ULCERATIVE COLITIS INSIGHTS: A PRIMER FOR PHARMACY TEAMS
Liz Fredrickson, PharmD, BCPS
Liz Fredrickson is an Associate Professor of Pharmacy Practice and Pharmaceutical Sciences at the Northeast Ohio Medical University (NEOMED) College of Pharmacy.
Topic Overview
Ulcerative colitis (UC) is a chronic, inflammatory disease of the colon and rectum, and, along with Crohn’s disease (CD), is one of two gastrointestinal (GI) disorders that comprise inflammatory bowel disease (IBD). This condition is relapsing and remitting in nature, with inflammation limited to the colon's mucosal layer. The appropriate diagnosis and treatment of UC is critical to maintaining patients' quality of life and avoiding disease complications. The most recent American College of Gastroenterology (ACG) guidelines focus on pharmacologic therapies and novel algorithms for starting, optimizing, and monitoring responses to these medications. These guidelines share evidence- based recommendations alongside the suggestion to treat patients with UC using a shared decision-making model. Pharmacists are vital to the care team. They can use their medication expertise to guide treatment decisions and counsel patients.
Accreditation Statement

RxCe.com LLC is accredited by the Accreditation Council for Pharmacy Education (ACPE) as a provider of continuing pharmacy education.
Universal Activity Number (UAN): The ACPE Universal Activity Number assigned to this activity is
Pharmacist 0669-0000-24-045-H01-P
Pharmacy Technician 0669-0000-24-046-H01-T
Credits: 2 contact hour(s) (0.2 CEU(s)) of continuing education credit
Type of Activity: Knowledge
Media: Internet/Home study Fee Information: $6.99
Estimated time to complete activity: 2 contact hour(s) (0.2 CEU(s)), including Course Test and course evaluation
Release Date: April 9, 2024 Expiration Date: April 9, 2027
Target Audience: This educational activity is for pharmacists and pharmacy technicians
How to Earn Credit: From April 9, 2024, through April 9, 2027, participants must:
Read the “learning objectives” and “author and planning team disclosures;”
Study the section entitled “Educational Activity;” and
Complete the Course Test and Evaluation form. The Course Test will be graded automatically. Following successful completion of the Course Test with a score of 70% or higher, a statement of participation will be made available immediately. (No partial credit will be given.)
Credit for this course will be uploaded to CPE Monitor®.
Learning Objectives: Upon completion of this educational activity, participants should be able to:
Describe the etiology and pathophysiology of ulcerative colitis (UC)
Compare and Contrast pharmacologic therapies for UC
Identify the mechanisms of action and side effect profiles of medications used to treat UC
Provide medication counseling points for patients with UC
Disclosures
The following individuals were involved in developing this activity: Liz Fredrickson, PharmD, BPCS, and Pamela Sardo, PharmD, BS. Pamela Sardo and Liz Fredrickson have no conflicts of interest or financial relationships regarding the subject matter discussed. There are no financial relationships or commercial or financial support relevant to this activity to report or disclose by RxCe.com or any of the individuals involved in the development of this activity.
© RxCe.com LLC 2024: All rights reserved. No reproduction of all or part of any content herein is allowed without the prior, written permission of RxCe.com LLC.
Educational Activity
Ulcerative Colitis Insights: A Primer for Pharmacy Teams Introduction
Ulcerative colitis is a chronic, inflammatory disease of the colon and rectum. This continuing education program will review its etiology, pathophysiology, and classification. The appropriate diagnosis and treatment of UC is critical to maintaining a patient’s quality of life and avoiding complications from the disease. Available pharmacologic treatments will be discussed, including their mechanisms of action, side effect profiles, and their recommended place in therapy. Evidence-based recommendations are also shared alongside the suggestion to treat patients with UC using a shared decision-making model. The role of pharmacists and pharmacy technicians as members of interdisciplinary care teams will also be discussed in relation to the diagnosis and management of this disease.
