URINARY TRACT INFECTIONS: AN INTERPROFESSIONAL APPROACH TO DIAGNOSIS, TREATMENT, AND STEWARDSHIP
Faculty:
L. Austin Fredrickson, MD, FACP
L. Austin Fredrickson is an Associate Professor of Internal Medicine at Northeast Ohio Medical University, where he serves as core faculty and teaches diagnostics, therapeutics, clinical skills, and health humanities. He is board-certified in general internal medicine and practices rural primary care.
Liz Fredrickson, PharmD, BCPS
Liz Fredrickson, PharmD, BCPS, is an Associate Professor of Pharmacy Practice and Pharmaceutical Sciences at the Northeast Ohio Medical University (NEOMED) College of Pharmacy, where she is course director of the Parenteral Products and Basic Pharmaceutics Lab courses.
Pamela Sardo, PharmD, BS
Pamela Sardo, PharmD, BS, is a freelance medical writer and licensed pharmacist. She is the founder and principal at Sardo Solutions in Texas. Pam received her BS from the University of Connecticut and her PharmD from the University of Rhode Island. Pam’s career spans many years in retail, clinics, hospitals, long-term care, Veterans Affairs, and managed health care responsibilities across a broad range of therapeutic classes and disease states.
Abstract
Urinary tract infections are among the most prevalent bacterial infections encountered in clinical practice, affecting individuals across all age groups and presenting distinct challenges in diagnosis, treatment, and prevention. The treatment of urinary tract infections involves eradicating the causative organism and averting recurrent infections. Complications associated with this condition may also arise. Antibiotics are used in treatment, but the causative organisms can become resistant to them. As the landscape of antibiotic resistance continues to evolve alongside shifts in patient demographics, it is increasingly imperative for healthcare professionals to stay well-informed about the latest insights and treatment strategies for urinary tract infections.
Accreditation Statements
In support of improving patient care, RxCe.com LLC is jointly accredited by the Accreditation CouncilTM for Continuing Medical Education (ACCME®), the Accreditation Council for Pharmacy Education (ACPE®), and the American Nurses Credentialing Center (ANCC®), to provide continuing education for the healthcare team.

This activity was planned by and for the healthcare team, and learners will receive 2 Interprofessional Continuing Education (IPCE) credits for learning and change.
Joint Universal Activity Number: The Joint Accreditation Universal Activity Numbers assigned to this activity are as follows:
Pharmacists: JA4008424-0000-26-047-H01-P
Pharmacy Technicians: JA4008424-0000-26-047-H01-T
Credits: 2 contact hour(s) (0.2 CEU(s)) of continuing education credit.
Credit Types:
IPCE Credits - 2 Credits
Pharmacy Credits- 2 Credits
Type of Activity: Application
Media: Computer-Based Training (i.e., online courses)
Estimated time to complete activity: 2 contact hour(s) (0.2 CEU(s)), including Course Test and course evaluation.
Release Date: April 9, 2026 Expiration Date: April 9, 2029
Target Audience: This educational activity is for Pharmacists and Pharmacy Technicians
How to Earn Credit: From April 9, 2026, through April 9, 2029, participants must:
Read the “learning objectives” and “author and planning team disclosures;”
Take the “Educational Activity Pre-Test;”
Study the section entitled “Educational Activity;” and
Complete the Educational Activity Post-Test and Activity Evaluation. The Educational Activity Post-Test will be graded automatically. Following successful completion of the Educational Activity Post-Test with a score of 70% or higher, a statement of participation will be made available immediately. (No partial credit will be given.)
CME Credit: Credit for this course will be uploaded to CPE Monitor® for pharmacists. Physicians may receive AMA PRA Category 1 Credit™️ and use these credits toward Maintenance of Certification (MOC) requirements. Physician Assistants may earn AAPA Category 1 CME credit, reportable through PA Portfolio. All learners shall verify their individual licensing board’s specific requirements and eligibility criteria.
Statement of Need
Urinary tract infections (UTIs) commonly affect both inpatient and outpatient populations. Misdiagnosis and inappropriate antibiotic use remain a healthcare challenge. Healthcare professionals must understand anatomy and pathophysiology to differentiate between uncomplicated and complicated infections. Identifying common causative organisms, such as E. coli, Klebsiella, and others, remains important. Accurate recognition of clinical signs and symptoms is essential for guiding diagnostic testing and reducing unnecessary antibiotic exposure. Evidence-based decision-making includes determining when to utilize urinalysis, urine culture, and point-of-care testing. A personalized approach to pharmacologic management requires understanding the mechanisms of action, adverse effects, and strategies to combat antimicrobial resistance. Optimal management of patients receiving pharmaceutical treatment often requires coordinated involvement across the healthcare team. This activity aims to address gaps in managing UTIs across diverse patient populations.
Learning Objectives: Upon completion of this educational activity, participants should be able to:
Describe the anatomy and pathophysiology of urinary tract infections, including common microorganism causes
Recall the clinical presentation of UTI, including signs and symptoms
Compare and contrast approaches to diagnosing UTI
Describe the pharmacologic management of UTIs, including mechanisms of action and side effects of commonly utilized antibiotic agents
Disclosures
The following individuals were involved in planning, developing, and/or authoring this activity: L. Austin Fredrickson, MD, FACP; Liz Fredrickson, PharmD, BCPS; and Pamela Sardo, PharmD, BS. None of the individuals involved in developing this activity has a conflict of interest or financial relationships related to the subject matter. There are no financial relationships or commercial or financial support relevant to this activity to report or disclose by RxCe.com or any of the individuals involved in the development of this activity.
© RxCe.com LLC 2026: All rights reserved. No reproduction of all or part of any content herein is allowed without the prior, written permission of RxCe.com LLC.
Educational Activity Pre-Test
According to the 2025 Infectious Diseases Society of America guidelines, what symptoms are indicative of a complicated urinary tract infection (UTI)?
Dysuria without fever
Infection confined to the bladder
Systemic symptoms such as fever and flank pain
Infection in a healthy, premenopausal woman
In severe sepsis due to suspected multidrug-resistant E. coli, which of the following empiric options is preferred?
Nitrofurantoin
Piperacillin–tazobactam
Cefepime–tazobactam
Meropenem–vaborbactam
Which antibiotic should generally be avoided in early pyelonephritis?
