to undergoing fertility treatments |
| Anovulatory infertility | Clomiphene citrate and letrozole to induce ovulation. Second-line medications, such as metformin, may also be needed, especially in women with obesity |
| Hypothalamic amenorrhea | Weight gain and decreased exercise, and resume ovulation following these changes. If this is not successful, gonadotropins can be used in low doses. |
| Diminished ovarian reserve | Proceed to IVF |
| Unexplained infertilitya | Oral medications can be started to increase the number of oocytes, and these can be used in conjunction with IUI to time ovulation. It is recommended to attempt this 3-6 times prior to starting IVF. |
aThe Fast Track and Standard Treatment Trial randomized women into two groups: 3 cycles of clomiphene-IUI followed by 3 cycles of clomiphene gonadotropins-IUI prior to IVF or 3 cycles of clomiphene-IUI followed by 6 cycles of IVF.48 Authors found that the time to pregnancy was significantly shorter in the clomiphene-to-IVF group.48 Because of this, gonadotropin-IUI is not recommended for unexplained infertility.48
Frequently Asked Questions3
Do fertility medications cause birth defects?
In 2012, a meta-analysis of 46 studies that included 124,468 children who were conceived via IVF or ICSI was completed to compare risk of birth defects. A higher risk of birth defects was associated with ART (RR 1.37).49 A separate study in Australia that included 327,420 births did not find an association between assisted conception and risk of birth defects (an exception was ICSI births). Further, no studies have found an increased risk of birth defects associated with the use of letrozole or clomiphene.49 A third meta-analysis found pregnancies from ART had a significantly higher risk of congenital malformation than spontaneous conceptions, but the risk was small, and no absolute risk rates were provided in the study.50
Do fertility medications place women at a higher risk for breast cancer?
Infertility itself places a woman at a higher risk for breast, endometrial, and ovarian cancers.3 However, it is unclear if undergoing fertility treatments incurs these same risks.3 One Cochrane review found women who used clomiphene citrate to conceive were at a higher risk of endometrial cancer than controls, but it is unclear if this was due to underlying medical conditions or the clomiphene itself.51 Available evidence regarding the risk of ovarian cancer has not identified a strong association between the use of these ovarian-stimulating drugs and this risk. Additionally, numerous systematic reviews have not found an association between risk of breast cancer and use of ovulation induction agents.3 Overall, there is currently no good evidence to suggest that use of fertility medications increases a patient’s risk of breast, ovarian, or endometrial cancer.3
What other complications may result from ART?
Studies suggest that ART may be associated with higher risks of pregnancy-related complications and adverse pregnancy outcomes.52 premature rupture of membranes, pregnancy-induced hypertension, gestational diabetes mellitus, preterm or very preterm birth, and low or very low birth weight. Further research is needed to determine which aspect of ART poses the greatest risk and how to minimize it.
The Role of the Pharmacy Staff
It is important for pharmacy staff to provide patient-centered care as part of multidisciplinary teams to maximize patient education and outcomes. To date, more than 100 pharmacies specialize in fertility therapies.6 Pharmacists can assist patients within 6 different realms related to fertility treatment. These include effectiveness, burden time, financial costs, potential risks, and genetic parentage.6 Provision of patient education is especially important, considering how important medication therapy plays a role in treatment. With any regimen, patients may utilize several different medications, and doses of these medications may change over the course of treatment depending on ovarian response.6 Demonstrating appropriate administration technique, counseling on potential side effects, and assisting with affording these medications can help patients greatly.6
Pharmacists and pharmacy staff can also provide vital emotional support for patients undergoing infertility treatment.6 When patients feel knowledgeable and empowered regarding their regimen, this can help to alleviate stress and anxiety. Pharmacists and their staff can assist by anticipating urgently needed medications as doses and therapies change. They can also anticipate when patients may require referrals for emotional support, such as to RESOLVE: The National Infertility Association.37
Other Resources RESOLVE: The National Infertility and Family Building Association helps people navigate their fertility journey by providing information about infertility, family building options, getting insurance coverage, financial resources, and more.53 Link: https://resolve.org/ |
|---|
Summary
Within the United States, an estimated 1 in 5 women are infertile. Defined as failure to achieve a pregnancy after 12 months of regular, unprotected sexual intercourse, infertility is a disease with not only health implications but also important economic and social ramifications. Current guidelines recommend couples consider infertility treatments after 12 months of regular sexual intercourse with no resulting pregnancy.