Etiology and Epidemiology
Ulcerative colitis (UC) is a chronic, inflammatory disease of the colon and rectum, and, along with Crohn’s disease (CD), is one of two gastrointestinal (GI) disorders that comprise inflammatory bowel disease (IBD).1 This condition is relapsing and remitting in nature, with inflammation limited to the colon's mucosal layer.1 Ulcerative colitis is an idiopathic disease with no clear etiology, but genetic, environmental, and immunologic factors may influence its development.1
Cases of UC have increased globally in recent years.1 Within the United States (US), the incidence is between 1.2 and 20 cases per 100,000 persons per year, and more than 1 million Americans have been diagnosed with the disease.1,2 Most patients are diagnosed with UC in the second or third decade of life, though individuals of all ages may receive the diagnosis.3 After the age of 45, females are between 13 and 32% less likely to be diagnosed with UC than males.4
Risk Factors
As with CD, the precise etiology of UC is unknown; however, there are numerous factors that, in combination, are believed to put patients at risk of developing this disease. These encompass genetic, infectious, immunologic, and environmental causes.1 Potential triggers of UC onset include recent smoking cessation, nonsteroidal anti-inflammatory drug use, and enteric infections.5-8
The Microbiome
Microorganisms are believed to play an important role in triggering inflammation and, ultimately, the development of IBD.9 This may begin with dysbiosis (an imbalance in gut microbes) or increased proinflammatory bacteria within the GI tract.9 Compared to healthy individuals, patients with IBD have fewer numbers and less diversity of microbiota in addition to the presence of more aggressive bacterial groups.9 Various microorganisms, including viruses, protozoans, and mycobacteria, have been thought to play a role in developing IBD.9
Genetics
A family history of IBD is a significant risk factor for its development; patients with a first-degree relative with IBD have a 20-fold increased risk of the disease.9,10 An individual’s genetic predisposition to IBD varies by population, with individuals of African American and Asian descent having lower risks.11
Immune-Mediated Risks
Autoimmune and nonautoimmune mechanisms may contribute to the development of IBD.9 The mucosal immune system encompasses 75% of all lymphocytes within healthy individuals, making it the largest component of the immune system.10 After the loss of effective intestinal barrier function,
lymphocytes, plasma cells, mast cells, macrophages, and neutrophils can infiltrate the bowel wall and form granulomas.9
Environment and Lifestyle
An individual’s environment and lifestyle, including their diet and medication use, may play important roles in the development of IBD.11,12 Various environmental risk factors have been noted for IBD, including cigarette smoking, antibiotic use during childhood, breastfeeding, use of oral contraceptives, low vitamin D levels, and tea and coffee consumption.11 These risk factors are compared between CD and UC in Table 1.11
Table 1
Environmental and Lifestyle Risk Factors for IBD Development11
| Risk Factor | Crohn’s Disease | Ulcerative Colitis |
| Smoking | Increased risk in Caucasians | Protective effect in Caucasians and Asians |
| Antibiotic use in childhood | Increased risk for Caucasians; Protective effect in Asians | Increased risk in Caucasians |
Breastfeeding | Protective effect in Asians and Caucasians in most studies | Protective effect in Asians and Caucasians in most studies |
| Oral contraceptive use | Risk in Caucasians | Inconclusive |
| Low vitamin D levels | Risk in Caucasians | Risk in Caucasians |
| Tea or coffee consumption | Risk in Caucasians | Protective in Asians |
It is thought that a Western diet, which includes a high intake of fat and sugar and a low intake of fiber, heightens the risk of IBD.11 Also, the hygiene hypothesis may explain the increased incidence of IBD in industrialized
countries. This theory suggests exposure to various microorganisms in early childhood through things such as family pets and living on a farm can protect against the development of autoimmune and allergic diseases.11
Pathophysiology
The pathophysiology of UC centers on interactions between an individual’s genes and their environment, which ultimately results in an abnormal immune response to microbes within the gut.10 The pathophysiologic features of UC and CD can overlap, complicating the diagnosis of one disease over the other. This is especially true when inflammation is limited to the colon.9 The main distinguishing features are the extent and distribution of inflammation and the depth of bowel wall involvement.9 These features are compared in Table 2.9
Table 2
Comparison of the Pathologic Features of CD and UC9
| Feature | CD | UC |
| Rectal involvement | Rare | Common |
| Ileal involvement | Very common | Rare |
| Strictures | Common | Rare |