Fosfomycin
Levofloxacin
Cefepime
Piperacillin-tazobactam
Educational Activity
Urinary Tract Infections:
An Interprofessional Approach to Diagnosis, Treatment, and Stewardship
Introduction
Urinary tract infections (UTIs) remain among the most frequently diagnosed bacterial infections worldwide, accounting for a significant share of outpatient visits, antibiotic prescriptions, and healthcare expenditures.1-4 Despite their common occurrence, UTIs pose ongoing clinical challenges in accurate diagnosis, optimal antibiotic selection, and prevention of recurrence.2 The management of these infections is further complicated by the rising prevalence of antimicrobial resistance, including extended-spectrum β-lactamase (ESBL)-producing and multidrug-resistant (MDR) pathogens that increasingly limit treatment options.5
Recent updates to national and international guidelines, including the 2025 Infectious Diseases Society of America (IDSA) guidance on complicated urinary tract infections, have revised definitions, diagnostic criteria, and empiric treatment recommendations to reflect this changing landscape.2 These updates emphasize shorter, evidence-based treatment durations, improved diagnostic stewardship, and the importance of tailoring antibiotic selection to local resistance data and patient-specific factors.2
This continuing education activity provides an interprofessional overview of contemporary approaches to UTI diagnosis, management, and prevention. Participants will learn about the latest guideline-based recommendations, pharmacologic and nonpharmacologic treatment strategies, and key principles of antimicrobial stewardship. By integrating these updates into practice, pharmacists, pharmacy technicians, and other healthcare professionals can play a critical role in improving patient outcomes, reducing recurrence, and optimizing antimicrobial stewardship.
Definitions and Classification
The IDSA has introduced a revised framework for classifying UTIS, shifting away from prior anatomic and demographic distinctions to a model based primarily on clinical presentation and severity (Table 1).2 In the updated classification, the distinction between uncomplicated and complicated UTIs is now based on the presence or absence of systemic findings; that is, complicated UTIs are characterized by the presence of systemic findings, such as fever, flank pain, or other signs of systemic illness, which indicate that the infection extends beyond the bladder.2 The revised approach emphasizes readily observable factors at the point of care, including vital signs and the presence of urinary catheters, rather than structural or functional urinary abnormalities requiring urologic evaluation.2
Under previous guidelines, uncomplicated UTIs were defined as acute cystitis in afebrile, nonpregnant, premenopausal women without diabetes or urologic abnormalities, while complicated UTIs encompassed all other infections, including those in men and patients with comorbidities or anatomic abnormalities.2 The new IDSA classifications instead define uncomplicated UTI as an infection confined to the bladder in afebrile women or men, while complicated UTI refers to infection extending beyond the bladder, such as pyelonephritis, febrile or bacteremic UTIs, or catheter-associated UTIs (CAUTIs).2 Cases of prostatitis are considered separately and are not covered under these guidelines.2
Urinary tract infections are considered recurrent when two or more occur within six months, or three or more occur within a year.1 If a different organism causes the second or third UTI, this is referred to as a reinfection.1 A relapse is a repeat infection with the initial organism, suggesting a persistent infectious source may be present.1
Asymptomatic bacteriuria (ASB) occurs when significant bacteriuria (greater than 105 bacteria/mL) is present despite no signs or symptoms of a UTI.1 This is more common among patients 65 years of age and older. Significant bacteriuria is the presence of microorganisms that cause a true infection rather than contamination of the urine.1
Table 1
Comparison of Previous and Updated IDSA Classifications of Urinary Tract Infections2
| Previous Classifications | Updated IDSA 2025 Classifications |
| Uncomplicated UTI: Acute cystitis in afebrile, nonpregnant, premenopausal women with no diabetes or urologic abnormalities. | Uncomplicated UTI: Infection confined to the bladder in afebrile women or men. |
| Acute Pyelonephritis: Acute kidney infection in women otherwise meeting the definition of uncomplicated UTI. | Complicated UTI: Infection extending beyond the bladder in women or men, including pyelonephritis, febrile or bacteremic UTI, and catheter-associated UTI (CAUTI). |
| Complicated UTI: All other UTIs (e.g., in men, diabetics, or those with structural abnormalities). | Prostatitis: Addressed separately and not included in these guideline recommendations. |
Epidemiology
Urinary tract infections are among the most common bacterial infections worldwide, accounting for an estimated 150 to 250 million cases annually and representing the most frequent healthcare-associated infection across hospital and community settings.1,5 They can affect individuals of all ages and genders, but occur most commonly in women, largely due to anatomical and hormonal factors that facilitate bacterial colonization.1,5 Epidemiologic studies indicate that up to 40% of women experience at least one symptomatic UTI during their lifetime, and approximately half will have an episode before age 35.5 Urinary tract infections are less common in men, affecting roughly 12–14% over a lifetime, although the prevalence increases with age and urinary tract abnormalities.5
The highest incidence of UTI is observed in sexually active women aged 16 to 64 years, with risk factors including sexual activity, poor hygiene, use of spermicides, urinary catheterization, and structural or functional urinary abnormalities.5 (See Table 2.) Globally, rising antimicrobial resistance among uropathogens has become a critical public health concern.5 Surveillance studies have demonstrated increasing rates of multidrug resistance, particularly in E. coli and K. pneumoniae, driven by widespread empiric antibiotic use without culture confirmation or susceptibility testing.5 These resistance trends contribute to longer hospital stays, higher healthcare costs, and increased morbidity and mortality, emphasizing the importance of antimicrobial stewardship and adherence to updated guidelines for diagnosis and management by all members of the healthcare team.5
Table 2
Urinary Tract Infections Risk Factors1-5
| Risk Factor Category | Examples / Details | Mechanism / Clinical Implication |
| Anatomic / Physiologic Factors | Female sex; Short urethral length; Postmenopausal estrogen deficiency; Pregnancy; Aging and bladder changes | Facilitate bacterial ascent and urinary stasis; hormonal changes alter protective vaginal flora. |
| Behavioral Factors | Sexual activity; Use of spermicides or diaphragms; Delayed voiding habits; Poor perineal hygiene | Promote bacterial entry and disrupt normal flora balance |
| Iatrogenic Factors | Urinary Catheterization Instrumentation (e.g., cystoscopy); Recent antibiotic therapy | Provide direct access for bacteria or select for resistant strains. |
