VACCINE AND IMMUNIZATION TRAINING
Steven Malen, PharmD, MBA
Dr. Steven Malen graduated with a dual degree: Doctor of Pharmacy (PharmD) and Master of Business Administration (MBA) from the University of Rhode Island. Over his career, he has worked as a clinical pharmacist in the retail, specialty, and compounding sectors.
Topic Overview
Vaccines are an effective life-saving public health intervention. Vaccines have saved roughly 37 million lives between 2000 and 2019 and are projected to save 69 million deaths by 2030. These statistics are even more impressive because much of the benefit has been in children under five. Pharmacy teams have significantly contributed to advancing public health by providing immunizations. Depending on state law, pharmacists and pharmacy technicians may play a greater role in screening patients for vaccine eligibility and administering injectable vaccines. Understanding vaccine administration best practices, contraindications, and which characteristics and populations may result in patients being recommended for one vaccine over another is essential to the safe and effective administration of vaccines.
Accreditation Statement

RxCe.com LLC is accredited by the Accreditation Council for Pharmacy Education (ACPE) as a provider of continuing pharmacy education.
Universal Activity Number (UAN): The ACPE Universal Activity Number assigned to this activity is
Pharmacist 0669-0000-25-002-H06-P
Pharmacy Technician 0669-0000-25-001-H06-T
Credits: 2 contact hour(s) (0.2 CEU(s)) of continuing education credit
Type of Activity: Knowledge
Media: Internet/Home study Fee Information: $6.99
Estimated time to complete activity: 2 contact hour(s) (0.2 CEU(s)), including Course Test and course evaluation
Release Date: January 17, 2025 Expiration Date: January 17, 2028
Target Audience: This educational activity is for pharmacists and pharmacy technicians.
How to Earn Credit: From January 17, 2025, through January 17, 2028 participants must:
Read the “learning objectives” and “author and planning team disclosures;”
Study the section entitled “Educational Activity;” and
Complete the Course Test and Evaluation form. The Course Test will be graded automatically. Following successful completion of the Course Test with a score of 70% or higher, a statement of participation will be made available immediately. (No partial credit will be given.)
Credit for this course will be uploaded to CPE Monitor®.
Learning Objectives: Upon completion of this educational activity, participants should be able to:
Identify vaccine administration clinical activities
Describe receiving, storing, and handling vaccines
Describe administration routes for routinely recommended vaccines
Describe the safe handling and disposal of sharps
Identify strategies and best practices to prevent vaccine administration errors
Identify contraindications for patients receiving vaccines
Identify how to recognize and manage an adverse reaction to vaccination
Describe immunization documentation procedures
Disclosures
The following individuals were involved in developing this activity: Steven Malen, PharmD, MBA, and Pamela Sardo, PharmD, BS. Pamela Sardo and Steven Malen have no conflicts of interest or financial relationships regarding the subject matter. There are no financial relationships or commercial or financial support relevant to this activity to report or disclose by RxCe.com or any of the individuals involved in the development of this activity.
© RxCe.com LLC 2025: All rights reserved. No reproduction of all or part of any content herein is allowed without the prior, written permission of RxCe.com LLC.
Educational Activity
Vaccine and Immunization Training Introduction
Vaccines are one of the most effective, life-saving inventions of all time. The introduction of vaccines dates back over 2,200 years ago. In modern times, vaccines have saved millions of lives. This makes vaccines one of the top public health interventions in history. Understanding vaccine administration best practices, contraindications, and which characteristics and populations may result in patients being recommended for one vaccine over another is essential to the safe and effective administration of vaccines.
The History of Vaccines
The history of events relating to the discovery of vaccines begins far in the past, as seen in Image 1 below.1 Before the development of vaccines, people understood that smallpox survivors were immune to the disease.2 This led to the practice of inoculation or variolation, which was used to expose a person to a virus.2 The earliest example of variolation may have been in China in 200 BC, where the smallpox virus was derived from cowpox scabs, and people were exposed to smallpox to give them immunity to the disease.1
Image 1
A History of Vaccines Timeline1
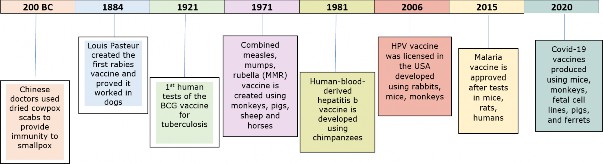
Adapted from: A history of vaccines timeline. Understanding Animal Research. 2024.
In the late 1700s, the smallpox vaccine was introduced.1,2 It was in 1796 that Edward Jenner met a young dairymaid named Sarah Nelms, whose arms and hands had fresh cowpox lesions.2 Jenner used a new procedure he called vaccination on an 8-year-old boy, James Phipps.2 The boy experienced moderate symptoms but then became well. A couple of months later, Jenner vaccinated the boy again, and this time, he showed no symptoms, leading Jenner to conclude that inoculation was successful and complete.2 In 1798, Jenner published his findings. He chose to call this new procedure “vaccination” from the Latin word “vacca,” which means cow, and the derivative word “vaccinia,” meaning cowpox.2 Other vaccines were to follow this momentous introduction of the cowpox vaccine.3 For example, in 1884, Louis Pasteur created the rabies vaccine.1,3
Most vaccines used today were created in the 1900s, with the tuberculosis vaccine leading the way.4 Finally, during the COVID-19 pandemic, vaccine research advanced further with mRNA technology.1 Common vaccines are listed in Table 1 below.5
| Inactive Vaccines | Live Vaccines |
| Influenza (IM) | Typhoid (Oral) |
| Typhoid (IM) | Yellow fever |
| Tdap | Varicella |
| Pneumococcal 23/7 valent | MMR |
| HPV | BCG |
| Hepatitis B | Polio |
| Hepatitis A | Smallpox |
| Hemophilus influenza B | Anthrax |
| Meningococcus | Rotavirus |
| Rabies | Adenovirus type 4, 7 (oral) |
Table 1 Common Vaccines
Types of Vaccines
“Vaccine modalities include live, attenuated viruses, inactivated pathogens, protein subunit vaccines, polysaccharides, conjugate vaccines, DNA platforms, mRNA platforms, and viral vectors.”6 Each of these modalities has its advantages and disadvantages. There are other advantages and disadvantages or challenges of vaccine modalities. Verdecia, et al. (2021), discuss these at greater length.6 There are many advantages to inactivated vaccines. They have a long history and a good safety profile. They are generally useful in administration to vulnerable patients, such as those who are immunosuppressed or pregnant.5-7
As depicted in Table 2 below, immunobiologics can be antigenic substances, such as vaccines or antibody-containing substances from human or animal donors, and are used for active or passive immunization.8 Live vaccines require a spacing of time between the vaccines, whereas inactivated vaccines do not.8,9 Inactivated vaccines may be less immunogenic than their live, attenuated counterparts. They do not replicate, which limits their activation of dendritic cells. “Inactivated vaccines can only activate innate responses at the injection site, making the site and administration route critical.”6
Vaccine Intervals Table 2
Timing and Spacing of Immunobiologics8,9
| Antigen combination | Recommended minimum interval between doses |
| Two or more non-live | May be administered simultaneously or at any interval between doses |
| Non-live and live | May be administered simultaneously or at any interval between doses |
| Two or more live injectable | 28 days minimum interval, if not administered simultaneously |
Effectiveness of Vaccines
Vaccines are one of the most effective life-saving inventions. Researchers have published that in low-income countries, the expansion of vaccinations will have averted 69 million deaths between 2000 and 2030.10 Reductions in mortality are a direct protection and benefit to vaccinated individuals. Indirect protection (herd immunity) within the remaining unvaccinated individuals in a population.10 These statistics are even more dramatic because the publication revealed that over 53% of deaths averted have impacted children under age five.
