PSORIASIS: PRACTICAL MANAGEMENT CONSIDERATIONS FOR THE HEALTHCARE TEAM
Faculty:
L. Austin Fredrickson, MD, FACP
L. Austin Fredrickson is an Associate Professor of Internal Medicine at Northeast Ohio Medical University, where he serves as core faculty and teaches diagnostics, therapeutics, clinical skills, and health humanities. He is board-certified in general internal medicine and practices rural primary care.
Liz Fredrickson, PharmD, BCPS, is an Associate Professor of Pharmacy Practice and Pharmaceutical Sciences at the Northeast Ohio Medical University (NEOMED) College of Pharmacy. She has eight years of experience in medical writing and research.
Pamela Sardo, PharmD, BS
Pamela Sardo, PharmD, BS, is a freelance medical writer and licensed pharmacist. She is the founder and principal at Sardo Solutions in Texas. Pam received her BS from the University of Connecticut and her PharmD from the University of Rhode Island. Pam’s career spans many years in retail, clinics, hospitals, long-term care, Veterans Affairs, and managed health care responsibilities across a broad range of therapeutic classes and disease states.
Abstract
Psoriasis is a chronic, immune-mediated inflammatory disease with heterogeneous clinical phenotypes and a substantial impact on quality of life. Contemporary management requires accurate assessment of severity and special-site involvement, recognition of common comorbidities, and thoughtful selection of topical, phototherapy, oral systemic, and biologic therapies. This continuing education activity reviews immunologic mechanisms of psoriasis and modern therapeutic targets. Learners will also compare established and emerging treatment classes and review real-world challenges, including variable treatment response, adverse effects, treatment access and cost, and medication switching. Practical, team-based considerations are emphasized, including patient counseling, safety monitoring concepts, adherence barriers, and strategies for coordinating care across outpatient settings.
Accreditation Statements
In support of improving patient care, RxCe.com LLC is jointly accredited by the Accreditation CouncilTM for Continuing Medical Education (ACCME®), the Accreditation Council for Pharmacy Education (ACPE®), and the American Nurses Credentialing Center (ANCC®), to provide continuing education for the healthcare team.

This activity was planned by and for the healthcare team, and learners will receive 2 Interprofessional Continuing Education (IPCE) credits for learning and change.
Credits: 2 contact hour(s) (0.2 CEU(s)) of continuing education credit.
Credit Types:
IPCE Credits - 2 Credits
AAPA Category 1 Credit™️ - 2 Credits
AMA PRA Category 1 Credit™️ - 2 Credits
Type of Activity: Application
Media: Computer-Based Training (i.e., online courses)
Estimated time to complete activity: 2 contact hour(s) (0.2 CEU(s)), including Course Test and course evaluation.
Release Date: April 10, 2026 Expiration Date: April 10, 2029
Target Audience: This educational activity is for Physicians and Physician Assistants
How to Earn Credit: From April 10, 2026, through April 10, 2029, participants must:
Read the “learning objectives” and “author and planning team disclosures;”
Study the section entitled “Educational Activity;” and
Complete the Course Test and Evaluation form. The Course Test will be graded automatically. Following successful completion of the Course Test with a score of 70% or higher, a statement of participation will be made available immediately. (No partial credit will be given.)
CME Credit: Credit for this course will be uploaded to CPE Monitor® for pharmacists. Physicians may receive AMA PRA Category 1 Credit™️and use these credits toward Maintenance of Certification (MOC) requirements. Physician Assistants may earn AAPA Category 1 CME credit, reportable through PA Portfolio. All learners shall verify their individual licensing board’s specific requirements and eligibility criteria.
Statement of Need:
Psoriasis is a chronic, immune-mediated disease with heterogeneous phenotypes and substantial quality-of-life and systemic impacts, yet severity and comorbidities remain underrecognized in routine care. Clinicians may underestimate disease burden when relying solely on body surface area and may fail to systematically evaluate high-impact sites or identify psoriatic arthritis, cardiometabolic risk, and associated mental health issues. Rapidly evolving guidelines and an expanding therapeutic armamentarium create uncertainty regarding regimen selection, risk-benefit considerations, monitoring, vaccination, and perioperative management. Healthcare professionals face increasing responsibility related to complex regimens, navigating access barriers, preserving cold-chain integrity, and counseling on administration, adherence, time to response, and adverse effects across multiple dosage forms. This activity aims to strengthen phenotype recognition, assessment considerations, evidence-based, and team-based use of treatments to optimize safety and long-term outcomes.
Learning Objectives: Upon completion of this educational activity, participants should be able to
Differentiate common psoriasis phenotypes and severity levels
Describe how to assess patients with psoriasis
Compare and contrast therapeutic options by expected benefits, key risks, and practical selection considerations
Disclosures
The following individuals were involved in planning, developing, and/or authoring this activity: L. Austin Fredrickson, MD, FACP; Liz Fredrickson, PharmD, BCPS; and Pamela Sardo, PharmD, BS. None of the individuals involved in developing this activity has a conflict of interest or financial relationships related to the subject matter. There are no financial relationships or commercial or financial support relevant to this activity to report or disclose by RxCe.com or any of the individuals involved in the development of this activity.
© RxCe.com LLC 2026: All rights reserved. No reproduction of all or part of any content herein is allowed without the prior, written permission of RxCe.com LLC.
Educational Activity Pre-Test
Which of the following features is most suggestive of moderate-to-severe psoriasis, regardless of total body surface area (BSA)?
Plaques limited to the elbows
Nail pitting and scalp involvement
Mild pruritus without visible lesions
Single localized plaque on the trunk
Which of the following is the most appropriate next step for a patient with psoriasis who reports morning joint stiffness lasting 45 minutes?
Increase topical corticosteroid potency
Initiate phototherapy
Screen for psoriatic arthritis and consider referral
Reassure the patient that the symptoms are unrelated
Which of the following best describes a key advantage of biologic therapies compared with oral systemic agents in psoriasis treatment?