| Fistulas | Common | Rare |
| Transmural involvement | Common | Rare |
| Crypt abscesses | Rare | Very common |
| Granulomas | Common | Rare |
| Linear clefts | Common | Rare |
| Cobblestone appearance | Common | Absent |
Whereas CD occurs throughout the entire bowel, UC is confined to the colon and rectum, affecting the mucosal and submucosal layers.9 An estimated 30-60% of patients will be diagnosed with proctitis (rectal inflammation), 16- 45% with left-side colitis (inflammation of the left side of the colon), and 14- 35% with extensive or pancolitis (widespread inflammation of the colon).2
Given its superficial mucosal involvement, UC is not associated with the fistulas, perforation, or obstructions seen with CD.9 However, abscesses can form in the crypts of Lieberkühn due to infiltration of lymphocytes, plasma cells, and granulomas.9 This damage can lead to significant diarrhea and bleeding.9
Ulcerative colitis complications include hemorrhoids, anal fissures, perirectal abscesses, and toxic megacolon.9 Toxic megacolon is a serious, major complication that occurs when systemic signs of toxicity occur and the colon distends more than six centimeters.2 Signs and symptoms of toxic megacolon include high fever, tachycardia, distended abdomen, elevated white blood cell count, and dilated colon.13
Individuals with UC also have an elevated risk of colorectal carcinoma (CRC), and this accounts for 10-15% of deaths among UC patients.9 Risk factors include diagnosis at a young age, long duration of disease, large extent of colonic involvement, active histological inflammation, family history of CRC before age 50, and male gender.14
Extraintestinal Manifestations of IBD
Ulcerative colitis and CD are associated with organ involvement outside the GI tract or extraintestinal manifestations.9 These complications are summarized in Table 3.9
| Complication | Details |
| Hepatobiliary | Fatty infiltration resulting malabsorption, protein-losing enteropathy, corticosteroid Includes nonalcoholic fatty liver disease, pericholangitis, autoimmune hepatitis, liver abscess, cirrhosis |
| Joint | Includes peripheral and axial arthropathies Symptoms can occur prior to development of IBD symptoms |
| Ocular | Includes dry eye, blepharitis, iritis, uveitis, episcleritis, conjunctivitis Occurs in ~29% of IBD patients |
| Dermatologic and Mucocutaneous | Includes erythema nodosum, pyoderma gangrenosum, aphthous ulceration, Sweet’s syndrome |
| Hematologic, Coagulation, Pulmonary, Metabolic Abnormalities | Includes anemia (prevalence of 74%), iron deficiency, chronic blood loss, inflammation, malnutrition, hemolysis, or bone marrow suppression due to medications Increased risk of venothromboembolism Osteopenia in 32-36% of patients Osteoporosis in 2-15% of patients Includes pneumonia, bronchiectasis, bronchiolitis obliterans |
Table 3 Extraintestinal Manifestations of IBD9
Clinical Presentation and Diagnosis Clinical Presentation
Patients with UC can present with various symptoms that depend on the disease's location and severity (Table 4).9,10 The most common symptoms include those indicative of an inflamed rectum: bleeding (90% of patients), increased stool frequency (90% of patients), and tenesmus, or a sense of pressure (75-90% of patients).9,10 If no other potential cause is present, these symptoms should trigger consideration of a UC diagnosis.1
Table 4
Signs and Symptoms of Ulcerative Colitis
Abdominal cramping
Frequent bowel movements, often with blood in the stool
Weight loss
Fever and tachycardia in severe disease
Blurred vision, eye pain, and photophobia with ocular involvement
Arthritis
Raised, red, tender nodules that vary in size from 1 cm to several centimeters
Most patients with UC will experience periods of symptoms intermixed with times of remission.1,9 This is known as a relapsing/remitting pattern of disease.1
Diagnosis
A full medical history, colonoscopy, stool examination, pathology, and laboratory findings are needed to diagnose UC.9,10 A thorough medical history is necessary to determine what may have triggered disease onset, the severity of the disease, and potential causes.1 The frequency of bowel movements should be determined, and bleeding can be assessed by asking about the number of bowel movements mixed with blood.1 Clinicians should also ascertain the presence of extraintestinal manifestations described above.1,9 When inflammation is limited to the colon, it can be challenging to differentiate CD from UC. Differentiating clinical features are detailed in Table 5.9
Table 5
Clinical Features of Crohn’s Disease and Ulcerative Colitis9
| Feature | Crohn’s Disease | Ulcerative Colitis |
| Clinical | ||
| Malaise, fever | Common | Uncommon |
| Rectal bleeding | Common | Common |
| Feature | Crohn’s Disease | Ulcerative Colitis |
| Abdominal tenderness | Common | May be present |
| Abdominal mass | Common | Absent |
| Abdominal pain | Common | Unusual |