The effectiveness of vaccines is often questioned because of the variable protection they provide from influenza vaccines. In some years, protection from the influenza vaccines may only be 40 to 60%.11 Most vaccines, however, have efficacy rates over 90% and, in some cases, over 99%.12
There are essential training programs that a pharmacist or pharmacy technician must take before administering vaccinations. In addition, a person administering vaccines must be certified in basic life support, which includes CPR (cardiopulmonary resuscitation) and how to use an automated external defibrillator or AED. Also, pharmacists and pharmacy technicians may be required to take additional training by the applicable pharmacy board or employer.
Identify Vaccine Administration Clinical Activities
Pharmacies will either administer vaccines in a waiting area with other patients or have a dedicated counseling room or room for vaccinations and other clinical services. A dedicated room is preferred as it offers privacy.
Making sure you have all the necessary supplies specific to each available vaccine is vital. Supplies include the syringe, vaccine vial, alcohol wipes, sanitizer, hand soap, medical gloves, sharps containers, literature for patients, and records, among other supplies. Organizing these supplies in a clear and user-friendly layout is essential to streamline the vaccination process
and double-check that the correct needle and vaccine are ready.13 Preparing vaccines requires using the aseptic technique and a good working environment. The vaccine administrator should be seated comfortably to ensure the preparation technique goes well.14
Different vaccines have varying requirements, so clinicians must ensure that all necessary supplies are readily available and appropriately arranged for vials, pre-filled syringes, and vaccines requiring dilution.13 In addition, medical emergency supplies are needed in case an adverse reaction occurs. These supplies may include a blood pressure monitor, telephone, epinephrine, antihistamines, first aid kits, and product-specific instructions, among other supplies.13
Regardless of whether a vial or prefilled syringe is used, a clinician must eliminate air bubbles in the syringe. With a finger flick against the wall of the syringe, bubbles will move to the top of the syringe, and then, by pushing the syringe slowly, the clinician can remove the bubbles with minimal risk of losing the medication dose.15
Some vaccines come in two parts: one is the diluent, and the other is the active ingredient.16,17 The diluent must be removed and added to the active ingredient before injecting. Before vaccinating a patient, review the prescribing information, review instructions for use, and practice the technique.
Table 3 below shows vaccines that come with a diluent.17 Notice that the diluent ingredients vary from vaccine to vaccine and are not interchangeable.
Some vaccines can be reconstituted and drawn up before an immunization appointment. Others must be reconstituted immediately before use. The “time allowed between reconstitution and use” varies substantially.17
Table 3 Vaccines With Diluents17
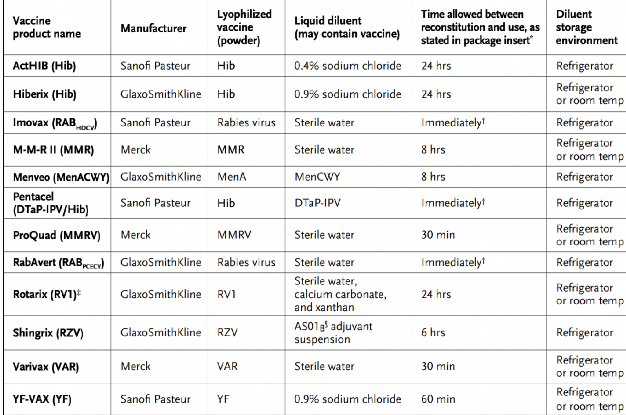
Boards of pharmacy require keeping track of temperatures in which vaccines are stored. Historically, the original temperature tracking method was handwritten logs. Electronic thermometers with automatic logging and an alert system are preferable today. Electronic systems alert clinicians if the temperature is too high or too low and can alert them of a problem when they are not there.
Administration Routes and Sites for Routinely Recommended Vaccines
Some vaccines, such as BCG or rabies, can be dosed intradermally with specialized training.18 In the pharmacy, the vaccines are generally administered by intramuscular or subcutaneous delivery. A subcutaneous
injection is administered at a 45-degree angle into the triceps area of the arm.19,20 An intramuscular injection does not require pinching the skin, is administered at a 90-degree angle, and is injected into the deltoid muscle. It is important not to inject too close to the shoulder to prevent injury.19,20 Always refer to your board-approved immunization training for proper technique and skills reinforcement.
Patients' skin and muscles also have different thicknesses, so a clinician must carefully ensure the depth is appropriate. If the patient is a man over
260 lbs. (118 kg) or a woman over 200 lbs. (90 kg), then use a 1.5-inch needle rather than the standard 1-inch needle for an intramuscular injection.19 The same goes for children who may require a ⅝-inch needle rather than the standard 1-inch needle. Another point worth mentioning is that pharmacists and technicians should try to use the thinnest needle possible, such as a 25-gauge needle. This can help decrease pain for the patient.
Image 2
Intramuscular Administration Site for an Adult20
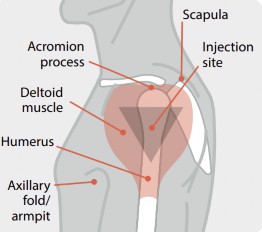
The picture above demonstrates the injection area in an adult receiving an intramuscular injection. Separate injection sites by one inch if multiple vaccines are administered in the same arm.