Lower cost and easier access
No need for baseline screening
Higher rates of near-complete skin clearance (PASI 90)
Daily oral administration
Educational Activity
Psoriasis: Practical Management Considerations for the Healthcare Team
Introduction
Psoriasis is a chronic, immune-mediated inflammatory disease with varying clinical phenotypes. It often has a substantial impact on patients' quality of life.1 This disease has traditionally been viewed as a primary disorder of keratinocytes but is now recognized as a complex "systems disease" driven by the IL-23/Th17 signaling axis.1-5 This systemic inflammation is extensive and leads to a high prevalence of clinically significant comorbidities that require proactive screening and integrated care pathways for patients.1-5
Appropriate management requires a team-based, multifaceted approach, including accurate assessment of disease severity (Body Surface Area and impact on special sites), early recognition of psoriatic arthritis (PsA), and thoughtful selection from an array of rapidly expanding pharmacologic therapies.1-5 Current treatment strategies range from advanced topicals and narrowband ultraviolet B (NB-UVB) phototherapy to oral small molecules and highly targeted biologic therapies.1-5
This continuing education activity reviews the immunologic mechanisms of psoriasis and modern therapeutic targets. Learners will also compare established and emerging treatment classes and review real-world challenges, including variable treatment response, adverse effects, treatment access and cost, and medication switching. Practical, team-based considerations are emphasized, including patient counseling, safety monitoring concepts, adherence barriers, and strategies for coordinating care across outpatient settings.
Guideline Updates
The American Academy of Dermatology/National Psoriasis Foundation (AAD–NPF) guideline series breaks psoriasis treatment into major modalities, including topical, phototherapy, systemic nonbiologic, and biologics.2-5 These guidelines emphasize a personalized approach that matches therapy to disease severity, anatomical site, and patient-specific risk factors rather than a rigid "step-therapy" ladder.2-5
Evolving Definitions of Severity
The traditional severity classification (mild, moderate, severe) of psoriasis previously relied on Body Surface Area (BSA). However, the 2024–2026 consensus updates from the NPF and International Psoriasis Council (IPC) emphasize that severity is multidimensional.6,7
High-Impact Sites: Involvement of the face, scalp, palms, soles, genitals, or nails is now considered the primary criterion for moderate-to-severe disease, regardless of a patient’s total BSA.
Treatment-Based Classification: Disease is categorized as "topical-candidate" versus "systemic-candidate." Patients failing two consecutive 4-week courses of appropriate topicals are now eligible for systemic escalation, reflecting a shift toward preventing "therapeutic inertia.”
Biologics as Standard of Care
Biologics are now considered first-line for systemic-candidate patients. Key clinical takeaways from recent updates include the following:
Vaccination Protocols: A 2024 NPF Delphi consensus specifies that most biologics do not require interruption for administration of non-live vaccines (e.g., flu, COVID-19).8 However, live vaccines (e.g., MMR) require therapy discontinuation for 2–3 half-lives before and 2–4 weeks after administration.8
Perioperative Management: For low-risk surgeries, biologics may often be continued. For moderate-to-high-risk procedures, treatment should typically be paused at the end of a dosing interval and resumed only after wound healing is complete (usually 10–14 days).2
The Role of Oral Systemics
Systemic nonbiologics remain essential because of their cost-effectiveness and utility in combination therapy. The selection of oral agents is increasingly driven by patient comorbidities.3
Apremilast: Often preferred for patients with mild-to-moderate disease who prioritize a favorable safety profile with no required laboratory monitoring.
Deucravacitinib: Best for patients desiring oral therapy with biologic-like efficacy
Exclusion Criteria: Screening for tuberculosis (TB), hepatitis B/C, and HIV remains a mandatory baseline for most systemics, particularly before initiating JAK or TNF inhibitors
Foundation of Care: Topicals and Phototherapy
Topical therapy remains the cornerstone for most patients. Recent guidelines highlight the role of non-steroidal topicals to avoid the long-term risks of corticosteroid-induced skin atrophy.5 For patients seeking non-systemic options, narrowband UVB (NB-UVB) phototherapy continues to be recommended as a highly effective, pregnancy-safe modality.4
Epidemiology and Burden
Psoriasis affects approximately 3.2% of the U.S. adult population, representing over 8 million individuals.1 While the disease can manifest at any age, it follows a bimodal distribution with peaks in early adulthood (ages 20–30) and late adulthood (ages 50–60).1
The Systemic "Psoriatic March"
Psoriasis is no longer viewed in isolation but rather as the cutaneous manifestation of a systemic inflammatory state.
Psoriatic Arthritis (PsA): Approximately 30% of patients with plaque psoriasis will develop PsA.9 Nail involvement (pitting, onycholysis) serves as a critical clinical marker. Patients with nail disease are significantly more likely to have or develop joint involvement.9
Cardiometabolic Risk: Chronic Th17-mediated inflammation contributes to a "psoriatic march" toward vascular inflammation.1 Patients with severe psoriasis have a significantly higher risk of myocardial infarction, stroke, and cardiovascular mortality compared to the general population.10
Mental Health: Depression and anxiety are more common in patients with psoriasis than in many other chronic conditions.1 These psychosocial effects often lead to poor adherence and "therapeutic inertia," where patients stop seeking care because they feel their disease is unmanageable.1
Etiology, Risk Factors, and Pathophysiology
Psoriasis is a chronic, immune-mediated inflammatory disease that arises from the interaction of genetic susceptibility, environmental triggers, and dysregulated immune signaling.11 Strong associations have been identified within the major histocompatibility complex, specifically the PSORS1 locus and HLA-C*06:02 allele.11 However, genetic predisposition alone is not enough to account for disease expression. Environmental factors, including streptococcal infection, skin trauma (Koebner phenomenon), psychological stress, obesity, smoking, alcohol use, and certain medications, can activate innate immune pathways in susceptible individuals.12 These triggers initiate inflammatory signaling cascades that ultimately lead to sustained cutaneous and systemic inflammation.12
At the immunologic level, psoriasis is driven by dendritic cell activation and stimulation of adaptive T-cell responses via the IL-23/Th17 axis.1,2
Initiation: Triggered dendritic cells produce IL-23, IL-12, and TNF-alpha
Amplification: IL-23 acts as the "master regulator," promoting the differentiation and expansion of Th17 cells.