Fertility evaluations should involve both partners, given the broad range of causes and high prevalence of unexplained infertility. Managing infertility from a nonpharmacologic perspective involves reducing risk factors that may contribute to infertility. Prior to initiating infertility treatment, it is important to manage identified underlying causes, including excess body weight, lack of exercise, poor diet, and medication use. Pharmacologic infertility treatments involve ovulation induction. It is important for pharmacists to provide patient-centered care as part of multidisciplinary teams to maximize patient education and outcomes.
References
Infertility. Centers for Disease Control and Prevention. March 2022. Accessed February 2023. Available at: https://www.cdc.gov/reproductivehealth/infertility/index.htm#:~:text=Is%20infertility%20a%20common%20problem,year%20of%20trying%20(infertility).
Dokras A, Hall JE. Infertility and Contraception. In: Loscalzo J, Fauci A, Kasper D, Hauser S, Longo D, Jameson J. eds. Harrison's Principles of Internal Medicine, 21e. McGraw Hill; 2022. Accessed February 10, 2023.
Carson SA, Kallen AN. Diagnosis and Management of Infertility: A Review. JAMA. 2021;326(1):65-76. doi:10.1001/jama.2021.4788
American College of Obstetricians and Gynecologists Committee on Gynecologic Practice and Practice Committee. Female age-related fertility decline: committee opinion No. 589. Fertil Steril. 2014;101(3):633-634. doi:10.1016/j.fertnstert. 2013.12.032
Ordonez ND. Infertility. In: O'Connell M, Smith JA. eds. Women's Health Across the Lifespan, 2e. McGraw Hill; 2019. Accessed February 10, 2023.
Hutcherson NEC, Harris JB, Karaoui LR, et al. Infertility Management and Pharmacotherapy: What Every Pharmacist Should Know. J Pharm Pract. 2021;34(4):635-647. doi:10.1177/0897190020930969
National Collaborating Centre for Women’s and Children’s Health (UK). Fertility: Assessment and Treatment for People with Fertility Problems. London: Royal College of Obstetricians & Gynaecologists; 2013 Feb. (NICE Clinical Guidelines, No. 156.) 8, Ovulation disorders.
Delbaere I, Verbiest S, Tydén T. Knowledge about the impact of age on fertility: a brief review. Ups J Med Sci. 2020;125(2):167-174. doi:10.1080/03009734.2019.1707913Female repro anatomy
Wikicommons. Female Reproductive System. Accessed February 10 2023. Available at: https://commons.wikimedia.org/wiki/Category:Human_female_reproductive_system#/media/File:Vaginal_Ultrasound.png
Hall JE, Dokras A. Disorders of the Female Reproductive System. In: Loscalzo J, Fauci A, Kasper D, Hauser S, Longo D, Jameson J. eds. Harrison's Principles of Internal Medicine, 21e. McGraw Hill; 2022.
El-Ibiary SY, Shrader SP, Ragucci KR. Contraception. In: DiPiro JT, Yee GC, Posey L, Haines ST, Nolin TD, Ellingrod V. eds. Pharmacotherapy: A Pathophysiologic Approach, 11e. McGraw Hill; 2020. Accessed January 18, 2023.
Reed BG, Carr BR. The Normal Menstrual Cycle and the Control of Ovulation. [Updated 2018 Aug 5]. In: Feingold KR, Anawalt B, Boyce A, et al., editors. Endotext [Internet]. South Dartmouth (MA): MDText.com, Inc.