Immunization Documentation
Pharmacists play a critical role in ensuring accurate vaccine administration and documentation, which is essential for public health and patient safety. Federal law requires pharmacists to document key details, including the vaccine manufacturer, lot number, administration date, and the name and title of the administering provider.21,22 For vaccines covered under the National Childhood Vaccine Injury Act (NCVIA), Vaccine Information Statements (VIS) must also be documented.21-24
Vaccine Information Statements include information about immunization, recommendations for the risks, and what to do if there is a problem.22 Vaccine Information Statements also include the date it was printed and the date provided to the patient or guardian.21,22 Adverse events following vaccination should be reported to the Vaccine Adverse Event Reporting System (VAERS), while administration errors must be reported to the ISMP National Vaccine Errors Reporting Program (VERP).25,26
Immunization Information Systems (IIS) are vital tools for pharmacists, providing a centralized and confidential database to track and consolidate vaccine records.27 These systems ensure accurate immunization histories are accessible to healthcare providers, supporting continuity of care and reducing duplicate vaccinations. Pharmacists must also provide patients with vaccine record cards to ensure the portability of their immunization history, particularly when records are needed outside their primary healthcare network.
It is important to have a completed questionnaire, and the rest of the information will be documented in the computer system, including lot numbers, expiration dates, etc. Ensure the lot number, expiration date, and which arm was injected are all documented on the questionnaire.
Pharmacists must handle undocumented or foreign vaccination histories with care, initiating age-appropriate vaccination schedules when records are unavailable and using serologic testing when appropriate. Written and dated documentation is preferred for all vaccines, though patient-reported histories are acceptable for influenza and pneumococcal polysaccharide vaccines. Pharmacists should also educate patients on the importance of maintaining accurate vaccination records and ensure VISs are provided before every vaccine dose. By following these best practices, pharmacy professionals can uphold high standards of care and enhance public confidence in immunization programs.
Strategies and Best Practices to Prevent Vaccine Administration Errors
Vaccine errors are among the most difficult for a pharmacy and its staff. Table 4 below lists different categories of errors and how to avoid them.28 This table was taken from the Centers for Disease Control and Prevention (CDC) website.28
Table 4 Avoiding Vaccine Errors
| ERROR(S) | POSSIBLE PREVENTIVE ACTION |
| Wrong vaccine, route, site, or dosage (amount); or, improperly prepared. | Circle important information on the packaging to emphasize the difference between the vaccines. Include the brand name with the vaccine abbreviation whenever possible (e.g., PCV13 [Prevnar13]) in orders, medical screens, etc. Separate vaccines into bins or other containers according to type and formulation. Use color- coded identification labels on vaccine storage containers. |
Store look-alike vaccines in different areas of the storage unit (e.g., pediatric and adult formulations of the same vaccine on different shelves in the unit). Do not list vaccines with look- alike names sequentially on computer screens, order forms, or medical records, if possible. Consider using "name alert" or "look-alike" stickers on packaging and in the areas where these vaccines are stored. Consider purchasing products with look-alike packaging from different manufacturers, if possible. Establish "Do NOT Disturb" or no- interruption areas or times when vaccines are being prepared or administered. Prepare vaccines for one patient at a time. Once prepared, label the syringe with the vaccine name. Do not administer vaccines prepared by someone else. Triple-check work before administering a vaccine and ask another staff member to check. Keep reference materials on recommended sites, routes, and needle lengths for each vaccine used in your facility in the medication preparation area. Clearly identify diluents if the manufacturer's label could mislead staff into believing the diluent is the vaccine itself. Integrate vaccine administration training into orientation and other appropriate education requirements. |
Provide education when new products are added to inventory or recommendations are updated. Use standing orders if appropriate. | |
| Wrong patient | Verify the patient's identity before administering vaccines. Educate staff on the importance of avoiding unnecessary distractions or interruptions when staff is administering vaccines. Prepare and administer vaccines to one patient at a time. If more than one patient needs vaccines during the same clinical encounter (e.g., a parent with two children), assign different providers to each patient, if possible. Alternatively, bring only one of the patient's vaccines into the treatment area at a time, labeled with the immunization and patient name. |
| Documentation errors | Do not use error-prone abbreviations to document vaccine administration (e.g., use intranasal route [NAS] to document the intranasal route— not IN, which is easily confused with IM). Use ACIP vaccine abbreviations. Change the appearance of look- alike names or generic abbreviations on computer screens, if possible. |
| Improperly stored and/or handled vaccine administered (e.g., expired vaccine given) | Integrate vaccine storage and handling training based on manufacturer guidance and/or requirements. Rotate vaccines so those with the earliest expiration dates are in |
the front of the storage unit. Use these first. Remove expired vaccines/diluents from storage units and areas where viable vaccines are stored. Isolate vaccines exposed to improper temperatures and contact the state or local immunization program and/or the vaccine manufacturer. | |
| Scheduling errors (e.g., vaccine doses in a series administered too soon) | Use standing orders if appropriate. Create procedures to obtain a complete vaccination history using the immunization information system (IIS), previous medical records, and personal vaccination records. Integrate vaccine administration training, including timing and spacing of vaccines, into orientation and other appropriate education requirements. For children-especially infants- schedule immunization visits after the child’s birthday. Post current immunization schedules for children and adults that staff can quickly reference in clinical areas where vaccinations may be prescribed and administered. Post reference sheets for timing and spacing in your medication preparation area. CDC has vaccine catch-up guidance for DTaP, Tdap, Hib, PCV13, and polio vaccines to assist healthcare personnel in interpreting the catch-up schedule for children. |
| Counsel parents and patients on how important it is for them to maintain immunization records. |
The best practices must be followed to prevent errors. Allowing trained pharmacists and technicians to vaccinate has greatly improved the pharmacy profession. It is important to review medication histories, assess the patient for needed immunizations, screen for contraindications, and educate the patient. The CDC provides a Screening Checklist for Contraindications to Vaccines for Adults and a Screening Checklist for Contraindications to Vaccines for Children and Teens.29
A preventable medical error is a failure to follow accepted practice and may include giving the wrong vaccine to a patient. The main tip recommended to help avoid this error is to read the patient’s name and the vaccine name out loud before giving a vaccine to a patient. This is also a good time to ask the patient if they have any last questions about the vaccine. This is equivalent to the checklist surgeons do to ensure all surgery supplies have been removed from a surgery site.