Effector Phase: These T cells secrete IL-17 (A and F), IL-22, and TNF-alpha, which act directly on keratinocytes.
The Feed-Forward Loop: Keratinocytes respond by proliferating rapidly and producing more chemokines, which recruit additional immune cells, creating a self-perpetuating cycle of inflammation.
Circulating pro-inflammatory cytokines contribute to endothelial dysfunction, insulin resistance, and heightened cardiovascular risk, which may help explain the association between psoriasis and cardiometabolic disease.1 Similar immune mechanisms cause psoriatic arthritis, in which IL-17 and TNF-α promote synovial inflammation and bone remodeling abnormalities.1
The risk of developing psoriasis is influenced by genetic susceptibility and environmental triggers. In clinical practice, common exacerbating factors include infection, stress, smoking, obesity/metabolic disease, alcohol overuse, and certain medications.1 Even when triggers are not easily modifiable, identifying them can meaningfully improve long-term control and may reduce the intensity of pharmacotherapy required.1
Clinical Presentation, Diagnosis, and Evaluation
Clinical Presentation
Psoriasis is a heterogeneous disease with several clinical phenotypes, the most common being chronic plaque psoriasis, which accounts for approximately 80–90% of cases.1,13 (See Table 1 below.) Plaque psoriasis (Figure 1) presents as well-demarcated, erythematous plaques with overlying silvery-white scale, most frequently involving the extensor surfaces (elbows, knees), scalp, lumbosacral region, and trunk.1,14 Lesions are typically symmetrical and may be pruritic, painful, or asymptomatic.1 Removal of scale may reveal pinpoint bleeding (Auspitz sign), reflecting underlying vascular dilation.
Figure 1
Plaque psoriasis
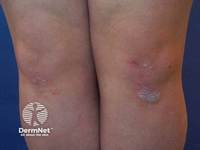
Other important phenotypes include the following.1,14
Guttate psoriasis: Small, drop-like papules often triggered by streptococcal infection; more common in children and young adults
Figure 2
Guttate Psoriasis
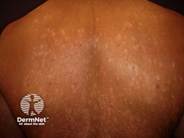
Inverse (intertriginous) psoriasis: Smooth, erythematous plaques in skin folds (axillae, groin, inframammary regions), often lacking prominent scale due to moisture
Pustular psoriasis: Characterized by sterile pustules; may be localized (palmar-plantar) or generalized and potentially life-threatening.
Figure 3
Pustular psoriasis
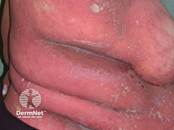
Erythrodermic psoriasis: Diffuse erythema covering most of the body surface area; may be associated with systemic symptoms such as fever and hemodynamic instability
Figure 4
Erythrodermic psoriasis

Nail psoriasis: Pitting, onycholysis, subungual hyperkeratosis, and oil-drop discoloration; nail involvement increases suspicion for psoriatic arthritis
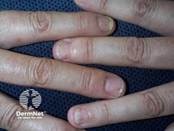
Table 1
Psoriasis Phenotypes and Key Features2
| Phenotype | Typical Appearance | Common Locations | Key Distinguishing Features | Clinical Notes |
| Plaque psoriasis | Well-demarcated erythematous plaques with silvery-white scale | Elbows, knees, scalp, trunk | Symmetrical distribution; Auspitz sign | Most common form (80–90%) |
| Guttate psoriasis | Small, drop-like papules | Trunk, proximal extremities | Often follows streptococcal infection | More common in children or young adults |
| Inverse psoriasis | Smooth, erythematous patches with minimal scale | Axillae, groin, inframammary | Lacks scale due to moisture | High irritation risk; steroid-sparing preferred |
| Pustular psoriasis | Sterile pustules on an erythematous base | Palms/soles or generalized | May be localized or systemic | Severe forms may require urgent care |
| Erythrodermic psoriasis | Diffuse erythema covering most of the body | Generalized | Associated with systemic symptoms | Medical emergency |
Symptoms typically extend beyond visible plaques, with patients commonly reporting pruritus, burning, skin tightness, fissuring, sleep disturbance, and embarrassment.1 Special-site involvement (scalp, palms, soles, face, genitals, nails) can significantly impair daily functioning even when total BSA involvement is limited. Therefore, disease impact cannot be assessed solely by lesion count.1
Diagnosis
Psoriasis is primarily a clinical diagnosis based on characteristic morphology and distribution.1,15 A thorough dermatologic examination is usually sufficient for diagnosis. Key features include sharply demarcated plaques, thick scale, chronic relapsing course, and symmetric distribution.1
Skin biopsy is rarely required but may be considered in atypical cases or when differentiating from conditions such as the following:1
Seborrheic dermatitis
Tinea corporis
Atopic dermatitis
Cutaneous T-cell lymphoma
Lichen planus
Secondary syphilis
If a biopsy is performed, histopathologic findings include acanthosis (epidermal thickening), elongated rete ridges, parakeratosis, diminished granular layer, dilated dermal capillaries, and neutrophilic collections.1,15
Laboratory testing is not necessary to confirm plaque psoriasis but may be used to evaluate comorbidities or guide systemic therapy selection.1,15
Severity Classification
Severity assessment is critical because it guides therapeutic decision-making. While no single metric fully captures disease burden, commonly used measures include the following:2,3,17
Body Surface Area (BSA)
Mild: <3–5%
Moderate: 5–10%
Severe: >10%
The "Palm Method": A patient’s palm (including fingers) represents approximately 1% BSA, providing a quick bedside assessment tool
Physician Global Assessment (PGA)
Psoriasis Area and Severity Index (PASI)
Dermatology Life Quality Index (DLQI) or similar patient-reported outcome measures
Table 2
Diagnostic Tools2,3,17
| Assessment Tool | Severity Classification or Description |
| Physician Global Assessment (PGA) | Clinician-rated assessment of overall plaque characteristics (erythema, induration, scaling); typically scored on a 0–4, 0–5, or 0–6 scale depending on system used |
| Psoriasis Area and Severity Index (PASI) | Composite score incorporating erythema, induration, scaling, and percentage of BSA involvement across body regions; range 0–72 |
| Dermatology Life Quality Index (DLQI) | Patient-reported outcome measure assessing impact on daily activities, work/school, relationships, and emotional well-being; score range 0–30 |
Importantly, patients with limited BSA may still have moderate-to-severe disease if they have any of the following:1,3
Special-site involvement (scalp, nails, palms/soles, genitals, face)
Significant pruritus or pain
Functional impairment
Substantial psychosocial distress
Modern guidelines emphasize integrating both objective disease extent and patient-reported impact when determining treatment intensity.