| Comorbid Conditions | Diabetes mellitus; Immunosuppression; Chronic kidney disease; Neurogenic bladder; Urinary tract obstruction (stones, strictures) | Impair host defenses or promote urinary retention, predisposing to complicated infections. |
| Recurrent UTI History | ≥2 infections in 6 months or ≥3 in 12 months | Indicates persistent colonization or uncorrected risk factors requiring further evaluation. |
Etiology
The most frequently isolated uropathogen remains Escherichia coli, followed by Klebsiella pneumoniae, Proteus species, and Enterobacter species.1,5 While fungal UTIs are less common, they do occur more frequently in patients with diabetes, recent antibiotic exposure, or indwelling catheters.1,5
In uncomplicated UTIs, the primary pathogens include Escherichia coli (E. coli), which accounts for 75-90% of community-acquired infections, as well as Staphylococcus saprophyticus, Klebsiella pneumoniae, Proteus spp., Pseudomonas aeruginosa, and Enterococcus spp.1 Notably, microorganisms such as lactobacilli, enterococci, Group B streptococci, and coagulase-negative staphylococci (excluding S. saprophyticus) are more likely to signify specimen contamination.3,6
Frequently encountered microorganisms in complicated UTIs include E. coli, Proteus spp., K. pneumoniae, Enterobacter spp., P. aeruginosa, staphylococci, and enterococci.1 Enterococci species have also emerged as a prevalent cause of UTIs for hospitalized patients, and vancomycin-resistant strains, including E. faecalis and E. faecium, that are more common among patients with prolonged hospital stays or residing in institutional healthcare settings.1
While UTIs are typically attributed to a single microorganism, certain patients, such as those with indwelling catheters or kidney stones, may have multiple pathogens because they can ‘seed’ more easily.1 In these cases, it is recommended that clinicians verify the causative bacteria through a repeat evaluation, as the presence of multiple microorganisms could possibly also be indicative of contamination.1,6
It is also essential to acknowledge that pregnancy can be a significant predisposing factor for UTIs, with an estimated 5-10% of women experiencing a UTI during their pregnancy.1,2 Within the spectrum of pregnancy-associated UTIs, bacteriuria can manifest as asymptomatic bacteriuria (ASB), cystitis, or pyelonephritis.1,2
Pathophysiology
The initiating event of a UTI is inoculation.7 Normally, the urinary tract is resistant to invading microorganisms.1 Important factors contributing to the infection are host factors, inoculum size, and the virulence of the microorganism.1,7 Bacteria can gain access to the urinary tract via three routes. These include the ascending, descending, and lymphatic pathways.1,7 As noted previously, UTIs are more likely in female patients, given their anatomic location and length of the urethra, and ascending UTIs are the most common.1 The female urethra can become colonized by bacteria that originate from fecal or vaginal flora due to proximity and activity.1 Descending pathways, including infection from an adjacent intra-abdominal organ or fistula, are less common. Lymphatics can also serve as pathways, typically from nearby infected tissues.1
Clinical Presentation
An uncomplicated UTI is confined to the bladder and does not suggest infection beyond the lower urinary tract.3 Patients with uncomplicated infections generally lack fever, flank pain, or systemic features such as chills, rigors, or hemodynamic instability unless attributable to another cause.3 Patients typically present with the following symptoms:3
Dysuria
Urinary urgency
Increased frequency
Suprapubic pain or discomfort
Complicated UTIs, on the other hand, are defined by symptoms or findings indicating that the infection has extended beyond the bladder.3 Common features include the following:3
Fever
Flank pain
Costovertebral angle tenderness
Other systemic signs (e.g., chills, rigors, malaise, hemodynamic instability)
Pyelonephritis and UTIs associated with transurethral, suprapubic, or intermittent catheterization are classified as complicated infections.3 In these patients, systemic manifestations or upper tract involvement must be presumed until ruled out.3
Older adults often present with non-specific or atypical symptoms, such as altered mental status, poor appetite, or gastrointestinal changes, rather than classic urinary complaints.1 In older women, acute-onset dysuria combined with new or worsening urinary symptoms remains a key diagnostic indicator.8,9 Other non-specific findings may include cloudy urine, pelvic discomfort, or transient urinary incontinence.8,9 Chronic symptoms such as nocturia or baseline incontinence are not specific for UTI.8,9
Diagnosing Urinary Tract Infections
In the diagnostic process for bacterial UTIs, it is crucial to emphasize that symptoms alone should not be the sole basis for diagnosis.1 Instead, a comprehensive approach involving physical examination and laboratory testing should be employed to differentiate between contamination and infection.1 Central to this diagnostic evaluation is the collection and examination of urine samples.1 The preferred method for this purpose is the midstream clean-catch technique, which involves voiding the first 20mL of urine and then collecting the subsequent portion of the urinary flow.1
Diagnosis of a UTI depends on isolating a substantial number of bacteria from a urine specimen.1,5 Specifically, when 105 colony-forming units per milliliter (CFU/mL) or more bacteria are detected, it indicates the presence of a UTI.1,5 However, it is important to note that approximately 50% of women presenting with UTI symptoms may exhibit lower bacterial counts.1 In several studies, a threshold of 102 CFU/mL or greater has demonstrated an 88-93% positive predictive value for bladder bacteriuria in patients with a strong suspicion of UTI.6 Table 3 lists uropathogens and the colony count thresholds used to diagnose UTIs.
Table 3
Urinary Tract Infections Pathogens
| Bacterial Pathogenicity | Representative Species | Colony Count Threshold |
| Primary pathogens | Escherichia coli, Staphylococcus saprophyticus | ≥ 10³ CFU/mL |
| Secondary pathogens | Enterobacter spp., Enterococcus spp., Klebsiella spp., Pseudomonas aeruginosa, Proteus mirabilis | Women: ≥ 10⁴ CFU/mL † Men: ≥ 10³ CFU/mL ‡ Women and men: ≥ 10⁵ CFU/mL § |
| Uncommon pathogens | Group B Streptococcus and other less common organisms | ≥ 10⁵ CFU/mL |
The confirmation of pyuria, which signifies the presence of leukocytes, further supports the diagnosis of significant bacteriuria when observed in symptomatic patients.1 Pyuria is defined as a white blood cell (WBC) count exceeding 10 x 106/L.1 It is imperative to recognize that pyuria is indicative of inflammation but not necessarily indicative of infection.1 To detect pyuria, a leukocyte esterase dipstick test can be effectively employed. Furthermore, it is essential to underscore that hematuria, the presence of blood in the urine, is nonspecific to UTIs.1 Dipstick tests can also be utilized to determine the presence of nitrite in urine, which is produced by bacteria as they reduce nitrate to nitrite, serving as an additional diagnostic indicator.1