Menstrual Cycle. Wikimedia Commons. Accessed January 2023. Available at: https://commons.wikimedia.org/wiki/File:MenstrualCycle2_en.svg
Bond JC, White KO, Abrams JA, Wesselink AK, Wise LA. Sexual dysfunction, distress, and care-seeking among females during the preconception period [published online ahead of print, 2023 Mar 29]. Am J Obstet Gynecol. 2023;S0002-9378(23)00222-3. doi:10.1016/j.ajog.2023.03.037
McGahuey CA, Gelenberg AJ, Laukes CA, et al. The Arizona Sexual Experience Scale (ASEX): reliability and validity. J Sex Marital Ther. 2000;26(1):25-40. doi:10.1080/009262300278623
Rosen RC, Cappelleri JC, Gendrano N 3rd. The International Index of Erectile Function (IIEF): a state-of-the-science review. Int J Impot Res. 2002;14(4):226-244. doi:10.1038/sj.ijir.3900857
Rosen R, Brown C, Heiman J, et al. The Female Sexual Function Index (FSFI): a multidimensional self-report instrument for the assessment of female sexual function. J Sex Marital Ther. 2000;26(2):191-208. doi:10.1080/009262300278597
Hakimi O, Cameron LC. Effect of Exercise on Ovulation: A Systematic Review. Sports Med. 2017;47(8):1555-1567. doi:10.1007/s40279-016-0669-8
Gaskins AJ, Chavarro JE. Diet and fertility: a review. Am J Obstet Gynecol. 2018;218(4):379-389. doi:10.1016/j.ajog.2017.08.010
Galic I, Negris O, Warren C, Brown D, Bozen A, Jain T. Disparities in access to fertility care: who's in and who's out. F S Rep. 2020;2(1):109-117. Published 2020 Nov 18. doi:10.1016/j.xfre.2020.11.001
Stentz NC, Griffith KA, Perkins E, Jones RD, Jagsi R. Fertility and Childbearing Among American Female Physicians. J Womens Health (Larchmt). 2016;25(10):1059-1065. doi:10.1089/jwh.2015.5638
Domar AD, Rooney K, Hacker MR, Sakkas D, Dodge LE. Burden of care is the primary reason why insured women terminate in vitro fertilization treatment. Fertil Steril. 2018;109(6):1121-1126. doi:10.1016/j.fertnstert.2018.02.130
Péloquin S, Garcia-Velasco JA, Blockeel C, et al. Educational needs of fertility healthcare professionals using ART: a multi-country mixed-methods study. Reprod Biomed Online. 2021;43(3):434-445. doi:10.1016/j.rbmo.2021.06.020
Carson A, Webster F, Polzer J, Bamford S. The power of potential: Assisted reproduction and the counterstories of women who discontinue fertility treatment. Soc Sci Med. 2021;282:114153. doi:10.1016/j.socscimed.2021.114153
Alirezaei S, Taghipour A, Latifnejad Roudsari R. The effect of infertility counseling interventions on marital and sexual satisfaction of infertile couples: A systematic review and meta-analysis. Int J Reprod Biomed. 2022;20(10):795-806. Published 2022 Nov 2. doi:10.18502/ijrm.v20i10.12264
Katz P, Showstack J, Smith JF, et al. Costs of infertility treatment: results from an 18-month prospective cohort study. Fertil Steril. 2011;95(3):915-921. doi:10.1016/j.fertnstert.2010.11.026
Ryan GL, Mengeling MA, Booth BM, Torner JC, Syrop CH, Sadler AG. Voluntary and involuntary childlessness in female veterans: associations with sexual assault. Fertil Steril. 2014;102(2):539-547. doi:10.1016/j.fertnstert.2014.04.042
Feferkorn I, Goldberg A, Bonchek M, et al. Fertility treatment after sexual trauma. Reprod Biomed Online. 2025;51(4):105022. doi:10.1016/j.rbmo.2025.105022
Morbidity and Mortality Weekly Report. Centers for Disease Control and Prevention. February 2018. Accessed February 2023. Available at: https://www.cdc.gov/mmwr/volumes/67/ss/ss6703a1.htm
Farquhar C, Marjoribanks J. Assisted reproductive technology: an overview of Cochrane Reviews. Cochrane Database Syst Rev. 2018;8(8):CD010537. Published 2018 Aug 17. doi:10.1002/14651858.CD010537.pub5
Sunderam S, Kissin DM, Zhang Y, et al. Assisted Reproductive Technology Surveillance - United States, 2018. MMWR Surveill Summ. 2022;71(4):1-19. Published 2022 Feb 18. doi:10.15585/mmwr.ss7104a1