Evaluation for Comorbidities
Psoriasis is a systemic inflammatory disease, and a comprehensive evaluation should extend beyond the skin.
Psoriatic Arthritis (PsA)
Early identification of psoriatic arthritis is essential to prevent irreversible joint damage. Clinicians should routinely screen for:1
Morning stiffness lasting >30 minutes
Joint swelling or tenderness
Dactylitis (“sausage digits”)
Enthesitis (heel pain, tendon insertion pain)
Nail disease
Validated screening tools such as the PEST (Psoriasis Epidemiology Screening Tool) questionnaire may be used in clinical practice.10 Suspicion warrants rheumatology referral.
Cardiometabolic Risk
Patients with moderate-to-severe psoriasis have an increased prevalence of the following conditions:1
Hypertension
Dyslipidemia
Type 2 diabetes
Obesity
Metabolic syndrome
Care teams should incorporate routine blood pressure measurement, lipid screening, weight assessment, and counseling on lifestyle modification into patient care plans.1
Mental Health
Depression, anxiety, and reduced quality of life are common. Clinicians should assess mood symptoms and consider referral to or integration of behavioral health support when appropriate.1
Infection and Vaccination Review
Before initiating systemic or biologic therapy for psoriasis, a comprehensive baseline evaluation is essential to ensure patient safety and reduce the risk of treatment-related complications.2,3 Because many systemic agents and biologics have immunomodulatory or immunosuppressive effects, screening for latent infections is a critical first step.2,3 Tuberculosis (TB) screening, using either an interferon-gamma release assay (IGRA) or tuberculin skin test, is recommended to identify latent TB infection, particularly before starting tumor necrosis factor (TNF) inhibitors or other biologics associated with reactivation risk. If screening is positive, appropriate treatment for latent TB should be initiated prior to or in coordination with immunosuppressive therapy.2,3
Testing for hepatitis B and hepatitis C infection is also recommended. Baseline hepatitis B surface antigen, surface antibody, and core antibody testing helps identify active or prior infection and guides the need for antiviral prophylaxis or hepatology referral. Hepatitis C screening should also be performed, particularly in patients with risk factors or when initiating agents that are hepatically metabolized or potentially hepatotoxic. HIV testing may be indicated based on patient risk factors, clinical suspicion, or local screening recommendations, as untreated HIV infection may complicate immunosuppressive treatment decisions and infection risk management.
A thorough review of immunization status is another key component of pre-treatment assessment. Patients should be up to date on age-appropriate vaccinations before starting systemic or biologic therapy. Live vaccines are generally contraindicated once immunosuppressive treatment has begun, so necessary live vaccines, such as measles-mumps-rubella (MMR) or varicella, should be administered beforehand when indicated. Inactivated vaccines, including influenza, pneumococcal, COVID-19, and hepatitis B vaccines, should be recommended as appropriate. Proactive evaluation and optimization of infectious risk and immunization status help mitigate preventable complications and support safe, long-term psoriasis management.
Practical Evaluation Framework
A structured evaluation for psoriasis can be summarized as follows and is depicted in Figure 1.
Confirm diagnosis and phenotype
Assess severity (BSA + impact + special sites)
Screen for psoriatic arthritis
Evaluate cardiometabolic and mental health comorbidities
Review medication history and triggers
Assess patient preferences and treatment goals
Determine candidacy for topical, phototherapy, systemic, or biologic therapy
Additional considerations are detailed in Table 3.
Table 3
Special Considerations in Evaluation1-5
| Clinical Consideration | Implications for Psoriasis Management |
| Pregnancy and Reproductive Planning | Medication selection must consider teratogenic risk, safety in pregnancy and lactation, and the need for contraception counseling. |
| Pediatric Patients | May present with higher rates of guttate psoriasis; treatment decisions should account for age-specific dosing, safety data, and long-term growth considerations. |
| Elderly Patients | Polypharmacy, comorbidities, altered pharmacokinetics, and increased risk of infection may influence therapy selection and monitoring. |
| Medication History Review | Assess for drugs known to trigger or exacerbate psoriasis (e.g., beta-blockers, lithium, antimalarials, withdrawal of systemic corticosteroids). |
| Adherence Barriers | Early identification of cost concerns, insurance coverage limitations, injection aversion, and regimen complexity can improve treatment persistence and outcomes. |
Patient Case
J.M. is a 45-year-old patient with 10–12% body surface area involvement from plaque psoriasis affecting the elbows and knees, with significant scalp disease. He reports sleep disruption due to pruritus and embarrassment at work. Medical history includes obesity and elevated blood pressure. He describes intermittent morning finger stiffness and has visible nail pitting.
Severity Assessment
Disease severity is moderate by BSA criteria (10–12%) and is further exacerbated by high-impact scalp involvement, affecting sleep and quality of life.
Comorbidity Screening
Morning stiffness and nail findings raise concern for possible psoriatic arthritis. Screening for inflammatory joint symptoms is warranted, potentially using a validated screening tool, with referral to rheumatology if indicated. Cardiometabolic risk assessment (blood pressure, weight, lipids, diabetes screening) should also be addressed in coordination with primary care.
Initial Management Plan
Optimize topical therapy with scalp-appropriate vehicles (e.g., foam or solution formulations) and a structured regimen for the trunk or extremities.
Provide explicit education regarding duration and maintenance strategies.