Table 4
Urinary Tract Infection Symptoms
| Category | Acute Simple Cystitis | Acute Complicated UTI |
| Key Clinical Features | Dysuria (painful urination) Urinary frequency Urinary urgency Suprapubic pain | Classic cystitis symptoms plus one or more:
Pelvic or perineal pain in males (prostatic involvement) |
| Alternative Presentations | New incontinence Acute-on-chronic dysuria or urgency | Flank pain or CVA tenderness without cystitis symptoms Fever or sepsis without another identifiable cause |
| Laboratory Findings | Pyuria and bacteriuria are present Positive leukocyte esterase or nitrite >10 WBCs/microliter on microscopy | Pyuria and bacteriuria are present Leukocyte casts may be present (suggests upper tract infection) |
| Urine Studies (When Needed) | Not essential for healthy females with previous cystitis and classic symptoms | Recommended for all suspected cases |
| Urine Culture | Not required in most recurrent, mild, or previously treated uncomplicated cases | Strongly recommended to guide therapy and assess resistance |
| Sample Collection | Midstream clean-catch specimen (labia spread for females) | Same method; ensure sterile collection for accurate culture |
| Key Points | Usually localized infection Lower urinary tract only | Potential for systemic infection or sepsis Requires broader evaluation and empiric therapy |
Pause and Ponder You are counseling a patient who comes to your office for her third antibiotic for a “UTI” in the past six months. She reports that each time symptoms improve briefly but then return, and she’s now worried her “infections just keep coming back.” What risk factors, diagnostic clues, or stewardship principles would you use to guide the next step in her care? |
Management of Urinary Tract Infections
Goals of Therapy
The management of UTIs focuses on four primary objectives:1
Eradicating the causative organism
Preventing or addressing complications
Reducing the risk of recurrence
Mitigating the development of antimicrobial resistance
Achieving these goals requires a systematic approach that includes careful diagnostic evaluation, evidence-based antibiotic selection, appropriate determination of treatment duration, and diligent follow-up to ensure complete resolution and minimize recurrence.1-3
Antimicrobial therapy remains the cornerstone of UTI management.1-3 When selecting an appropriate agent, care teams should consider several critical factors, including the following:1-3
Severity of illness (e.g., presence of sepsis or systemic involvement)
Site of infection (lower vs. upper urinary tract)
Classification (complicated vs. uncomplicated)
Local resistance patterns and antibiograms
Patient-specific factors, such as allergies, renal function, pregnancy status, and recent antimicrobial exposure
The Four-Step Approach
The updated IDSA guidelines recommend a structured four-step approach to guide empiric antibiotic selection for complicated urinary tract infections (cUTIs).3 This stepwise process promotes precision, minimizes the development of resistance, and aligns empiric therapy with current evidence-based recommendations.3
Step One: Assess Severity- Clinicians should assess the severity of illness, determining whether the patient is stable, febrile, or septic, as this dictates the urgency and route of therapy.3
Step Two: Evaluate Resistance Risk- Evaluate risk factors for antimicrobial resistance, including prior urine culture results, recent antibiotic exposure, and local resistance trends.3
Step Three: Consider Patient-Specific Factors- Consider patient-specific factors such as drug allergies, comorbid conditions, renal function, pregnancy status, and potential drug interactions to ensure both safety and efficacy.3
Step Four: Use Local Antibiogram- Care teams should use local antibiogram data, particularly in septic patients, to select empiric therapy guided by local susceptibility patterns and sepsis risk modeling.3
Nonpharmacologic Therapies
Beyond pharmacologic therapy, nonpharmacologic interventions can play an important role in UTI management and prevention. Increasing fluid intake is recommended to promote diuresis, dilute bacterial concentration, and help flush pathogens from the urinary tract.1 Cranberry juice or extracts have been proposed as adjunctive prophylactic measures, with potential benefits attributed to fructose and proanthocyanidins, which interfere with bacterial adhesion to uroepithelial cells.1 Although clinical evidence remains inconsistent, some studies suggest that individuals with recurrent UTIs may benefit from daily cranberry products providing approximately 36 mg of proanthocyanidins.1 In contrast, the use of ascorbic acid (vitamin C) to acidify urine is not recommended, as it has not been shown to significantly alter urinary pH or prevent bacterial growth.1
For patients experiencing bothersome dysuria or suprapubic discomfort, the urinary analgesic phenazopyridine may provide short-term symptom relief.1,10 This medication acts locally on the bladder mucosa to reduce irritation and discomfort, though it does not treat the underlying infection.10 The recommended dosage is 200 mg taken orally up to three times daily, with use limited to one to two days in conjunction with antimicrobial therapy.10 Common side effects include orange-red discoloration of urine and body fluids, while rare adverse effects include hypersensitivity reactions and, infrequently, acute renal injury.10
Pharmacologic Management
Acute, uncomplicated UTIs are the most common form and most likely to occur in females of childbearing age.1 Therapy should target E. coli, as this is the most common microorganism.1 If patients present with lower tract symptoms and significant bacteriuria is present, a short-course therapy of antibiotics can be initiated (Table 5).1 Alternative therapies are presented in Table 6.1 If clinical failure occurs, a urine culture can be performed to determine the presence of microbes.1 If this is positive, the patient can be retreated for two weeks with a follow-up urine culture.1 If the culture remains positive after two weeks, whether a relapse or reinfection is present should be determined.1 Relapses should prompt a urologic work-up, while reinfection, if frequent, should prompt a discussion about urologic hygiene, risk factors, and possibly suppressive postcoital therapy.1 Fluoroquinolones are not recommended for initial therapy and should be saved for patients with pyelonephritis.1
Table 5
First-Line Options for Acute, Uncomplicated Urinary Tract Infections
| Drug | Dose | Duration |
| Nitrofurantoin extended release 11 | 100 mg orally twice daily | 5 days (female) 7 days (male) |
| Trimethoprim-sulfamethoxazole (TMP-SMX)12 | One double-strength tablet (160 mg/800 mg) orally twice daily | 3 days (female) 7 days (male) |
| Pivmecillinam13 | 400 mg HCl orally three times daily | 3-5 days (female) 7 days (male) |
| Fosfomycin14 | Single 3-gram dose | Single dose |
Table 6
Alternative Options for Acute, Uncomplicated Urinary Tract Infections
| Drug | Dose | Duration (Females) | Duration (Males) | Notes / Key Considerations |
Amoxicillin- clavulanate15 | 500 mg orally twice daily | 5–7 days | 7 days | Dose based on the amoxicillin component. |