| Abdominal wall and internal fistulas | Common | Absent |
| Distribution | Discontinuous | Continuous |
| Aphthous or linear ulcers | Common | Rare |
Laboratory Testing
In some cases, patients may have normal bloodwork.12 Various laboratory tests can help identify the presence of UC, including the inflammatory markers, erythrocyte sedimentation rate (ESR) and C-reactive protein (CRP). While nonspecific, these markers are commonly elevated in active UC.1,9 However, up to a quarter of patients may have normal CRP levels despite having endoscopically active disease.1 Up to 70% of cases will have positive perinuclear antineutrophil cytoplasmic antibodies (pANCAs); elevated pANCA levels, in combination with negative anti-Saccharomyces cerevisiae antibodies, can help to establish the diagnosis.1 Fecal calprotectin (FCal) can be used to distinguish IBD from irritable bowel syndrome.1 Common laboratory tests are detailed in Table 6.9
| Test | Finding |
| Hematocrit/hemoglobin | Decreased |
| ESR, CRP, fecal calprotectin | Increased |
| White blood cell count | Increased |
| Albumin | Decreased |
Table 6 Laboratory Tests9
| Fecal calprotectin | Increased |
| Perinuclear antineutrophil cytoplasmic antibodies | Positive test |
Imaging
Ulcerative colitis should be histologically confirmed via a lower gastrointestinal endoscopy.1 A complete colonoscopy assists clinicians in assessing the full extent of the disease and ruling out CD.1 If a patient has severe disease at diagnosis, a sigmoidoscopy with a biopsy can be considered instead, as colonoscopy increases the risk of perforation.1 Ulcerative colitis will appear as vascular markings, granularity, and friability of the mucosa on endoscopy. Deep ulcerations and spontaneous bleeding may also be seen in patients with severe disease.1
Disease Extent and Severity
Once UC has been diagnosed, it is important to determine which sections of the colon are involved, as this will affect prognosis and treatment decisions.9 Distal disease is inflammation limited to areas distal to the splenic flexure.9 This is also called “left-sided” disease. When inflammation extends proximally to the distal flexure, this is termed “extensive disease.”9 Proctitis is inflammation confined to the rectal area, and proctosigmoiditis is inflammation involving the rectum and sigmoid colon.9 The extent of disease can be categorized per the Montreal classification system as proctitis (E1), left-sided colitis (E2), or extensive colitis (E3).15 Notably, the extent of disease can worsen over time; in one systematic review, 30% of patients had more extensive disease 10 years after their diagnosis.16
Clinicians can use the Mayo endoscopic scoring system to categorize UC based on severity (Table 7).1,17 Higher scores indicate worse disease.17 The severity assessment of a patient with UC should also determine the need for colectomy, response to medications, and predictors of aggressive disease courses.1
Table 7
Mayo Endoscopic Scoring System
| Mild | Moderate | Severe | |
| Stool frequency | 1-2 stools/day above baseline (1 point) | 3-4 stools/day above baseline (2 points) | 5 or more stools/day above baseline (3 points) |
| Rectal bleeding | Streaks of blood with stool less than half the time (1 point) | Obvious blood with stool most of the time (2 points) | Blood passed alone (3 points) |
| Physical global assessment | Mild disease (1 point) | Moderate disease (2 points) | Severe disease (3 points) |
| Endoscopic assessment | Erythema, decreased vascular pattern (1 point) | Marked erythema, absent vascular pattern, friability, erosions (2 points) | Spontaneous bleeding, ulcerations (3 points) |
The American Gastroenterological Association (AGA) guidelines provide another definition of acute severe UC, which requires hospitalization and having greater than six bloody stools per day and at least one marker of systemic toxicity: heart rate greater than 90 beats per minutes, temperature greater than 37.8°C, hemoglobin less than 10.5 g/dL (105 g/L; 6.52 mmol/L), and/or ESR greater than 30 mm/hr. (8.3 µm/s).18 Patients can also be categorized based on risk for colectomy. Low risk encompasses patients with limited anatomic extent and mild endoscopic disease, and high risk encompasses extensive colitis, age less than 40 years, deep ulcers, high ESR/CRP, steroid dependence, history of hospitalization, and C. difficile or cytomegalovirus infection.9
Management of Ulcerative Colitis
Importantly, there is no known cure for UC. Thus, UC treatment aims to increase a patient’s quality of life, decrease disease complications, promote mucosal healing, control inflammation, control symptoms resulting from active inflammation, and minimize adverse effects of drug therapies.1,9 Guidelines for treating UC include novel algorithms for starting, optimizing, and monitoring responses to pharmacologic therapies.1 Pharmacists are vital to the care team and can use their medication expertise to guide treatment decisions and counsel patients.