Introduce discussion of next-step options if response is inadequate, including phototherapy or systemic therapy
Given a BSA near 10–12% and possible joint involvement, candidacy for systemic therapy should be introduced early. Shared decision-making should compare oral systemic options with biologic therapies, incorporating safety profile, monitoring requirements, and patient preference.
Management of Psoriasis
Psoriasis management requires a structured, longitudinal, and patient-centered approach.1-5 Therapy selection should be individualized based on disease severity and distribution, prior treatment response, reproductive considerations, history of infection and malignancy, organ function, and safety profile, while also accounting for access, cost, and patient preference.1-5 A thorough medication history is essential to identify potential psoriasis-triggering agents or contraindications.
Current AAD-NPF guidelines suggest a "Treat-to-Target" approach. Rather than simply settling for improvement, clinicians should set clear, evidence-based goals with the patient. They may include the following:
Primary Target: Achieving a BSA </1% or a PASI 90/100 response within 3 to 4 months of initiating therapy.2
Maintenance: Sustaining this level of clearance at 6-month intervals.2
Escalation Thresholds: If the target is not met (BSA remains > 3% or PASI improvement is < 75%), the clinician should re-evaluate adherence and triggers, and consider a class switch or dose optimization.1
Ongoing monitoring for safety and therapeutic response is critical, with planned reassessment intervals to guide dose adjustment, escalation, switching, or maintenance strategies.1
Table 4
Treatment Algorithm1-5
| Step | Clinical Focus | Key Actions & Considerations |
| Step 1 | Confirm Phenotype & Assess Severity | • Confirm psoriasis type and rule out mimics • Estimate BSA (mild often <3–5%, but escalate if special-site or QoL impact) • Document PGA and symptom severity (itch, pain, sleep disruption) • Use QoL measure (e.g., DLQI) when feasible • Assess special sites: scalp, nails, palms/soles, face, intertriginous/genital areas |
| Step 2 | Initiate or Optimize Topical Therapy (for mild-to-moderate localized disease or adjunct use) | • Select vehicle and potency based on anatomic site • Educate on duration, fingertip-unit dosing, and maintenance strategies (e.g., weekend therapy) • Add keratolytics or vitamin D analogs if scaling is prominent • Use steroid-sparing agents for sensitive sites • Continue topicals as an adjunct even with systemic therapy |
| Step 3A | Phototherapy | • Consider NB-UVB, excimer, or PUVA (selected cases) • Appropriate for patients preferring non-systemic therapy • Requires adherence to scheduled visits or home phototherapy access |
| Step 3B | Systemic Oral Therapy | • For more extensive disease • For refractory special-site disease • When phototherapy is not feasible |
| Step 3C | Biologic Therapy | • For moderate-to-severe disease • Significant QoL impact • Failure/intolerance of other treatments • Presence of psoriatic arthritis requiring coordinated target |
| Step 4 | Treat-to-Target & Reassess | • Reassess at expected time-to-response — Topicals: weeks — Phototherapy: several weeks — Systemic/biologics: typically ~12–16 weeks • If partial response: optimize adherence, dose, vehicle; consider combination therapy • If inadequate response: switch within/across classes; individualize transition strategy |
Therapies
The management of psoriasis has undergone a paradigm shift, moving away from a rigid, one-size-fits-all "treatment ladder" toward a highly individualized strategy. Therapy selection should be guided by a shared decision-making process that balances clinical efficacy with patient-specific factors, including disease phenotype, anatomical distribution, comorbid conditions, and lifestyle preferences. Psoriasis treatments are available in multiple dosage forms, including topical creams, ointments, foams, and solutions; oral tablets and capsules; and subcutaneous injectable biologics.
In the tables below, topical therapies are listed in Table 5, phototherapy in Table 6, systemic therapies in Table 7, and classification and clinical Profiles of biologic and targeted therapies are covered in Table 8.
Table 5
Topical Therapies2,3
| Class | Examples | Typical Role | Key Counseling or Safety Pearls |
| Topical corticosteroids (by potency) | High/super-potent: clobetasol, halobetasol Medium potency: triamcinolone Low potency: hydrocortisone | First-line for most plaque lesions; rapid symptom relief | Match potency to anatomic site (avoid super-potent agents on face, genitals, or intertriginous areas); limit duration; consider taper or weekend maintenance; monitor for skin atrophy, striae, telangiectasia, and tachyphylaxis |
| Vitamin D analogs | Calcipotriene, calcitriol | Steroid-sparing option; maintenance therapy; often combined with topical corticosteroids | Counsel on consistent application; mild irritation possible; fixed-dose combination products may improve adherence |
| Topical retinoid | Tazarotene | Adjunct for thick, hyperkeratotic plaques | Irritation is common, often combined with corticosteroids; contraindicated in pregnancy |
| Calcineurin inhibitors | Tacrolimus, pimecrolimus (commonly used off-label for psoriasis) | Steroid-sparing therapy for face, intertriginous, and genital psoriasis | Transient burning or stinging is common; apply a thin layer, which is useful for long-term maintenance in sensitive areas |
| Keratolytics and adjunctive therapies | Salicylic acid, coal tar, and emollients | Reduce scale, enhance penetration of other topicals, and provide symptomatic relief | Avoid large-area salicylic acid use in children; coal tar may stain and has an odor; consistent moisturization supports barrier function and adherence |
Table 6
Phototherapy4
| Modality | Typical Candidates | Practical Notes | Key Risks or Monitoring |
| Narrowband UVB (NB-UVB) | Widespread plaque psoriasis; patients preferring non-systemic therapy | Requires reliable attendance or home phototherapy protocol; often combined with topical therapy | Erythema, burning, photoaging; long-term skin cancer risk appears lower than PUVA, but counsel on UV safety |
| Excimer laser/light | Localized plaques; scalp or limited involvement | Targeted therapy reduces overall UV exposure | Local erythema or burning; availability may vary |
| PUVA (psoralen + UVA) | Selected refractory cases | More intensive regimen; less commonly used now | Nausea (psoralen), phototoxicity; higher long-term skin cancer risk compared with NB-UVB; use judiciously |
Table 7
Systemic Therapies1-3,5