Adverse Reactions to Vaccines
Adverse reactions to vaccines, while rare, can range from mild to severe, necessitating vigilance and preparedness.30,31 Local reactions such as redness or swelling are common and generally resolve without intervention. Systemic reactions, like fever or fatigue, occur less frequently, while allergic reactions, including anaphylaxis, are rare but require immediate attention.30,31 To prevent adverse outcomes, pharmacists should obtain a thorough allergy history before vaccination, identify potential risk factors, and communicate effectively with patients about the benefits and risks of immunization in culturally sensitive and clear language. Proper observation of patients for 15 minutes post-vaccination, particularly adolescents and adults, helps prevent and manage immediate reactions like syncope.30,31
In the event of an adverse reaction, pharmacy teams must be proficient in recognizing and managing vaccine-related emergencies, especially anaphylaxis. Rapid administration of intramuscular epinephrine is the cornerstone of treatment for anaphylaxis and should be readily available alongside airway management equipment. Continuous monitoring of vital signs, oxygen levels, and patient positioning (elevating the legs in hypotension) is essential. For respiratory symptoms resistant to epinephrine, adjunct treatments such as albuterol nebulization or glucagon for beta- blocker-treated patients may be necessary. Pharmacists should also be familiar with protocols for specific scenarios, such as managing allergies to vaccine components like egg proteins or latex. Reporting adverse events to the Vaccine Adverse Event Reporting System (VAERS) ensures compliance and contributes to public health data that identifies trends and improves vaccine safety practices. By maintaining these protocols and engaging in effective patient communication, pharmacy professionals can safeguard patient well-being and build trust in immunization programs.
Safe Handling and Disposal of Sharps
In the pharmacy setting, the safe handling and disposal of sharps are critical to preventing needlestick injuries, which pose serious risks to staff due to potential exposure to bloodborne pathogens. Proper handling begins with engineering controls such as sharps with integrated safety features, like retractable needles or needleless systems. A safety syringe has a shield that can be put on the syringe safely to prevent needlesticks.32 A retracting syringe is another option that automatically retracts all the way when it is pushed in.32 The needle retracts while it is inside the patient’s body. This removes an additional step by the person administering the vaccine, reducing needlestick risk.
It is important not to recap needles. Pharmacists and pharmacy technicians should be trained not to recap needles, as this practice is a common cause of injuries. Instead, used sharps should be immediately placed into puncture-resistant sharps disposal containers that are strategically
located to ensure convenient access during procedures. These containers must be labeled, leak-proof, and disposed of in accordance with regulatory guidelines.
Establishing clear protocols for handling spills or overfilled sharps containers is vital to avoid accidental exposure. Pharmacy teams should foster a culture of safety through ongoing education and adherence to OSHA's Bloodborne Pathogens Standard, which emphasizes the importance of work- practice controls and personal protective equipment. Regular training sessions should review proper handling techniques, the use of safety devices, and the correct procedures for reporting and responding to any injuries. A comprehensive approach that combines engineering solutions, administrative policies, and education will minimize risks and ensure a safer working environment for all pharmacy staff.
Current Immunization Resources
The Advisory Committee on Immunization Practices (ACIP) is a part of the CDC and dictates the immunization schedule.33 The protocols that pharmacies have to vaccinate generally go hand in hand with these schedules, so it is important to be familiar with them. The schedules not only tell you who is eligible for a certain vaccine by age but also by a medical condition.33
All 50 states, the District of Columbia, and Puerto Rico allow pharmacists to immunize in some capacity.34 More than 320,000 pharmacists have been trained nationwide to administer vaccines.34 Pharmacists are part of the healthcare team that helps promote community access to immunization through collaboration and communication with other healthcare providers. This collaborative approach is referred to as the “immunization neighborhood,” something the American Pharmacists Association and the National Adult and Influenza Immunization Summit have promoted for years now.34
Some states allow pharmacists to independently administer vaccines without a patient-specific prescription from another healthcare provider, and
others do not.35 These laws are typically based on the type of vaccine and the patient's age.35 The laws governing when a pharmacist may administer a vaccine without a physician’s prescription vary by state.35
If a patient has a side effect from a vaccine, it is important to report the event to VAERS via their website: www.vaers.hhs.gov.25 The patient, pharmacy, or physician can report it. Finally, if a patient suffers severe harm from a vaccine, they can apply for injury compensation through HRSA: www.hrsa.gov. This government fund is in place because vaccine manufacturers often have immunity and cannot be sued for injuries caused by their vaccine products.
Assess for Appropriateness and Need
Assessing a patient for vaccine appropriateness could include age, medical condition(s), and reviewing possible contraindications.29 For example, the MMR vaccine prescribing information shows five contraindications.36 These include hypersensitivity to any component of the vaccine, immunosuppression, moderate or severe febrile illness, active untreated tuberculosis and pregnancy.36 Vaccines may be medical condition dependent. Patients may not be appropriate to receive a vaccine if they say they are pregnant. Pregnancy, as with MMR, is a contraindication with many vaccines. Take time to reread the forms the patient completed before vaccination, and double-check that a vaccine is appropriate for the patient. Always review the specific vaccine FDA-prescribing information to double-check the warnings, precautions, contraindications, and dosing for age and weight.
During product shortages, contact the prescriber and review the prescribing information. Thoroughly counsel patients if an alternative is available.