For some patients, UC will be self-limited.9 The ACG guidelines note UC management must include a quick, accurate diagnosis, assessment of outcomes, and initiation of therapies that are not only effective but safe and tolerable.1 Treatments should be selected based on inflammatory activity and disease prognosis.1
A broad goal of UC management is to achieve a sustained, steroid-free remission period.1 Clinicians should also work to ensure patients have the necessary psychosocial support and a good quality of life while preventing surgery, hospitalization, and the prevention of cancer.1 Finally, shared decision-making models should be utilized. Mucosal healing can occur before symptom relief, and patients should be educated regarding the expected course of disease.1
Nonpharmacologic and Lifestyle Approaches
Patients may be counseled to alter their diet depending on disease severity.9 In general, if a specific food has been found to cause or worsen symptoms, patients may try eliminating that food to see if symptoms improve.9 However, it is critical that this is done deliberately to avoid malnutrition, and dieticians may be consulted to assist with this process. Specific diets, including low FODMAP and the specific carbohydrate diet (SCD) may also be trialed as these have been shown to improve IBD symptoms in some cases.9 The specific carbohydrate diet involves the restriction of all
carbohydrates except for monosaccharides, including glucose, fructose, and galactose.9 Milk, canned fruits and vegetables, and processed meats are also eliminated with this diet.9
The addition of probiotics as adjuncts to pharmacologic therapies has been evaluated, as these agents can establish normal gut bacterial flora and promote beneficial effects on host immune response.9 Some studies have found benefits related to the induction and remission of UC; however, these trials may be limited by variable methodologies and the probiotic formulations studied.19,20
Clinicians should also work closely with patients to assess and manage stress, depression, and anxiety. Numerous studies have found these psychological issues have the potential to increase symptoms of IBD. Perceived stress can derive from issues around disease management and the impact of UC on a patient’s life.21 Additionally, patients with anxiety or major depression also have a greater risk of surgery and use of healthcare resources.22
Pharmacologic Approaches
The choice of medication in UC is determined based on the patient’s specific diagnosis, disease activity, and disease prognosis.1 Treating based on prognosis and not just inflammatory activity is crucial. Pharmacists play a key role in the management of UC by reviewing medication lists to avoid drug- drug interactions, monitoring for and mitigating adverse effects of UC therapies, and assisting with therapeutic drug monitoring. Pharmacists can also provide critical patient education to optimize therapy adherence. Pharmacologic therapy aims to improve symptoms and quality of life while preventing treatment complications.1,10 Recommended agents are reviewed below.
Mesalamine (5-aminosalicylic acid)
Mesalamine is a topical anti-inflammatory agent shown to be effective within the lumen of the intestine.9 This medication is the active component of sulfasalazine, with an unclear mechanism of action.9 It may exert benefits via scavenging free radicals, inhibiting leukocyte motility, and interfering with TNF-alpha.9,23 It can be used in an enema formulation to treat left-sided disease or in suppository form to treat proctitis.9 Side effects include delayed hypersensitivity reactions with potential fever, myocarditis/pericarditis, interstitial nephritis, liver injury, pancreatitis, and interstitial pulmonary disease.23 Intolerance syndrome (diarrhea, fever, and abdominal pain may occur in 7-14% of patients) and renal effects (interstitial nephritis, renal failure syndrome) are also risks.23 Dosing is detailed in Table 8 below.
Sulfasalazine is a combination of mesalamine with sulfapyridine, which is a carrier that delivers 5-aminosalicylic acid (aka 5-ASA) to the colon.24 It is available in both immediate and delayed-release formulations, with a recommended dose of 3-6 grams per day in divided doses for up to 16 weeks.