Centers for Disease Control. Policy Documents. Assisted Reproductive Technology (ART). ART Success Rates. CDC. February 7, 2025. Accessed April 24, 2026. https://www.cdc.gov/art/success-rates/index.html
CLOMIPHENE CITRATE- clomiphene citrtae tablet. Prescribing Information. Appco Pharma LLC. Updated November 18, 2024. Accessed April 24, 2026. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=2d257ec2-827c-4bc2-8f17-cf24520c3cad
Yang AM, Cui N, Sun YF, Hao GM. Letrozole for Female Infertility. Front Endocrinol (Lausanne). 2021;12:676133. Published 2021 Jun 16. doi:10.3389/fendo.2021.676133
LETROZOLE- letrozole tablets tablet, film coated. Prescribing Information. Avet Pharmaceuticals Inc. Updated September 30, 2025. Accessed April 24, 2026. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=fe0e4d4b-436d-a36d-e053-6294a90a973d
Wang R, Kim BV, van Wely M, et al. Treatment strategies for women with WHO group II anovulation: systematic review and network meta-analysis. BMJ. 2017;356:j138. Published 2017 Jan 31. doi:10.1136/bmj.j138
Franik S, Eltrop SM, Kremer JA, Kiesel L, Farquhar C. Aromatase inhibitors (letrozole) for subfertile women with polycystic ovary syndrome. Cochrane Database Syst Rev. 2018;5(5):CD010287. Published 2018 May 24. doi:10.1002/14651858.CD010287.pub3
Legro RS, Brzyski RG, Diamond MP, et al; NICHD Reproductive Medicine Network. Letrozole versus clomiphene for infertility in the polycystic ovary syndrome. N Engl J Med. 2014;371(2):119-29. doi: 10.1056/NEJMoa1313517
Weiss NS, Kostova E, Nahuis M, Mol BWJ, van der Veen F, van Wely M. Gonadotrophins for ovulation induction in women with polycystic ovary syndrome. Cochrane Database Syst Rev. 2019;1(1):CD010290. Published 2019 Jan 16. doi:10.1002/14651858.CD010290.pub3
MENOPUR- menotropins kit. Prescribing Information. Ferring Pharmaceuticals Inc. Updated May 31, 2018. Accessed April 24, 2026. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=22c8db95-c3db-1770-8086-31356fbabe35
GONAL-F- follitropin alfa kit. Prescribing Information. EMD Serono, Inc. Updated September 2, 2025. Accessed April 24, 2026. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=ae85b9c3-e7eb-4981-a5d9-346050e8f189
FOLLISTIM AQ- follitropin injection, solution. Prescribing Information. Merck Sharp & Dohme LLC. Updated October 3, 2025. Accessed April 24, 2026. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=96f11b5d-cc7b-48aa-830a-a28e1962d871
OVIDREL- choriogonadotropin alfa injection, solution. Prescribing Information. EMD Serono, Inc. Updated December 26, 2023. Accessed April 24, 2026. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=a683e58a-63ea-44b8-a326-1a99a537bcf2
ELIGARD- leuprolide acetate kit. Prescribing Information. TOLMAR Inc. Updated April 29, 2019. Accessed April 24, 2026. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=b78d1919-9dee-44fa-90f9-e0a26d32481d
CETRORELIX ACETATE- cetrorelix acetate for injection kit. Prescribing Information. Apotex Corp. Updated June 12, 2025. Accessed April 24, 2026. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=95b852d6-26a4-4aaf-a4c3-26340a24723d
GANIRELIX ACETATE injection. Prescribing Information. Amphastar Pharmaceuticals, Inc. Updated April 19, 2022. Accessed April 24, 2026. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=9c286f64-8039-44ec-8fdd-d1a2f52b4c3c
New Zealand Medicines and Medical Devices Safety Authority. NEW ZEALAND DATA SHEET 1 PRODUCT NAME CETROTIDE ® 250 Microgram Powder for Injection Vial with Diluent Syringe. 2020. Accessed April 24, 2026. https://www.medsafe.govt.nz/profs/datasheet/c/Cetrotideinj.pdf