COVID-19 and Other New Vaccine Information
COVID-19 vaccines are novel not only in their ingredients but also in their expiration dates. Most vaccines will have their expiration date on the vial
like all medications; however, because of storage requirements and the Emergency Use Authorization by the FDA, the expiration dates were not printed on the vaccines.37 Moderna vaccine expiration dates can be found on their website.37
In 2024, ACIP recommended a second dose of 2024-2025 COVID-19 for adults ages 65 years and older.38 If previously unvaccinated and receiving Novavax, two doses are recommended as an initial vaccination series followed by a third dose of any age-appropriate 2024-2025 COVID-19 vaccine 6 months (minimum interval 2 months) after the second dose.38
ACIP also recommended a second dose of the 2024-2025 COVID-19 vaccine for people ages 6 months to 64 years who are moderately or severely immunocompromised.38 If previously unvaccinated or receiving an initial vaccination series, at least two doses of the 2024-2025 vaccine are recommended, and depending on vaccination history, more may be needed. This additional 2024-2025 vaccine dose is recommended 6 months (minimum interval 2 months) after completing the initial vaccination series. ACIP also recommended additional doses (i.e., three or more doses) of the 2024-2025 COVID-19 vaccine for people ages 6 months and older who are moderately or severely immunocompromised with shared decision-making.38
A difference between routine, catch-up, and risk-based recommendations and shared clinical decision-making recommendations is the decision to vaccinate.39 For routine, catch-up, and risk-based recommendations, the decision should be to vaccinate the patient based on age group or indication unless contraindicated. For shared clinical decision- making recommendations, the decision about whether or not to vaccinate may be informed by who may benefit, the individual’s characteristics and preferences, the health care provider’s clinical discretion, and the characteristics of the vaccine.39
The Pfizer-BioNTech COVID-19 vaccine products come in vials with different cap colors to assist pharmacists and technicians in identifying the appropriate Pfizer-BioNTech COVID-19 vaccine for each age group.40 Table 5, below, provides that a maroon cap indicates the vaccine is appropriate for 5
months through 4 years of age, an orange cap indicates the vaccine is appropriate for 5 through 11 years of age, and a gray or purple cap indicates the vaccine is appropriate for 12 years of age and older.40 Moreover, the CDC’s Pfizer-BioNTech COVID-19 Vaccine Products At-A-Glance provides an excellent reference of other important information for pharmacists and technicians who are administering Pfizer-BioNTech COVID-19 vaccines.41
Table 5
COVID-19 Vaccine Products At-A-Glance
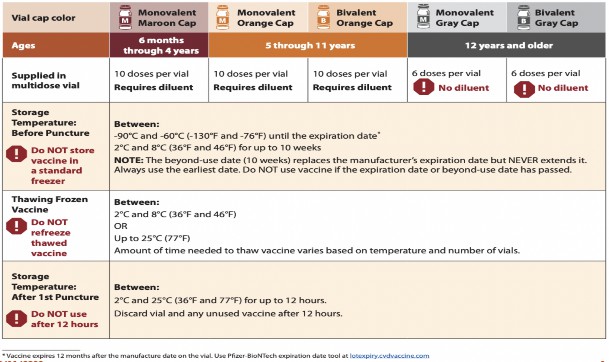
The expiration date for Pfizer vaccines is complicated as it depends on the vaccine sequence being in transit, in the ultra-cold freezer, in the regular freezer, in thawing, and finally in regular storage. For more details, look at the package insert for the Pfizer COVID-19 vaccine being given.42-45
Table 6 below shows the differences between COVID-19 vaccines. It is also important to remember that the age requirements and other criteria are
ever-changing with these vaccines. The schedule for COVID-19 vaccines is relatively complicated as they have multiple boosters.46
Table 6
COVID- 19 Vaccines
| Moderna (2024-2025) | Novavax | Pfizer-BioNTech | |
| FDA approval | no, authorized for emergency use by FDA under an Emergency Use Authorization (EUA) | no, authorized for emergency use by FDA under an Emergency Use Authorization (EUA) | yes, and Emergency Use Authorization (EUA) |
| Age | COVID-19 vaccine under EUA for children 6 months to 11 years Spikevax is FDA- approved for ages 12+ | Ages 12+ | COVID-19 vaccine EUA for children 6 months to 11 years Comirnaty is FDA- approved for ages 12+ |
| Type of Vaccine | mRNA | Protein subunit | mRNA |
| Storage | ultracold/frozen | refrigerator between 2 to 8 °C (36 to 46 °F) Do not freeze. | ultracold/frozen |
| After first puncture | Discard after single use | Discard after single use | Discard after single use |
| Preparation | Do not dilute Do not refreeze | Do not dilute Do not shake | Do not dilute Do not refreeze |
Respiratory Syncytial Virus (RSV)
The 2023–2024 RSV season was the first during which RSV vaccination was recommended for U.S. adults aged ≥60 years, using shared clinical decision-making.47 A single dose of any Food and Drug Administration-approved RSV vaccine (Arexvy, Abrysvo, or mResvia) is now recommended for all adults aged ≥75 years and those aged 60–74 years at increased risk for severe RSV disease. Eligibility for RSV vaccination is based on clinical assessment and patient attestation of a risk factor. The CDC recommends infants be protected by maternal vaccination or infant receipt of nirsevimab. Beware of medication errors due to the difference between the RSV vaccine and the RSV monoclonal antibody.
Pneumococcal Vaccines
In 2024, ACIP recommended a pneumococcal conjugate vaccine (PCV) for all PCV-naïve adults aged ≥50 years.48 Adults aged 19 to 64 with risk conditions are recommended to receive either 15 or 20 valent pneumococcal conjugate vaccine (PCV).48 ACIP recommended PCV 21 as an option for adults aged ≥19 years who are currently recommended to receive PCV 15 or PCV 20. It contains eight serotypes not included in other vaccines.48
Meningococcal Vaccines
In 2024, ACIP recommended MenB-4C (Bexsero®) be administered as a 2-dose series at 0 and 6 months when given to healthy adolescents and young adults aged 16–23 years based on shared clinical decision-making for the prevention of serogroup B meningococcal disease.48
ACIP recommends MenB-4C (Bexsero®) be administered as a 3-dose series at 0, 1–2, and 6 months when given to persons aged ≥10 years at increased risk for serogroup B meningococcal disease (i.e., persons with anatomic or functional asplenia, complement component deficiencies, or complement inhibitor use; microbiologists routinely exposed to N. meningitidis isolates; and persons at increased risk during an outbreak).48
The Role of the Pharmacy Technician in Vaccination
Pharmacy technicians can play a vital role in supporting vaccination services, helping to address the growing demand for immunizations.49 Depending on state law, pharmacy technicians may screen patients for vaccine eligibility and administer injectable vaccines.49 By taking on these tasks, pharmacy technicians may improve workflow efficiency, free up pharmacists for clinical responsibilities, and enhance patient outcomes.49
The use of pharmacy technicians to administer vaccines is highly regulated. State-specific, accredited vaccine administration training for technicians and oversight by a pharmacist strengthens this capacity, making vaccination services safer and more accessible to patients.49
By embracing professional practice advancements and leveraging the benefits of technology, pharmacies can expand their vaccination services efficiently and reach a broader patient population.49
Vaccine Billing
Billing vaccines can be accomplished through a pharmacy benefit plan, medical benefit plan, or both. Coverage can differ depending on the billing used and requires extensive learning and experience. The processes can also be very different from pharmacy to pharmacy.50
Transactions covered under the pharmacy benefit are processed directly between the pharmacy and the pharmacy benefits manager (PBM).50 These claims are adjudicated in real-time, with immediate feedback on payment amounts based on contracted rates.50
While representing a smaller percentage of transactions, medical coverage involves high-value claims such as specialty drugs, clinical services, durable medical equipment (DME), and certain vaccines. Unlike pharmacy benefit claims, medical benefit claims are processed directly between the pharmacy and the payer. These claims are submitted in batches using the American National Standards Institute X12 837 standard or a CMS 1500 form. Due to the delayed batch processing, denials may occur weeks after submission, necessitating proactive use of claim adjudication technology to minimize risks.50
When considering vaccine coverage, it depends on the payer and the type of vaccine. Pharmacies must navigate these variations to ensure proper reimbursement.50
Hypothetical Patient Case
A 55-year-old diabetic male enters a pharmacy in October to pick up prescriptions. What vaccines is he eligible for? He has only received a flu shot since childhood. The goal is to see which vaccines this patient is eligible to receive.22 This information may be found on the CDC’s website under Immunization Schedules made available to healthcare providers and in the Additional Resources table below.22
The CDC provides guidance for patients with underlying medical conditions that may be associated with a higher risk for severe COVID-19.23 Since it is October, it is appropriate to offer the flu shot as it is needed once a year. The next vaccine you can recommend is the Td or Tdap, which requires a booster every 10 years. The MMR vaccine is also recommended as the patient was born after 1957; however, you must double-check if they received it. Of note, before vaccines were available, nearly everyone was infected with
measles, mumps, and rubella viruses during childhood, which is presumed in those born before 1957.51 There are state vaccine registries linked to pharmacies and doctors, so you can offer to double-check the registry for the patient. For example, Florida has an IIS program called FL SHOTS.52,53 A Florida patient’s immunization records may be accessed by a clinician at https://flshotsusers.com/. The FL SHOTS program is authorized under Florida law.54
The next eligible vaccine is the zoster or shingles vaccine since the patient is over 50. The PPSV23 is Pneumovax or one of the pneumonia vaccines and is recommended because of the patient’s diabetes. Finally, the patient may be eligible for their COVID-19 booster based on the schedule.