24 Significant side effects include blood dyscrasias, GI effects (nausea, vomiting, diarrhea), and delayed hypersensitivity reactions.24 Pharmacists can assist the care team in determining the best formulation of ASA agents to increase adherence, considering the patient's preferences.
| Drug | Brand Name | Starting Dose | Dosing Range |
| Mesalamine suppository | Canasa | 1 g | 1 g daily to three times weekly |
| Mesalamine enema | Rowasa | 4 g | 4 g daily to three times weekly |
Table 8 Mesalamine dosing9
| Mesalamine (oral) | Asacol HD Apriso Lialda Pentasa Delzicol | 1.6 g/day 1.5 g/day 1.2-2.4 g/day 2 g/day 1.2 g/day | 2.8-4.8 g/day 1.5 g/day once daily 1.2-4.8 g/day once daily 2-4 g/day 2.4-4.8 g/day |
| Olsalazine | Dipentum | 1.5 g/day | 1.5-3 g/day |
| Balsalazide | Colazal | 2.25 g/day | 2.25-6.75 g/day |
Corticosteroids
Corticosteroids modulate the immune system and inhibit the production of cytokines and mediators. They can be given via oral, parenteral, or rectal routes.9 Budesonide is a topical agent with a large first-pass effect, which minimizes a patient’s systemic exposure and lessens side effects. Budesonide has shown superiority compared with continuing 5-ASA or placebo in patients who failed to respond to 5-ASA.1 Budesonide MMX has also shown efficacy in inducing remission in patients with mildly active UC.1
Immunomodulators
Immunomodulator medications include azathioprine, 6-mercaptopurine, and cyclosporine.1 These agents are slow-acting and ineffective at inducing remission in moderately-severely active UC.1
Azathioprine is the prodrug of mercaptopurine, and both agents can be used long-term to treat UC.25 Thiopurine methyltransferase (TMPT) testing is important to consider before starting azathioprine or 6- mercaptopurine.1 Dosing is 50 mg once daily. This can be titrated up to
2.5 mg/kg once daily over ≥12 weeks as indicated and tolerated. Adverse effects include allergic reactions, pancreatitis, myelosuppression, nausea, infections, hepatotoxicity, and malignancies that include nonmelanoma skin cancer, and lymphoma.25
Cyclosporine inhibits the production and release of interleukin II and inhibits interleukin II-induced activation of resting T-lymphocytes.26 The intravenous dose is 2 to 4 mg/kg/day, infused continuously over 24 hours.26 Cyclosporine (modified) can be given orally at a dose of 2.3 to
3 mg/kg every 12 hours. Side effects include nephrotoxicity and neurotoxicity.26
Biologics
Anti-TNF agents include infliximab, adalimumab, and golimumab. All have similar efficacy rates.9 The choice often depends on the patient’s preference, medication cost, and administration route.9 The agents are detailed in Table 9.9
Table 9
Anti-TNF Agents9,27-29
| Generic (Brand) | Description | Dose |
| Infliximab (Remicade) | Chimeric mouse-human IgG1 monoclonal antibody | 5 mg/kg at 0, 2, and 6 weeks, followed by 5 mg/kg every 8 weeks thereafter |
| Adalimumab (Humira) | Fully human IgG1 monoclonal antibody directed against TNF-α | Initial: 160 mg (given over 1 or 2 days), then 80 mg 2 weeks later (day 15). Maintenance: 40 mg every other week beginning day 29. If a disease flare occurs, some experts increase to 40 mg every week |
| Golimumab (Simponi) | Human monoclonal antibody that binds to human tumor necrosis factor-alpha (TNFα), thereby interfering with endogenous TNFα activity | Induction: 200 mg at week 0, then 100 mg at week 2, followed by maintenance therapy of 100 mg every 4 weeks |
Patients at risk of serious side effects from anti-TNF agents include those with prior demyelinating disorders, congestive heart failure, and prior lymphoma or malignancies.1 Clinicians should assess patients for active or latent tuberculosis before starting anti-TNF therapies.1 They should also be screened for opportunistic infections, including histoplasmosis and blastomycosis.1 For patients at high risk of TB, this testing should also be considered before starting corticosteroids or other immunomodulators. If TB is discovered, the patient will require chemoprophylaxis for weeks to months prior to starting anti-TNF medications.1 Patients should also be assessed for viral hepatitis before starting therapy, and carriers should receive treatment to avoid a hepatitis B flare and liver failure.1
Pharmacists can assist the care team in ensuring patients are up to date on vaccinations, including pneumococcal pneumonia, varicella, human papillomavirus, inactivated influenza, hepatitis A, and herpes zoster.1 These should be given before starting therapy.1 Live attenuated vaccines should be avoided.1
Biosimilar anti-TNF agents include biosimilar infliximab and biosimilar adalimumab.9 A biosimilar is a product highly similar to the reference product with minor differences in clinically inactive components and no clinically meaningful differences between the products regarding safety, potency, or purity.9 Biosimilars for infliximab include infliximab-dyyb, CT-P13, and infliximab-abda, infliximab-qbtx.2 Adalimumab has adalimumab-atto and adalimumab-adbm.1,9 Every biosimilar has a structural complexity and different glycosylation patterns, though the amino acid sequence remains the same.1 Biosimilars are not the same as generic medications because they differ in solubility, stability, clearance, and immunogenicity from one another.9
Other Agents
Ustekinumab is an anti-p40 antibody that inhibits IL-12 and 23.1 Overall, it has a good safety profile. It is an option for the long-term management of moderate to severe UC and is dosed based on the patient’s
weight. Side effects include potential posterior reversible encephalopathy syndrome, serious allergic reactions, lung inflammation, and cancer.