| Cefadroxil16 | 500 mg orally twice daily | 5–7 days | 7 days | — |
| Cefpodoxime17 | 100 mg orally twice daily | 5–7 days | 7 days | — |
| Cephalexin18 | 500 mg orally twice daily | 5–7 days | 7 days | — |
| Cefdinir19 | 300 mg orally twice daily | 5–7 days | 7 days | Does not achieve high urinary concentrations but may be effective for cystitis with susceptible organisms |
| Ciprofloxacin20,21 | 250 mg twice daily or 500 mg ER once daily; Cipro XR extended release form not available in the U.S. | 3 days | 5 days | Useful for males with possible prostatitis. |
| Levofloxacin22 | 250 mg orally once daily | 3 days | 5 days | Useful for males with possible prostatitis. |
| Gepotidacin23 | 1500 mg (two 750 mg tablets) orally twice daily | 5 days | Uncertain | For infections with no other options. Retains activity against MDR organisms. Avoid if: Concern for early pyelonephritis. Not well studied in males. |
Complicated Urinary Tract Infections
Patients who present with high-grade fevers and severe flank pain necessitate immediate attention for the management of acute pyelonephritis.1 While milder cases may be effectively treated with oral antibiotics, severely ill patients will invariably require intravenous antibiotics and hospital admission.1 The duration of oral antibiotic therapy typically spans 7 to 14 days, with the specific choice of agent influencing the duration.1
In cases of mild to moderate pyelonephritis, fluoroquinolones are the first-line treatment, with TMP-SMX as a viable second-line option.3 For instances of severe pyelonephritis, a regimen with broad-spectrum coverage becomes imperative.1 Here, therapeutic options encompass fluoroquinolones, aminoglycosides in conjunction with or without ampicillin, as well as extended-spectrum cephalosporins either with or without aminoglycosides and carbapenems.1 Furthermore, it is essential to consider the potential involvement of Pseudomonas and enterococci in patients who have been hospitalized within the past six months, those with urinary catheters, or individuals residing in nursing homes.1 These factors warrant heightened clinical suspicion and careful consideration in antibiotic selection. Fosfomycin is not indicated for the treatment of pyelonephritis because it does not achieve therapeutic serum or renal tissue concentrations adequate for this condition.1,14
Complicated UTI Management Algorithm
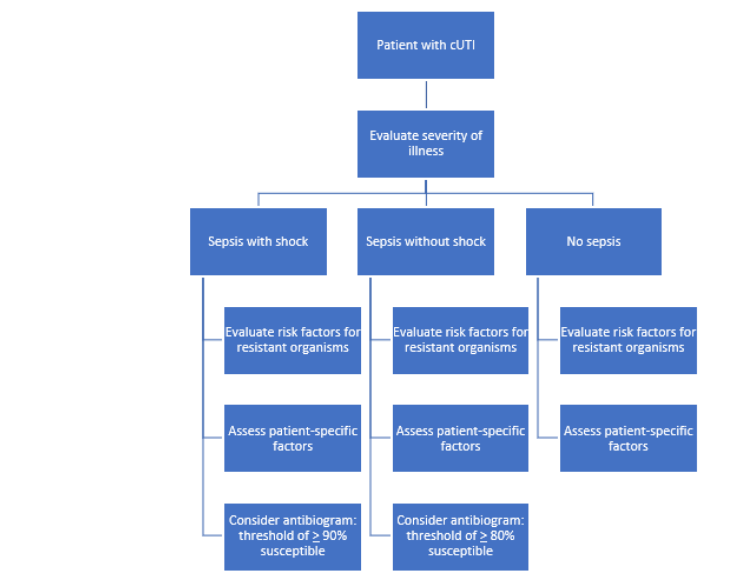
Table 7
Antibiotics for Complicated Urinary Tract Infections
| Condition of the Patient | Preferred Options | Alternative Options |
| Sepsis (with or without shock) | - Third or fourth-generation cephalosporins - Carbapenems -Piperacillin–tazobactam - Fluoroquinolones | - Novel β-lactam/β-lactamase inhibitors - Cefiderocol - Plazomicin - Older aminoglycosides |
| Without sepsis (IV route of therapy) | - Third or fourth-generation cephalosporins - Piperacillin–tazobactam - Fluoroquinolones | - Carbapenems - Newer agents (novel β-lactam/β-lactamase inhibitors, cefiderocol, plazomicin) - Older aminoglycosides |
| Without sepsis (oral route of therapy) | - Fluoroquinolones - Trimethoprim–sulfamethoxazole | - Amoxicillin–clavulanate - Oral cephalosporins |
Table 8
Doses of Antibiotics For UTI From Clinical Trials
| Drug | Dosing Regimen Used in Clinical Trials (Normal Renal Function) |
| Cefepime24 | 1–2 g every 8–12 hours |
| Cefepime–enmetazobactam25 | 2 g / 0.5 g (infused over 2 hours) every 8 hours |
| Cefiderocol26 | 2 g (infused over 3 hours) every 8 hours |
| Cefotaxime27 | 1–2 g every 8 hours |
| Ceftazidime28 | 1–2 g every 8 hours |
| Ceftazidime–avibactam29 | 2.5 g (infused over 2 hours) every 8 hours |
| Ceftolozane–tazobactam30 | 1.5 g every 8 hours |
| Ceftriaxone31 | 1–2 g daily |
| Ertapenem32 | 1 g daily |
| Fosfomycin (IV)33 | 6 g every 8 hours |
| Imipenem–cilastatin34 | 500 mg every 6 hours or 1 g every 8 hours |
Imipenem–cilastatin– relebactam35 | 500 mg / 125 mg every 6 hours |
| Meropenem36 | 1 g every 8 hours |
| Meropenem–vaborbactam37 | 2 g / 2 g (infused over 3 hours) every 8 hours |
| Piperacillin–tazobactam38 | 4.5 g every 8 hours |
| Plazomicin39 | 10–15 mg/kg daily |
Symptomatic Bacteriuria
Symptomatic bacteriuria is present when patients present with dysuria and pyuria, with fewer than 105 bacteria/mL.1 Patients may have some other infection present (such as Chlamydia), which requires treatment.1
Asymptomatic Bacteriuria
Relapse and reinfection are common with ASB, making it challenging to treat and eradicate.1 If patients are not pregnant, treatment is controversial.1 Patients with neurocognitive baseline deficits may find it more difficult to ascertain if symptom changes are due to UTI or another source.
Emerging Antimicrobial Agents in Urinary Tract Infection Management
As antimicrobial resistance among uropathogens continues to increase, several novel antibiotics have expanded the therapeutic landscape for cUTIs. These agents target MDR Gram-negative bacteria, including ESBL and carbapenem-resistant Enterobacterales (CRE), as well as difficult-to-treat Pseudomonas aeruginosa. The most significant additions include new β-lactam/β-lactamase inhibitor (BL/BLI) combinations, cefiderocol, plazomicin, and gepotidacin.23,26,39
Among the BL/BLI combinations, ceftazidime-avibactam, meropenem-vaborbactam, and imipenem-cilastatin-relebactam represent important therapeutic advancements.29,35,37 Ceftazidime-avibactam combines a third-generation cephalosporin with avibactam, a non-β-lactam β-lactamase inhibitor that restores activity against many ESBL-, AmpC-, and KPC-producing organisms.29 This agent demonstrated efficacy comparable to carbapenems and is a valuable alternative for treating infections caused by resistant E. coli and Klebsiella pneumoniae.40,41
Meropenem-vaborbactam combines a carbapenem backbone with vaborbactam, a boronic acid β-lactamase inhibitor that enhances activity against KPC-producing CRE. It maintains the broad Gram-negative coverage and safety profile of meropenem while providing strong activity against several carbapenemase-producing strains. Similarly, imipenem-cilastatin-relebactam extends the spectrum of imipenem by adding relebactam, which inhibits certain ESBLs, AmpC, and KPC enzymes, offering another potent option for infections involving resistant Enterobacterales and P. aeruginosa. Another promising agent, cefepime-taniborbactam, remains under FDA review and is expected to exhibit activity against both serine- and metallo-β-lactamases, including KPC- and NDM-producing strains.42
Beyond the BL/BLI combinations, cefiderocol is a first-in-class siderophore cephalosporin that uses an iron-transport mechanism to penetrate bacterial cell walls. Cefiderocol demonstrates potent in vitro activity against a wide range of MDR Gram-negative pathogens, including those producing ESBLs, AmpC, KPC, and NDM, as well as Acinetobacter baumannii. It is currently approved for the treatment of cUTI, including pyelonephritis. Cefiderocol’s unique mechanism provides stability against all four Ambler β-lactamase classes (A, B, C, and D), though clinicians should exercise caution in severely ill patients, as some studies have reported higher mortality in select populations.