Reindollar RH, Regan MM, Neumann PJ, et al. A randomized clinical trial to evaluate optimal treatment for unexplained infertility: the fast track and standard treatment (FASTT) trial. Fertil Steril. 2010;94(3):888-899.doi:10.1016/j.fertnstert.2009.04.022
Wen J, Jiang J, Ding C, et al. Birth defects in children conceived by in vitro fertilization and intracytoplasmic sperm injection: a meta-analysis. Fertil Steril. 2012;97(6):1331-1337. doi:10.1016/j.fertnstert.2012.02.0
Davies MJ, Moore VM, Willson KJ, et al. Reproductive technologies and the risk of birth defects. N Engl J Med. 2012;366(19):1803-1813. doi: 10.1056/NEJMoa1008095
Skalkidou A, Sergentanis TN, Gialamas SP, et al. Risk of endometrial cancer in women treated with ovary-stimulating drugs for subfertility. Cochrane Database Syst Rev. 2017;3(3):CD010931. doi:10 1002/14651858.CD010931.pub2
Qin J, Wang H, Sheng X, Liang D, Tan H, Xia J. Pregnancy-related complications and adverse pregnancy outcomes in multiple pregnancies resulting from assisted reproductive technology: a meta-analysis of cohort studies. Fertil Steril. 2015;103(6):1492-508.e1, 7. doi:10.1016/j.fertnstert. 2015.03.018
Resolve. The National Infertility and Family Building Association. resolve.org. 2026. Accessed April 24, 2026. https://resolve.org/
Lukish DA, Cross CI, Gornet ME, Christianson MS. Teaming in the contemporary fertility clinic: creating a culture to optimize patient care. Fertil Steril. 2022;117(1):15-21. doi:10.1016/j.fertnstert.2021.09.032
Romeiro J, Caldeira S, Venicios Lopes M. "Impaired Resilience (00210)" in patients under fertility treatment: Clinical validation study [published online ahead of print, 2022 Aug 29]. Int J Nurs Knowl. 2022;10.1111/2047-3095.12391. doi:10.1111/2047-3095.12391
DISCLAIMER
The information provided in this course is general in nature, and it is designed solely to provide participants with continuing education credit(s). This course and materials are not meant to substitute for the independent, professional judgment of any participant regarding that participant’s professional practice, including but not limited to patient assessment, diagnosis, treatment, and/or health management. Medical and pharmacy practices, rules, and laws vary from state to state, and this course does not cover the laws of each state; therefore, participants must consult the laws of their state as they relate to their professional practice.
Healthcare professionals must consult their employer, healthcare facility, hospital, or other organization for guidelines, protocols, and procedures to follow. The information provided in this course does not replace those guidelines, protocols, and procedures, but is for academic purposes only, and this course’s limited purpose is for the completion of continuing education credits.
Participants are advised and acknowledge that information related to medications, their administration, dosing, contraindications, adverse reactions, interactions, warnings, precautions, or accepted uses is constantly changing. Any person taking this course understands that such a person must make an independent review of medication information before any patient assessment, diagnosis, treatment and/or health management. Any discussion of off-label use of any medication, device, or procedure is informational only, and such uses are not endorsed hereby.
Nothing contained in this course represents the opinions, views, judgments, or conclusions of RxCe.com LLC. RxCe.com LLC is not liable or responsible to any person for any inaccuracy, error, or omission with respect to this course or course material.
© RxCe.com LLC 2026: All rights reserved. No reproduction of all or part of any content herein is allowed without the prior, written permission of RxCe.com LLC.
RxCe.com
© RxCe.com LLC 2025: All rights reserved.