Additional Resources
| Immunization Schedule by Age | Resource (Table) |
| Child and Adolescent Immunization Schedule by Age | https://www.cdc.gov/vaccines/hcp/i mz-schedules/child-adolescent- age.html#table-1 |
| Vaccines in the Adult Immunization Schedule | https://www.cdc.gov/vaccines/hcp/i mz-schedules/adult-age.html |
Summary
Before the pandemic, vaccinations were difficult to prepare, administer, and bill. Since the pandemic, these difficulties have only increased. This course reviews a brief history of vaccines, describes common vaccines, shares select ACIP 2024 updates, and recommends error prevention methods to advance best practices around vaccine administration. The risk of contraindications must be monitored for the safe and effective administration of vaccines.
It is important to stay current on COVID-19 vaccinations since they are continually changing. When discussing immunization opportunities and status with patients, reinforce that vaccinations can reduce the risk of serious diseases and improve patients' health outcomes. These discussions can also build better relationships with patients.
Course Test
Any two non-live vaccines can be given
at the same time.
one week apart.
two weeks apart.
three weeks apart.
Keeping a log to track temperatures in which vaccines are stored
is NOT a requirement of the board of pharmacy in every state, so technicians need to check their local rules.
only affects medication efficacy; it has nothing to do with medication safety.
is a requirement of the board of pharmacy in every state.
can only be done using electronic thermometers.
True or False: All vaccines require the same supplies.
True
False
vaccines have the lowest risk of a needlestick injury.
Single-dose vial
Prefilled syringe
Reconstituted
Multi-dose vial
Intramuscular vaccines are given in the
triceps area.
gluteal area.
deltoid area.
trapezoid area.
Subcutaneous vaccines require pinching the skin and injecting at a -degree angle.
0
45
90
180
What technique is used to monitor patient safety after vaccine administration?
Proper observation of patients should occur for 15 minutes on site post-vaccination
Immediately send the patient away, but have them call you if they get short of breath
Send the immunization record to the patient’s physician since monitoring is their responsibility
All patients must be monitored for an hour in the pharmacy post- vaccination
To avoid vaccine errors in children-especially infants-
only schedule immunization visits after the child’s fifth birthday.
never pinch the skin.
schedule immunization visits after the child’s birthday.
only use retracting syringes.
Which of the following options is NOT a method to avoid vaccine errors?
Circle important information on the vaccine package to highlight differences between vaccines
Implement vaccine administration training as part of the new employee orientation process
Do not rotate vaccines. Keep the ones with the earliest expiration at the back of the shelf
Avoid using error-prone abbreviations when documenting vaccine administration
Which of the options below include contraindications to the MMR vaccine?
ear infections, rashes
pregnancy, immunosuppression
psoriasis, cataracts
arthritis, cold sores
References
History of vaccines timeline. Understanding Animal Research. 2023. https://www.understandinganimalresearch.org.uk/resources/infographic s/history-of-vaccines-timeline. Accessed December 19, 2024.
Riedel S. Edward Jenner and the history of smallpox and vaccination. Proc (Bayl Univ Med Cent). 2005;18(1):21-25. doi:10.1080/08998280.2005.11928028
Plotkin S. History of vaccination. Proc Natl Acad Sci U S A.
2014;111(34):12283-12287. doi:10.1073/pnas.1400472111
Luca S, Mihaescu T. History of BCG Vaccine. Maedica (Bucur). 2013;8(1):53-58.
Tanrıöver MD, Akar S, Türkçapar N, Karadağ Ö, Ertenli İ, Kiraz S. Vaccination recommendations for adult patients with rheumatic diseases. Eur J Rheumatol. 2016;3(1):29-35. doi:10.5152/eurjrheum.2016.16100
Verdecia M, Kokai-Kun JF, Kibbey M, Acharya S, Venema J, Atouf F. COVID-19 vaccine platforms: Delivering on a promise?. Hum Vaccin Immunother. 2021;17(9):2873-2893. doi:10.1080/21645515.2021.1911204
Arvas A. Vaccination in patients with immunosuppression. Turk Pediatri Ars. 2014;49(3):181-185. Published 2014 Sep 1. doi:10.5152/tpa.2014.2206
Kroger AT, Atkinson WL, Marcuse EK, Pickering LK; Advisory Committee on Immunization Practices (ACIP) Centers for Disease Control and Prevention (CDC). General recommendations on immunization: recommendations of the Advisory Committee on Immunization Practices (ACIP) [published correction appears in MMWR Morb Mortal Wkly Rep. 2006 Dec 8;55(48):1303] [published correction appears in MMWR Morb Mortal Wkly Rep. 2007 Mar 23;56(11):256] [published correction appears in Pediatrics. 2007 May;119(5):1008]. MMWR Recomm Rep. 2006;55(RR-15):1-48.
Centers for Disease Control and Prevention. Timing and Spacing of Immunobiologics. July 24, 2024. CDC.https://www.cdc.gov/vaccines/hcp/imz-best-practices/timing- spacing- immunobiologics.html#:~:text=There%20is%20no%20evidence%20th at,(Table%203%2D4). Accessed December 19, 2024.
Li X, Mukandavire C, Cucunubá ZM, et al. Estimating the health impact of vaccination against ten pathogens in 98 low-income and middle- income countries from 2000 to 2030: a modelling study [published correction appears in Lancet. 2021;397(10275):670]. Lancet. 2021;397(10272):398-408. doi:10.1016/S0140-6736(20)32657-X
Centers for Disease Control and Prevention. Flu Season Has Started but there is Still Time to Benefit from Getting a Flu Vaccine. Influenza. CDC. December 20, 2024. https://www.cdc.gov/flu/whats-new/2024-2025-
flu-activity-vaccine.html. Accessed December 21, 2024.