Plazomicin is a next-generation aminoglycoside developed to overcome traditional aminoglycoside resistance mechanisms.39 It retains activity against several ESBL-producing and carbapenem-resistant Enterobacterales by evading aminoglycoside-modifying enzymes. Administered intravenously once daily, plazomicin has been shown to achieve high urinary concentrations, making it particularly useful for cUTIs caused by MDR Gram-negative bacteria when first-line options are limited.39 However, as with all aminoglycosides, clinicians must monitor renal function and auditory toxicity closely.39
Recurrent Infections
It is estimated that 80% of recurrent infections are caused by a different microorganism than the initial infection.1 Management is patient-specific and depends on predisposing factors, the number of episodes per year, and the patient’s preference.1,4 It is recommended that clinicians obtain a thorough and complete patient history and perform a pelvic examination in women with recurrent UTIS.4 To diagnose a recurrent infection, clinicians must document positive urine cultures associated with prior symptomatic episodes.4 Further, guidelines recommend clinicians obtain a urinalysis, urine culture, and sensitivity with each symptomatic acute cystitis episode before initiating treatment in patients with recurrent UTIs.4
Asymptomatic bacteriuria should not be treated.4 Management options include self-administered antibiotic therapy, postcoital therapy, and continuous, low-dose prophylaxis.1 First-line therapies include nitrofurantoin, TMP-SMX, and Fosfomycin. The choice depends on the local antibiogram for the treatment of symptomatic UTIs in women.1 Guidelines suggest clinicians offer patient-initiated treatment (self-start treatment) to select patients with acute episodes while awaiting urine cultures.4 Short-course therapies are preferred in these settings.4 Prophylactic therapy options include TMP-SMX (1/2 single-strength tablet), trimethoprim 100 mg daily, levofloxacin 500 mg daily, and nitrofurantoin 50 or 100 mg daily.1 Therapy duration is commonly 6 months.1
Antimicrobial Stewardship
Resistance among various urologic pathogens has increased significantly over the past two decades.4 This includes an increase in ESBL-producing bacteria.4 Because an uncomplicated UTI is among the most common reasons for women to be exposed to antibiotics, it is critical that clinicians practice antimicrobial stewardship to avoid contributing to resistance levels and having their patients experience untoward outcomes.4 Antimicrobial stewardship programs are multifactorial and involve reducing the use of inappropriate treatments, decreasing the use of broad-spectrum antibiotics, utilizing antimicrobial therapy for the shortest, most effective duration, and adhering to current guideline recommendations.4
Collaborative Care Approach
The effective management of UTIs requires coordinated interprofessional collaboration among physicians, pharmacists, nurses, and other healthcare professionals.43,44 Physicians play a significant role in diagnosis and overall clinical decision-making, while pharmacists provide critical input on antimicrobial selection, dosing, renal adjustments, and stewardship principles to optimize outcomes and minimize resistance. Nurses and advanced practice providers (APPs) contribute by early recognition of signs of infection, ensuring proper urine specimen collection, and monitoring treatment response.43
In complex or high-risk cases, infectious disease specialists can provide guidance on advanced therapy options, including newer antimicrobial agents or step-down strategies.43 Collaborative care also extends beyond the acute setting, emphasizing transitions of care, patient education, adherence monitoring, and recurrence prevention through behavioral interventions and management of modifiable risk factors.43,44 By integrating these complementary roles, interprofessional teams can improve diagnostic accuracy, individualize antibiotic therapy, and enhance patient outcomes while promoting responsible antimicrobial use.43
Active Learning
For each clinical statement in Column A, write the letter “A” or “B” in Column B, as appropriate, to match the clinical statement to the correct diagnosis. A = Acute Simple Cystitis, B = Acute Complicated UTI. The correct answers may be found after the course summary.
| Column A – Clinical Statements | Column B – Condition |
| 1. Dysuria, urinary frequency, and urgency without systemic signs | |
| 2. Flank pain or CVA tenderness with or without cystitis symptoms | |
| 3. Not essential to obtain urine studies in healthy females with prior cystitis and classic symptoms | |
| 4. Leukocyte casts suggest possible upper-tract involvement | |
| 5. Strongly recommended to obtain a urine culture to guide therapy | |
| 6. May present with fever, chills, rigors, hypotension, or signs of sepsis |
Patient Case
A 72-year-old woman presents with fever, flank pain, and dysuria. Her last urine culture (3 months ago) showed E. coli resistant to TMP-SMX but susceptible to cefepime and piperacillin–tazobactam.
Complicated UTI Classification: Under the 2025 IDSA definitions, this patient meets criteria for a complicated UTI because her infection demonstrates systemic features—fever and flank pain—indicating extension beyond the bladder, consistent with pyelonephritis. Additionally, her advanced age (≥65 years), potential comorbidities (e.g., renal impairment, postmenopausal status), and history of resistant E. coli infection further support classification as a complicated UTI.
Applying the Four-Step Approach:
Step One: Assess Severity- The presence of fever and flank pain indicates at least a moderate infection; sepsis is possible if vital signs are unstable. Hospitalization and intravenous (IV) therapy are warranted.
Step Two: Evaluate Resistance Risk- Her prior culture revealed TMP-SMX resistance but susceptibility to cefepime and piperacillin-tazobactam. Recent antimicrobial exposure in the past 3 months increases the risk of resistant organisms.
Step Three: Consider Patient-Specific Factors- At 72 years old, renal function should be assessed to guide antibiotic dosing. Allergies, comorbid conditions, and the ability to tolerate IV therapy should also be evaluated.
Step Four: Use Local Antibiogram- Based on the patient’s stability and susceptibility data, an empiric regimen such as cefepime or piperacillin–tazobactam would be appropriate, as both offer broad Gram-negative coverage and align with local resistance trends. If sepsis were suspected or the patient demonstrated hemodynamic instability, escalation to a carbapenem (e.g., meropenem) could be justified pending culture results.
Once culture and sensitivity data are available, therapy should be de-escalated to the narrowest effective agent and continued for an appropriate duration, typically 7 to 10 days for cUTI or pyelonephritis in older adults. Interprofessional follow-up should include reassessment of clinical response, verification of renal dosing, and education to prevent recurrence.
Which factors place her in the “complicated UTI” category under the 2025 IDSA definitions?
How would you apply the four-step approach (severity, resistance risk, patient factors, antibiogram) to select empiric therapy?
Summary
Urinary tract infections remain among the most common bacterial infections globally, affecting millions annually and driving substantial antibiotic use. The 2025 IDSA guideline updates redefined UTI classification based on clinical presentation and severity rather than patient demographics alone, emphasizing evidence-based diagnosis, shorter treatment durations, and stewardship-focused management. This activity reviews updated definitions, diagnostic principles, pharmacologic and nonpharmacologic strategies, and highlights newly approved antimicrobial agents, including novel agents.
Through interprofessional collaboration, pharmacists, pharmacy technicians, and clinicians can enhance antimicrobial stewardship, tailor therapy using the four-step IDSA approach, and prevent recurrence. Pharmacy technicians play an essential support role in reinforcing adherence, monitoring adverse effects, and promoting appropriate antibiotic use. By integrating these strategies, healthcare teams can improve outcomes, reduce resistance, and advance patient-centered UTI management.