| Agent | Mechanism / Role | Typical Dosing Concepts | Key Risks & Monitoring |
| Methotrexate | Immunomodulatory; long-standing systemic option | Weekly dosing (commonly 15–20 mg once weekly) with folic acid supplementation | Hepatic, hematologic, and pulmonary toxicity; monitor CBC, LFTs, renal function; assess hepatitis risk; contraindicated in pregnancy |
| Cyclosporine | Rapid onset immunosuppressive agent; useful for severe flares or erythrodermic psoriasis | Typically used short-term | Nephrotoxicity, hypertension, infection risk; monitor blood pressure and renal function; assess drug interactions |
| Acitretin | Oral retinoid affecting keratinocyte differentiation | Often slower onset; may combine with phototherapy | Highly teratogenic; mucocutaneous adverse effects; hyperlipidemia; monitor lipids and liver enzymes |
| Apremilast | PDE-4 inhibitor; oral option with moderate efficacy | Titration schedule to reduce GI effects; maintenance typically 30 mg twice daily; adjust in severe renal impairment | GI upset, weight loss, mood changes; dose adjustment if CrCl <30 mL/min |
| Other / less common agents | Additional agents discussed in guidelines (e.g., tofacitinib in selected contexts; fumarates outside the US) | Varies by agent | Use specialist guidance; safety monitoring individualized |
Table 8
Classification and Clinical Profiles of Biologic and Targeted Therapies1,2
| Class | Examples (Adult Plaque Psoriasis) | Strengths or Best Fit | Key Safety Considerations |
| TNF-α inhibitors | Adalimumab, etanercept, infliximab, certolizumab | Extensive long-term data; useful when psoriatic arthritis is present | Screen for TB and hepatitis; infection risk counseling; injection reactions; consider malignancy or heart failure history |
| IL-12/23 (p40 inhibitor) | Ustekinumab | Convenient dosing schedule; durable efficacy | Infection risk counseling; injection reactions |
| IL-17 pathway inhibitors | Secukinumab, ixekizumab, brodalumab | High skin efficacy; rapid onset for many patients | Increased risk of mucocutaneous candidiasis; caution in inflammatory bowel disease; brodalumab carries a boxed warning for suicidal ideation |
| IL-23 (p19 inhibitors) | Guselkumab, tildrakizumab, risankizumab | Strong PASI90 response rates; convenient dosing intervals | Generally favorable safety profile; upper respiratory infections and injection-site reactions are the most common |
Translating Clinical Trial Endpoints into Patient Goals
When discussing treatment efficacy in a clinical setting, translating abstract trial endpoints into meaningful, understandable goals is essential for shared decision-making. Psoriasis trials commonly utilize the Psoriasis Area and Severity Index (PASI) to report outcomes such as PASI 75, PASI 90, and PASI 100, which represent 75%, 90%, and 100% improvement in skin clearance, respectively.1 While formal PASI scoring is not routinely calculated in outpatient practice, these benchmarks serve as critical standardized measures for comparing therapies and setting realistic expectations.1 Trials also frequently report the Investigator Global Assessment (IGA) 0/1, which indicates "clear" or "almost clear" skin.1 These validated measures anchor therapeutic discussions, helping to align patient expectations with evidence-based outcomes.1
Comparative Efficacy of Oral and Biologic Therapies
Current evidence indicates that oral nonbiologic options generally demonstrate lower PASI 75 response rates than biologic agents.1 For example, at week 16, approximately 36% of patients treated with methotrexate and 33% of those treated with apremilast achieved PASI 75, compared to placebo rates of 19% and 5%, respectively.1 These data are clinically useful when counseling patients who prefer oral administration or face barriers to injectable therapy. While oral agents remain reasonable, evidence-based options for moderate disease, patients with high disease burden or ambitious clearance goals often require biologic therapy to achieve near-clearance targets.1,3
Modern biologic therapies have fundamentally raised clinical expectations, shifting the primary therapeutic target from PASI 75 to PASI 90 (near-clearance).1 Newer agents, such as the IL-23 inhibitor risankizumab, achieved a PASI 90 of approximately 75% and an IGA 0/1 of 86% at week 16.1 While direct cross-trial comparisons are limited by variations in study design, the practical takeaway is that IL-17 and IL-23 pathway inhibitors offer a high probability of substantial, durable skin clearance with infrequent maintenance dosing. This reinforces a "treat-to-target" mindset where near-total clearance is no longer an exceptional outcome but an increasingly attainable clinical standard.1,2
Care Team Perspective: Oral vs Biologic Therapy
From a medication management standpoint, the health care team plays a key role in differentiating oral systemic and biologic therapies based on the following:
Monitoring Burden
Oral systemics (e.g., methotrexate, cyclosporine): require routine laboratory monitoring (CBC, LFTs, renal function)
Biologics: require baseline infection screening but generally less ongoing lab monitoring
Cost and Access
Oral agents: typically lower cost, fewer access barriers
Biologics: high cost, often require prior authorization and specialty pharmacy coordination
Adherence Considerations
Oral therapies: daily or weekly dosing may reduce adherence
Biologics: less frequent dosing (every 2–12 weeks) may improve adherence but require injection training; generally require less routine laboratory monitoring after baseline screening compared to oral systemic therapies
Safety and Risk Communication
Across biologic classes, the most frequently reported adverse effects include injection-site reactions and upper respiratory tract infections.1 Importantly, large-scale registry data and pooled analyses have not identified a consistent signal for increased internal malignancy or serious infection compared to background population risks.1 In clinical practice, safety is maintained through established standards of care, including mandatory screening for latent tuberculosis and hepatitis prior to initiation, and counseling patients to temporarily hold therapy during serious active infections.2
Patient Counseling and Adherence Support
Psoriasis treatment failure most often results from treatment friction rather than pharmacologic inefficacy. Common barriers include inconvenient topical vehicles, unrealistic application schedules, delayed perceived benefit, adverse effects, injection anxiety, laboratory monitoring burden, insurance barriers, and refill gaps. A structured, team-based approach can substantially improve persistence and outcomes.