Orenstein WA, Ahmed R. Simply put: Vaccination saves lives. Proc Natl Acad Sci U S A. 2017;114(16):4031-4033. doi:10.1073/pnas.1704507114
Centers for Disease Control and Prevention. Satellite, Temporary and Off-site Vaccination Clinic Supply Checklist. CDC. August 11, 2020. https://www.cdc.gov/vaccines/hcp/admin/mass-clinic- activities/vaccination-clinic-supply-checklist.html. Accessed December 21, 2024.
Administration for Strategic Preparedness and Response (ASPR) 200.
U.S. Department of Health and Human Services. COVID-19 Vaccine Ancillary Supply and Mixing Kits. HHS. Undated. https://aspr.hhs.gov/SNS/Pages/COVID19-Vaccine-Ancillary- Supply.aspx. Accessed December 19, 2024.
Prueksaanantakal N, Manomaipiboon A, Phankavong P, et al. Effectiveness of the Air-Filled Technique to Reduce the Dead Space in Syringes and Needles during ChAdox1-n CoV Vaccine Administration. Vaccines (Basel). 2023;11(4):741. Published 2023 Mar 27. doi:10.3390/vaccines11040741
Centers for Disease Control and Prevention. Vaccine Storage and Handling Toolkit. CDC. January 2023. https://www.cdc.gov/vaccines/hcp/admin/storage/toolkit/storage- handling-toolkit.pdf. Accessed December 19, 2024.
immunize.org. Vaccines with Diluents: How to Use Them. immunize.org. No Date. https://www.immunize.org/wp- content/uploads/catg.d/p3040.pdf. Accessed December 19, 2024.
Kim YC, Jarrahian C, Zehrung D, Mitragotri S, Prausnitz MR. Delivery systems for intradermal vaccination. Curr Top Microbiol Immunol. 2012;351:77-112. doi: 10.1007/82_2011_123
Centers for Disease Control and Prevention. Vaccine Administration. Vaccines and Immunizations. CDC. June 18, 2024. https://www.cdc.gov/vaccines/hcp/imz-best-practices/vaccine- administration.html#:~:text=For%20women%20who%20weigh%20%3 E200,subcutaneous%20tissues%20are%20not%20bunched. Accessed December 21, 2024.
Centers for Disease Control and Prevention. You Call the Shots. Vaccine Administration: Intramuscular (IM) Injection Adults 19 Years of Age or Older. CDC. November 16, 2020. https://www.cdc.gov/vaccines/hcp/admin/downloads/IM-Injection- adult.pdf. Accessed December 19, 2024.
Centers for Disease Control and Prevention. Documenting Vaccinations. Document the Vaccination(s). CDC. May 7, 2019. https://www.cdc.gov/vaccines/hcp/admin/document-vaccines.html. Accessed December 19, 2024.
Centers for Disease Control and Prevention. Vaccines & Immunizations. Vaccination Records. CDC. July 17, 2024. https://www.cdc.gov/vaccines/hcp/imz-best-practices/vaccination- records.html?CDC_AAref_Val=https://www.cdc.gov/vaccines/hcp/acip- recs/general-recs/records.html. Accessed December 21, 2024.
Centers for Disease Control and Prevention. Vaccines & Immunizations. About VISs. CDC. August 2, 2024. https://www.cdc.gov/vaccines/hcp/about-vis/index.html. Accessed December 19, 2024.
Centers for Disease Control and Prevention. Vaccines & Immunizations. Current VISs. October 17, 2024. https://www.cdc.gov/vaccines/hcp/vis/current-vis.html. Accessed December 19, 2024.
Samad F, Burton SJ, Kwan D, et al. Strategies to Reduce Errors Associated with 2-Component Vaccines [published correction appears in Pharmaceut Med. 2021 Jan;35(1):65. doi: 10.1007/s40290-020-00369- 2]. Pharmaceut Med. 2021;35(1):1-9. doi:10.1007/s40290-020-00362-
9
Centers for Disease Control and Prevention. Vaccine Safety Systems. About the Vaccine Adverse Event Reporting System (VAERS). CDC. August 7, 2024. https://www.cdc.gov/vaccine-safety- systems/vaers/index.html . Accessed Accessed December 21, 2024.
Centers for Disease Control and Prevention. Immunization Information Systems. Immunization Information Systems Resources. CDC. May 17, 2024. https://www.cdc.gov/iis/about/index.html. Accessed December 19, 2024.
Centers for Disease Control and Prevention. You Call the Shots Vaccine Administration: Preventing Vaccine Administration Errors. CDC. January 5, 2021. https://www.cdc.gov/vaccines/hcp/admin/downloads/vaccine-
administration-preventing-errors.pdf. Accessed December 19, 2024.
Centers for Disease Control and Prevention. Vaccines & Immunizations. Screen for Contraindications and Precautions. CDC. May 7, 2019. https://www.cdc.gov/vaccines/hcp/admin/screening.html. Accessed December 19, 2024.
Centers for Disease Control and Prevention. Preventing and Managing Adverse Reactions. CDC. July 25, 2024. https://www.cdc.gov/vaccines/hcp/imz-best-practices/preventing- managing-adverse-reactions.html. Accessed December 19, 2024.
Picard M, Stone CA Jr, Greenhawt M. Management of patients with immediate reactions to COVID-19 vaccines. J Allergy Clin Immunol. 2023;151(2):413-415. doi:10.1016/j.jaci.2022.09.003
Reddy VK, Lavoie MC, Verbeek JH, Pahwa M. Devices for preventing percutaneous exposure injuries caused by needles in healthcare personnel. Cochrane Database Syst Rev. 2017;11(11):CD009740. Published 2017 Nov 14. doi:10.1002/14651858.CD009740.pub3
Centers for Disease Control and Prevention. Advisory Committee on Immunization Practices (ACIP). ACIP Recommendations. CDC. August 29, 2024. https://www.cdc.gov/acip/vaccine-
recommendations/index.html. Accessed December 21. 2024.
Westrick SC, Patterson BJ, Kader MS, Rashid S, Buck PO, Rothholz MC. National survey of pharmacy-based immunization services. Vaccine. 2018;36(37):5657-5664. doi:10.1016/j.vaccine.2018.07.027
Centers for Disease Control and Prevention. Vaccine Information for Adults. Where to Find Vaccines. CDC. July 10, 2024. https://www.cdc.gov/vaccines-adults/recommended-vaccines/vaccine- planning.html. Accessed December 21. 2024.