Answers to Active Learning
| Column A – Clinical Statements | Column B – Condition |
| 1. Dysuria, urinary frequency, and urgency without systemic signs | A |
| 2. Flank pain or CVA tenderness with or without cystitis symptoms | B |
| 3. Not essential to obtain urine studies in healthy females with prior cystitis and classic symptoms | A |
| 4. Leukocyte casts suggest possible upper-tract involvement | B |
| 5. Strongly recommended to obtain a urine culture to guide therapy | B |
| 6. May present with fever, chills, rigors, hypotension, or signs of sepsis | B |
References
Fernandez JM, Coyle EA. Urinary Tract Infections. In: DiPiro JT, Yee GC, Haines ST, Nolin TD, Ellingrod VL, Posey L. eds. DiPiro’s Pharmacotherapy: A Pathophysiologic Approach, 12th Edition. McGraw Hill; 2023.
Kurotschka PK, Gágyor I, Ebell MH. Acute uncomplicated UTIs in adults: Rapid evidence review. Am Fam Physician. 2024;109(2):167-174
Trautner BW, Cortés-Penfield NW, Gupta K, et al. Clinical Practice Guideline by Infectious Diseases Society of America (IDSA): 2025 Guideline on Management and Treatment of Complicated Urinary Tract Infections: Duration of Antibiotics for Complicated UTI. Clin Infect Dis. Published online December 20, 2025. doi:10.1093/cid/ciaf462
Abou Heidar NF, Degheili JA, Yacoubian AA, Khauli RB. Management of urinary tract infection in women: A practical approach for everyday practice. Urol Ann. 2019;11(4):339-346. doi:10.4103/UA.UA_104_19
Anger JT, Bixler BR, Holmes RS, Lee UJ, Santiago-Lastra Y, Selph SS. Updates to Recurrent Uncomplicated Urinary Tract Infections in Women: AUA/CUA/SUFU Guideline. J Urol. 2022;208(3):536-541. doi:10.1097/JU.0000000000002860
Hooton TM, Roberts PL, Cox ME, Stapleton AE. Voided midstream urine culture and acute cystitis in premenopausal women. N Engl J Med. 2013;369(20):1883-1891. doi:10.1056/NEJMoa1302186
Boscia JA, Kobasa WD, Abrutyn E, Levison ME, Kaplan AM, Kaye D. Lack of association between bacteriuria and symptoms in the elderly. Am J Med. 1986;81(6):979-982. doi:10.1016/0002-9343(86)90391-8
Nicolle LE, Gupta K, Bradley SF, et al. Clinical Practice Guideline for the Management of Asymptomatic Bacteriuria: 2019 Update by the Infectious Diseases Society of America. Clin Infect Dis. 2019;68(10):e83-e110. doi:10.1093/cid/ciy1121
Stamm WE, Counts GW, Running KR, Fihn S, Turck M, Holmes KK. Diagnosis of coliform infection in acutely dysuric women. N Engl J Med. 1982;307(8):463-468. doi:10.1056/NEJM198208193070802
PHENAZOPYRIDINE HYDROCHLORIDE- phenazopyridine tablet. Prescribing Information. Amneal Pharmaceuticals LLC. Updated October 30, 2024. Accessed April 3, 2026. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=b424b50f-990b-4018-a815-0311693cabb3
MACROBID- nitrofurantoin monohydrate/macrocrystalline capsule. Prescribing Information. Almatica Pharma LLC. Updated October 14, 2025. Accessed April 3, 2026. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=8f270a9f-12a1-44d4-bc7e-873613555801
SULFAMETHOXAZOLE AND TRIMETHOPRIM DOUBLE STRENGTH- sulfamethoxazole and trimethoprim tablet. Prescribing Information. New Horizon Rx Group, LLC. Updated December 30, 2013. Accessed April 3, 2026. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=804f8363-8ea1-4a57-bbd1-7fd9b6b4d29b
PIVYA- pivmecillinam tablet, coated. Prescribing Information. Alembic Pharmaceuticals Inc. Updated October 10, 2025. Accessed April 3, 2026. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=440a5f17-ab11-0fc8-e063-6394a90a58e9
FOSFOMYCIN TROMETHAMINE granule, for solution. Prescribing Information. Amneal Pharmaceuticals NY LLC. Updated April 2, 2024. Accessed April 3, 2026. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=aacd04a4-f2d5-45a2-b9e7-0e92244a5880
AMOXICILLIN AND CLAVULANATE POTASSIUM- amoxicillin and clavulanate potassium tablet, film coated. Prescribing Information. Micro Labs Limited. Updated July 21, 2025. Accessed April 3, 2026. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=0c2d1fbc-f80c-4737-a15e-9cfa6bf99769
CEFADROXIL capsule. Prescribing Information. Aurobindo Pharma Limited. Updated January 30, 2024. Accessed April 3, 2026. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=7bb90096-14d3-400e-94d3-048bff6b1292
CEFPODOXIME PROXETIL tablet, film coated. Prescribing Information. Amici Pharmaceuticals, LLC. Updated July 13, 2023. Accessed April 3, 2026. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=9d6d7f50-9838-47e3-8214-53e2dbeeadea
CEPHALEXIN capsule. Prescribing Information. AMELLA PHARMA, LLC. Updated October 10, 2025. Accessed April 3, 2026. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=602fae70-3b8a-4f09-8db1-41fef4a2ecd7
CEFDINIR capsule. Prescribing Information. American Health Packaging. Updated June 7, 2024. Accessed April 3, 2026. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=c493e78b-afa3-4f74-8305-75e5f3817df0
CIPROFLOXACIN- ciprofloxacin hydrochloride tablet, film coated. Prescribing Information. Aurobindo Pharma Limited. Updated April 24, 2025. Accessed April 3, 2026. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=6a935846-3c97-4517-bc4e-2377f673285e
CIPROFLOXACIN- ciprofloxacin hydrochloride tablet, film coated, extended release. Prescribing Information. Dr. Reddy's Laboratories Limited. Updated August 30, 2011. Accessed April 3, 2026. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=4ab110e4-7b91-42aa-8052-f24cd79293f0
LEVOFLOXACIN- levofloxacin tablet, film coated. Prescribing Information. Camber Pharmaceuticals, Inc. Updated August 14, 2024. Accessed April 3, 2026. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=b43e9b51-568c-4d84-bbd1-d237120056c4
BLUJEPA- gepotidacin tablet, film coated. Prescribing Information. GlaxoSmithKline LLC. Updated February 10, 2026. Accessed April 3, 2026. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=80b57cfe-7819-4d95-a57d-014af42f118d
CEFEPIME- cefepime hydrochloride injection, powder, for solution. Prescribing Information. Qilu Pharmaceutical Co., Ltd. Updated November 15, 2021. Accessed April 3, 2026. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=80b4c811-4a71-47fb-8565-85ce9d0f782c
EXBLIFEP- cefepime hydrochloride, enmetazobactam injection, powder, forsolution. Prescribing Information. Allecra Therapeutics SAS. Revised 2/2024. Accessed April 3, 2026. https://www.bing.com/ck/a?!&&p=72c6873efbf0a3766a7060643cf06281770cfc96f3d3334f256525faca292118JmltdHM9MTc3NTI2MDgwMA&ptn=3&ver=2&hsh=4&fclid=04b4bed2-ccb5-649d-35b3-a881cd986507&u=a1aHR0cHM6Ly9kYWlseW1lZC5ubG0ubmloLmdvdi9kYWlseW1lZC9kb3dubG9hZHBkZmZpbGUuY2ZtP3NldElkPTJkYTAzMWIxLTA3ZmYtNDhmMy04OGQyLTdlODZiYTkzMDQ4YQ
FETROJA- cefiderocol sulfate tosylate injection, powder, for solution. Prescribing Information. Shionogi Inc. Updated July 1, 2025. Accessed April 3, 2026. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=75c0c785-38e0-4049-a6fb-b77581f5b35c