Table 9
Key Questions for the Healthcare Team
| Domain | Key Questions | Clinical Implication |
| Prior treatments | What therapies have been used? What worked or failed? | Guides escalation and avoids repetition of ineffective therapy |
| Adherence | Were medications used as prescribed? Any barriers? | Identifies “treatment friction” vs true treatment failure |
| Tolerability | Any adverse effects or intolerance? | May require an alternative class or formulation |
| Access and cost | Insurance coverage? Affordability concerns? | Influences therapy selection (oral vs biologic vs topical) |
Normalize Chronicity and Set Milestones
Patients benefit from understanding that psoriasis is a chronic inflammatory condition requiring chronic management.1 Setting clear expectations, including reassessment at 8 to 16 weeks depending on therapy, supports a treat-to-target strategy.1,2 For example, explaining that near-clearance may take several months prevents premature discontinuation and reduces frustration.
Simplify the Topical Plan
Topical regimens should be kept as simple as possible. A practical strategy includes one clearly defined “weekday plan” and one “weekend or maintenance plan,” with site-based potency assignments and specific stop rules.4 Visual education tools, such as fingertip unit diagrams, improve dosing accuracy and reduce underuse.4 When patients understand exactly how much to apply and for how long, adherence improves, and safety concerns decrease.
Injection Technique Coaching for Biologic Therapy
For patients starting biologic therapy, pharmacists and nurses play a crucial role in education. Instruction should include proper storage, allowing the medication to reach room temperature before injection, site rotation, sharps disposal, and identification of situations requiring temporary discontinuation (e.g., febrile illness or serious infection).3 Patients should be instructed to report signs of infection, including fever, while receiving systemic or biologic therapy. Addressing anxiety related to injections can proactively improve confidence and long-term adherence.
Oral Systemic Monitoring Literacy
Patients prescribed oral systemic therapies will benefit from understanding why laboratory monitoring is required. Education should clarify the purpose of CBC, liver function tests, renal monitoring, and blood pressure checks, as well as which symptoms warrant urgent communication (including unexplained shortness of breath with methotrexate, severe headaches, or blood pressure elevation with cyclosporine).3
Managing Expectations
Managing patient expectations regarding the onset of action is a primary counseling goal to prevent premature treatment discontinuation. While patients using topical steroids may feel itch relief within days and notice plaque thinning within 2 weeks, systemic therapies require more patience.3,4 IL-17 inhibitors are known for their rapid onset, with many patients seeing visible improvement by week 2 to 4. Conversely, IL-23 inhibitors and oral systemics (like apremilast or methotrexate) typically have a slower trajectory, with peak clinical efficacy and near-clearance targets usually achieved between weeks 12 and 16.1,2 Counseling patients to stay the course through the first three months is essential for long-term success.
Clinical Reflection What steroid-sparing strategies could reduce long-term risk while maintaining disease control? |
Interprofessional Roles
Effective use of advanced therapies in atopic dermatitis requires coordinated, interprofessional oversight. Physicians and physician assistants are responsible for selecting appropriate systemic agents and screening for key contraindications, such as avoiding TNF inhibitors in patients with advanced heart failure or demyelinating disease. Pharmacists play a critical role in verifying these risks during medication reviews, identifying potential safety concerns, and reinforcing appropriate therapy selection based on comorbidities.
All members of the healthcare team contribute to optimizing topical therapy, which remains foundational across all disease severities. Prescribers select appropriate agents and vehicles based on lesion type and location, while pharmacists reinforce proper use and help tailor recommendations to patient preferences and lifestyle.18 Recognizing that the vehicle significantly impacts effectiveness and patient acceptance, the team can improve outcomes by aligning treatment strategies with both clinical needs and real-world use.
Return to Patient Case
J.M. demonstrates partial improvement but continues to experience functionally limiting scalp disease and persistent morning stiffness. Adherence to topical therapy has been inconsistent due to time burden and irritation.
Clinical Management Pearls1-4
| Pearl | Key Insight | Clinical Application |
| Pearl 1: BSA Alone Can Be Misleading | Limited body surface area does not always indicate mild disease. | Even 2–3% BSA involvement may represent moderate-to-severe disease if special sites (scalp, face, genitals, hands, feet, nails) are affected or if quality of life is significantly impaired. Always assess special-site involvement and patient-reported burden before labeling disease as “mild.” |
| Pearl 2: Nail Disease Is a Clue to Psoriatic Arthritis | Nail findings increase suspicion for joint involvement. | Nail pitting, onycholysis, and oil-drop discoloration should prompt active screening for psoriatic arthritis symptoms, including morning stiffness, joint swelling, dactylitis, and enthesitis. |
| Pearl 3: Itch Matters | Pruritus is common and clinically meaningful. | Many patients experience significant itch that disrupts sleep and daily functioning. Symptom severity should influence escalation decisions and therapeutic targets. |
| Pearl 4: Think Systemic Inflammation | Psoriasis is associated with cardiometabolic disease. | Incorporate blood pressure measurement, weight assessment, and lipid screening into routine care, particularly in moderate-to-severe disease. |
| Pearl 5: If It’s Not Responding, Reconsider the Diagnosis | Treatment resistance warrants diagnostic reassessment. | Evaluate for mimickers such as tinea corporis, seborrheic dermatitis, cutaneous T-cell lymphoma, or medication-induced eruptions when response is atypical. |
| Pearl 6: Avoid Chronic Systemic Corticosteroids | Systemic steroids carry risk in psoriasis. | Chronic systemic corticosteroid use is generally discouraged due to risk of rebound flares and potential precipitation of pustular or erythrodermic psoriasis. |
Summary
Psoriasis is now understood as a multidimensional, systemic inflammatory disease driven by the IL-23/Th17 axis. Treatment requires a shift in management from simple skin-directed care to a comprehensive "treat-to-target" model. Modern guidelines emphasize that severity is not defined by body surface area alone but by the profound impact on "high-impact" sites and the presence of comorbidities like psoriatic arthritis and cardiometabolic disease. The current therapeutic landscape offers a diverse array of medication options, including advanced nonsteroidal topicals, oral small molecules such as TYK2 inhibitors, and highly targeted biologics.