Centers for Disease Control and Prevention. Vaccines & Immunizations. Routine Measles, Mumps, and Rubella Vaccination. CDC. January 26, 2021.
https://www.cdc.gov/vaccines/vpd/mmr/hcp/recommendations.html. Accessed December 19, 2024.
Holm MR, Poland GA. Critical aspects of packaging, storage, preparation, and administration of mRNA and adenovirus-vectored COVID-19 vaccines for optimal efficacy. Vaccine. 2021;39(3):457-459. doi:10.1016/j.vaccine.2020.12.017
Centers for Disease Control and Prevention. Advisory Committee on Immunization Practices (ACIP). ACIP Recommendations. CDC. August 29, 2024. https://www.cdc.gov/acip/vaccine-
recommendations/index.html. Accessed December 21. 2024.
ACIP Shared Clinical Decision-Making Recommendations. Advisory Committee on Immunization Practices (ACIP). Centers for Disease Control and Prevention. September 3, 2024. https://www.cdc.gov/vaccines/hcp/vis/vis-statements/mmrv.pdf . Accessed December 21, 2024.
Centers for Disease Control and Prevention. Pfizer-BioNTech COVID-19 Vaccine Products At-A-Glance. 2022. CDC. https://www.memorialcare.org/sites/default/files/_images/content/Vacc ine-Resources/MHS_VaccineClinics_Pfizer%20Vaccine-at-a- glance_2022_06_24.pdf. Accessed December 21, 2024.
ine-Resources/MHS_VaccineClinics_Pfizer%20Vaccine-at-a- glance_2022_06_24.pdf. Accessed December 21, 2024.
U.S. Food and Drug Administration. FDA Approves First COVID-19 Vaccine. FDA. August 23, 2021. https://www.fda.gov/news- events/press-announcements/fda-approves-first-covid-19-vaccine. Accessed December 19, 2024.
U.S. Food and Drug Administration. Moderna COViD-19 Vaccine. FDA. August 22, 2024. https://www.fda.gov/vaccines-blood- biologics/coronavirus-covid-19-cber-regulated-biologics/moderna-covid- 19-vaccine. Accessed December 19, 2024.
COVID-19 Vaccine, Adjuvanted (2024-2025 Formula). Novavax. 2024. https://us-hcp.novavaxcovidvaccine.com/?gclid=CjwKCAiApY- 7BhBjEiwAQMrrEUgYLcSPu3- iXVJeb75L_q0HioJ7e9ENcyC0ol0X33Pl3MBfnirolxoCjiYQAvD_BwE&gad_s ource=1. Accessed December 19, 2024.
North Dakota Health & Human Services. COVID-19 Vaccine Comparison Table For Individuals 12 Years and Older. North Dakota HHS. September 2024. https://www.hhs.nd.gov/sites/www/files/documents/Adult_COVID- 19_Vaccine_Comparison_Table.pdf. Accessed December 19, 2024.
Centers for Disease Control and Prevention. Stay Up to Date with COVID-19 Vaccines Including Boosters. 2023. CDC. https://www.cdc.gov/covid/vaccines/stay-up-to- date.html?CDC_AAref_Val=https://www.cdc.gov/coronavirus/2019- ncov/vaccines/stay-up-to-date.html. Accessed December 21, 2024.
Britton A, Roper LE, Kotton CN, et al. Use of Respiratory Syncytial Virus Vaccines in Adults Aged ≥60 Years: Updated Recommendations of the Advisory Committee on Immunization Practices - United States, 2024. MMWR Morb Mortal Wkly Rep. 2024;73(32):696-702. Published 2024 Aug 15.
doi:10.15585/mmwr.mm7332e1
Kobayashi M, Leidner AJ, Gierke R, et al. Use of 21-Valent Pneumococcal Conjugate Vaccine Among U.S. Adults: Recommendations of the Advisory Committee on Immunization Practices - United States, 2024. MMWR Morb Mortal Wkly Rep. 2024;73(36):793-798. Published 2024 Sep 12.
doi:10.15585/mmwr.mm7336a3
DeMarco M, Carter C, Houle SKD, Waite NM. The role of pharmacy technicians in vaccination services: a scoping review. J Am Pharm Assoc (2003). 2022;62(1):15-26.e11. doi:10.1016/j.japh.2021.09.016
King J. Navigating the World of Vaccine Billing. Pharmacy Times. June 24, 2019. https://www.pharmacytimes.com/view/navigating-the-world-
of-vaccine-billing. Accessed December 21, 2024.
Measles, Mumps, and Rubella Vaccination: What Everyone Should Know. Vaccines and Immunizations. Centers for Disease Control and Prevention. January 26, 2021. https://www.cdc.gov/vaccines/vpd/mmr/public/index.html. Accessed December 21, 2024.
Centers for Disease Control and Prevention. Immunization information systems (IIS). IIS Policies: Florida. CDC. August 9, 2024. https://www.cdc.gov/iis/policy- legislation/florida.html#:~:text=Florida%27s%20IIS%20is%20called% 20Florida,381.003(1)(e). Accessed December 21, 2024.
Florida Health. Florida Shots. Florida Health. 2024. https://flshotsusers.com/. Accessed December 21, 2024.
Florida Statutes Annotated, § 381.003(1)(e). 2024.
DISCLAIMER
The information provided in this course is general in nature, and it is solely designed to provide participants with continuing education credit(s). This course and materials are not meant to substitute for the independent, professional judgment of any participant regarding that participant’s professional practice, including but not limited to patient assessment, diagnosis, treatment, and/or health management. Medical and pharmacy practices, rules, and laws vary from state to state, and this course does not cover the laws of each state; therefore, participants must consult the laws of their state as they relate to their professional practice.
Healthcare professionals, including pharmacists and pharmacy technicians, must consult with their employer, healthcare facility, hospital, or other organization, for guidelines, protocols, and procedures they are to follow. The information provided in this course does not replace those guidelines, protocols, and procedures but is for academic purposes only, and this course’s limited purpose is for the completion of continuing education credits.
Participants are advised and acknowledge that information related to medications, their administration, dosing, contraindications, adverse reactions, interactions, warnings, precautions, or accepted uses are constantly changing, and any person taking this course understands that such person must make an independent review of medication information prior to any patient assessment, diagnosis, treatment and/or health management. Any discussion of off-label use of any medication, device, or procedure is informational only, and such uses are not endorsed hereby.
Nothing contained in this course represents the opinions, views, judgments, or conclusions of RxCe.com LLC. RxCe.com LLC is not liable or responsible to any person for any inaccuracy, error, or omission with respect to this course, or course material.
© RxCe.com LLC 2025: All rights reserved. No reproduction of all or part of any content herein is allowed without the prior, written permission of RxCe.com LLC.