CEFOTAXIME- cefotaxime injection powder, for solution. Prescribing Information. SteriMax Inc. Updated December 12, 2024. Accessed April 3, 2026. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=8efe3bd0-c43a-2bd6-e053-2a95a90a92fa
CEFTAZIDIME injection, powder, for solution. Prescribing Information. Sagent Pharmaceuticals. Updated January 3, 2024. Accessed April 3, 2026. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=aec64360-8452-4987-a366-a5a4f8762ab6
AVYCAZ- ceftazidime, avibactam powder, for solution. Prescribing Information. Allergan, Inc. Updated April 25, 2025. Accessed April 3, 2026. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=d9c2803f-dc9c-4b19-b4a3-8303bc8c15fd
ZERBAXA- ceftolozane and tazobactam injection, powder, lyophilized, for solution. Prescribing Information. Merck Sharp & Dohme LLC. Updated November 4, 2024. Accessed April 3, 2026. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=70ac1d90-eff3-4f0b-9f46-5846c571b32f
CEFTRIAXONE- ceftriaxone sodium injection, powder, for solution. Prescribing Information. Samson Medical Technologies, LLC. Updated January 15, 2023. Accessed April 3, 2026. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=5cd2d96f-83e5-4326-ae87-d0ede4ba493a
ERTAPENEM- ertapenem sodium injection. Prescribing Information. Apotex Corp. Updated December 13, 2023. Accessed April 3, 2026. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=468105b9-48b3-4477-82c7-e7fb92460e28
CONTEPO- fosfomycin disodium injection, powder, for solution. Meitheal Pharmaceuticals Inc. Updated November 4, 2025. Accessed April 3, 2026. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=855574f2-39f8-43bc-a3d2-79dbb578cb94
IMIPENEM AND CILASTATIN- imipenem and cilastatin sodium injection, powder, for solution. Prescribing Information. Fresenius Kabi USA, LLC. Updated October 6, 2022. Accessed April 3, 2026. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=6ab0ca9e-9a79-4cab-9f09-470eda06952e
RECARBRIO- imipenem anhydrous, cilastatin, and relebactam anhydrous injection, powder, for solution. Prescribing Information. Merck Sharp & Dohme LLC. Updated December 9, 2025. Accessed April 3, 2026. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=be32f468-738b-40df-a425-591e12f65159
MEROPENEM injection. Prescribing Information. ARMAS PHARMACEUTICALS INC. Updated July 29, 2022. Accessed April 3, 2026. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=e1122310-c3ed-42f1-b189-301c221ff1f0
VABOMERE- meropenem-vaborbactam injection, powder, for solution. Prescribing Information. Melinta Therapeutics, LLC. Updated February 27, 2026. Accessed April 3, 2026. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=e237fd0f-3050-42a3-b43e-b6fb4824e93d https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=e237fd0f-3050-42a3-b43e-b6fb4824e93d
PIPERACILLIN AND TAZOBACTAM injection, powder, for solution. Prescribing Information. Apotex Corp. Updated May 21, 2025. Accessed April 3, 2026. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=2c8f06a2-49b2-4169-94ef-182d13d248bf
ZEMDRI (PLAZOMICIN)- plazomicin injection. Prescribing Information. Cipla USA Inc. Updated December 20, 2024. Accessed April 3, 2026. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=eccf9fd9-53ac-4252-a106-5567b3a82a70
Torres A, Wible M, Tawadrous M, et al. Efficacy and safety of ceftazidime/avibactam in patients with infections caused by β-lactamase-producing Gram-negative pathogens: a pooled analysis from the Phase 3 clinical trial programme. J Antimicrob Chemother. 2023;78(11):2672-2682. doi:10.1093/jac/dkad280
Shields RK, Nguyen MH, Chen L, et al. Ceftazidime-Avibactam Is Superior to Other Treatment Regimens against Carbapenem-Resistant Klebsiella pneumoniae Bacteremia. Antimicrob Agents Chemother. 2017;61(8):e00883-17. Published 2017 Jul 25. doi:10.1128/AAC.00883-17
Wagenlehner FM, Gasink LB, McGovern PC, et al. Cefepime-Taniborbactam in Complicated Urinary Tract Infection. N Engl J Med. 2024;390(7):611-622. doi:10.1056/NEJMoa2304748
Peiffer-Smadja N, Allison R, Jones LF, et al. Preventing and Managing Urinary Tract Infections: Enhancing the Role of Community Pharmacists-A Mixed Methods Study. Antibiotics (Basel). 2020;9(9):583. Published 2020 Sep 7. doi:10.3390/antibiotics9090583
Agency for Healthcare Research and Quality. Improving healthcare safety by enhancing care coordination: AHRQ-funded patient safety project highlights. U.S. Department of Health and Human Services. May 2025. Accessed April 2, 2026. https://www.ahrq.gov/sites/default/files/wysiwyg/patient-safety/highlights/ps-project-highlights-care-coordination.pdf
DISCLAIMER
The information provided in this course is general in nature, and it is designed solely to provide participants with continuing education credit(s). This course and materials are not meant to substitute for the independent, professional judgment of any participant regarding that participant’s professional practice, including but not limited to patient assessment, diagnosis, treatment, and/or health management. Medical and pharmacy practices, rules, and laws vary from state to state, and this course does not cover the laws of each state; therefore, participants must consult the laws of their state as they relate to their professional practice.
Healthcare professionals must consult their employer, healthcare facility, hospital, or other organization for guidelines, protocols, and procedures to follow. The information provided in this course does not replace those guidelines, protocols, and procedures, but is for academic purposes only, and this course’s limited purpose is for the completion of continuing education credits.
Participants are advised and acknowledge that information related to medications, their administration, dosing, contraindications, adverse reactions, interactions, warnings, precautions, or accepted uses is constantly changing. Any person taking this course understands that such a person must make an independent review of medication information before any patient assessment, diagnosis, treatment and/or health management. Any discussion of off-label use of any medication, device, or procedure is informational only, and such uses are not endorsed hereby.
Nothing contained in this course represents the opinions, views, judgments, or conclusions of RxCe.com LLC. RxCe.com LLC is not liable or responsible to any person for any inaccuracy, error, or omission with respect to this course or course material.
© RxCe.com LLC 2026: All rights reserved. No reproduction of all or part of any content herein is allowed without the prior, written permission of RxCe.com LLC.
RxCe.com
© RxCe.com LLC 2025: All rights reserved.