References
Armstrong AW, Read C. Pathophysiology, clinical presentation, and treatment of psoriasis: A review. JAMA. 2020;323(19):1945–1960. doi:10.1001/jama.2020.4006
Menter A, Strober BE, Kaplan DH, et al. Joint AAD–NPF guidelines of care for the management and treatment of psoriasis with biologics. J Am Acad Dermatol. 2019;80(4):1029–1072. doi:10.1016/j.jaad.2018.11.057
Menter A, Gelfand JM, Connor C, et al. Joint AAD–NPF guidelines of care for the management of psoriasis with systemic nonbiologic therapies. J Am Acad Dermatol. 2020;82(6):1445–1486. doi:10.1016/j.jaad.2020.02.044
Elmets CA, Lim HW, Stoff B, et al. Joint AAD–NPF guidelines of care for the management of psoriasis with phototherapy. J Am Acad Dermatol. 2019;81(3):775–804. doi:10.1016/j.jaad.2019.04.042
Menter A, Korman NJ, Elmets CA, et al. Guidelines of care for the management of psoriasis and psoriatic arthritis: Section 3. Guidelines of care for the management and treatment of psoriasis with topical therapies. J Am Acad Dermatol. 2009;60(4):643–659. doi:10.1016/j.jaad.2008.12.032
Blauvelt A, Prussick R, Merola JF, et al. Clarifying psoriasis disease severity: A position statement from the National Psoriasis Foundation Medical Board. J Am Acad Dermatol. Published online December 30, 2025. doi:10.1016/j.jaad.2025.12.087
Strober B, Zhong Y, Sima A, et al. Criteria for Identifying Candidates for Systemic Psoriasis Treatment in the Real World: Application of the International Psoriasis Council Guidelines in Patients in North America. J Psoriasis Psoriatic Arthritis. 2024;10(1):22-27. Published 2024 Nov 21. doi:10.1177/24755303241302070
Chat VS, Ellebrecht CT, Kingston P, et al. Vaccination recommendations for adults receiving biologics and oral therapies for psoriasis and psoriatic arthritis: Delphi consensus from the medical board of the National Psoriasis Foundation. J Am Acad Dermatol. 2024;90(6):1170-1181. doi:10.1016/j.jaad.2023.12.070
Gisondi P, Bellinato F, Maurelli M, et al. Reducing the Risk of Developing Psoriatic Arthritis in Patients with Psoriasis. Psoriasis (Auckl). 2022;12:213-220. Published 2022 Aug 10. doi:10.2147/PTT.S323300
Zwain A, Aldiwani M, Taqi H. The Association Between Psoriasis and Cardiovascular Diseases. Eur Cardiol. 2021;16:e19. Published 2021 May 13. doi:10.15420/ecr.2020.15.R2
Campanati A, Marani A, Martina E, Diotallevi F, Radi G, Offidani A. Psoriasis as an Immune-Mediated and Inflammatory Systemic Disease: From Pathophysiology to Novel Therapeutic Approaches. Biomedicines. 2021;9(11):1511. Published 2021 Oct 21. doi:10.3390/biomedicines9111511
Liu S, He M, Jiang J, et al. Triggers for the onset and recurrence of psoriasis: a review and update. Cell Commun Signal. 2024;22(1):108. Published 2024 Feb 12. doi:10.1186/s12964-023-01381-0
Kimmel GW, Lebwohl M. Psoriasis: Overview and Diagnosis. Evidence-Based Psoriasis. 2018;1-16. Published 2018 Jul 1. doi:10.1007/978-3-319-90107-7_1
DermNet. Psoriasis. dermnetnz.org. Last reviewed: June 2023. Accessed March 22, 2026. https://dermnetnz.org/topics/psoriasis
Garner KK, Hoy KDS, Carpenter AM. Psoriasis: Recognition and Management Strategies. Am Fam Physician. 2023;108(6):562-573.
Bożek A, Reich A. The reliability of three psoriasis assessment tools: Psoriasis area and severity index, body surface area and physician global assessment. Adv Clin Exp Med. 2017;26(5):851-856. doi:10.17219/acem/69804
Strober B, Ryan C, van de Kerkhof P, et al. Recategorization of psoriasis severity: Delphi consensus from the International Psoriasis Council. J Am Acad Dermatol. 2020;82(1):117-122. doi:10.1016/j.jaad.2019.08.026
Aoki KC, Wong S, Duong JQ, Feldman SR. Adherence to Psoriasis Therapies. Dermatol Clin. 2024;42(3):495-506. doi:10.1016/j.det.2024.02.010
DISCLAIMER
The information provided in this course is general in nature, and it is designed solely to provide participants with continuing education credit(s). This course and materials are not meant to substitute for the independent, professional judgment of any participant regarding that participant’s professional practice, including but not limited to patient assessment, diagnosis, treatment, and/or health management. Medical and pharmacy practices, rules, and laws vary from state to state, and this course does not cover the laws of each state; therefore, participants must consult the laws of their state as they relate to their professional practice.
Healthcare professionals must consult their employer, healthcare facility, hospital, or other organization for guidelines, protocols, and procedures to follow. The information provided in this course does not replace those guidelines, protocols, and procedures, but is for academic purposes only, and this course’s limited purpose is for the completion of continuing education credits.
Participants are advised and acknowledge that information related to medications, their administration, dosing, contraindications, adverse reactions, interactions, warnings, precautions, or accepted uses is constantly changing. Any person taking this course understands that such a person must make an independent review of medication information before any patient assessment, diagnosis, treatment and/or health management. Any discussion of off-label use of any medication, device, or procedure is informational only, and such uses are not endorsed hereby.
Nothing contained in this course represents the opinions, views, judgments, or conclusions of RxCe.com LLC. RxCe.com LLC is not liable or responsible to any person for any inaccuracy, error, or omission with respect to this course or course material.
© RxCe.com LLC 2026: All rights reserved. No reproduction of all or part of any content herein is allowed without the prior, written permission of RxCe.com LLC.
RxCe.com
© RxCe.com LLC 2025: All rights reserved.