Vedolizumab selectively inhibits α4β7 integrin interaction with mucosal addressin cell adhesion molecule-1.31 This makes it relatively specific for leukocyte trafficking to the gut.31 The onset of effect is slower than with anti- TNF agents; however, it can take up to 10 weeks.31 IV dosing is 300 mg at 0, 2, and 6 weeks and then every eight weeks thereafter. Therapy should be discontinued in patients who show no evidence of therapeutic benefit by week
14. Maintenance dosing is 108 mg SUBQ once every two weeks, beginning after at least 2 IV infusions.31
Tofacitinib and upadacitinib inhibit intracellular Janus kinase (JK) proteins.32,33 Ozanimod is inhibits sphingosine-1 phosphate (S1P).34 Induction dosing for tofacitinib is 10 mg twice daily for at least eight weeks; based on the therapeutic response, may continue 10 mg twice daily for a maximum of
16 weeks or transition to the maintenance dose.32 Discontinue therapy if inadequate response is achieved after 16 weeks using 10 mg twice daily.32 Maintenance dosing is mg twice daily. If the patient experiences a loss of response of 5 mg twice daily, then use 10 mg twice daily after assessing the benefits and risks and use it for the shortest duration.32 Induction dosing for upadacitinib is 45 mg once daily for eight weeks.33 Maintenance dosing is 15 mg once daily, and this may be increased to 30 mg once daily in patients with refractory, severe, or extensive disease.33 Discontinue if an adequate response is not achieved with the 30 mg dose.33 Side effects of these medications include increased risk of serious infections, increased risk of major adverse cardiovascular events, cancer/immune system problems, thrombosis, stomach perforations, and allergic reactions.32,33
Ozanimod is dosed at 0.23 mg once daily on days 1 through 4, then
0.46 mg once daily on days 5 through 7.34 The maintenance dosing is 0.92 mg once daily starting on day 8.34 The side effects include upper respiratory tract infection, increased liver function tests, headache, pyrexia, nausea, arthralgia.34
Induction Therapy for Mild-Moderate Active Disease
A treatment algorithm for UC is depicted in the Figure below.
Treatment of UC Based on Disease Severity10
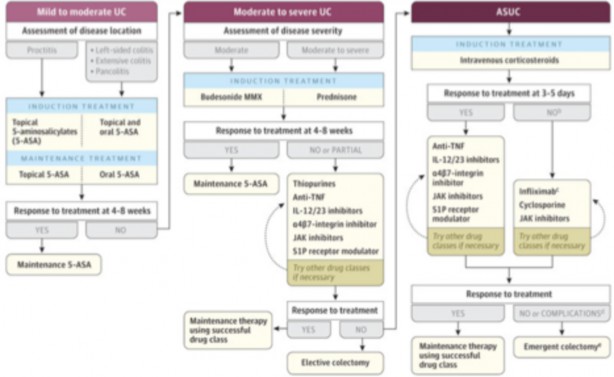
For patients with mild-moderate disease, management with either oral or topical 5-ASA or oral or rectal budesonide is reasonable.9 For isolated proctitis, topical 5-ASA is recommended. A meta-analysis that included 11 randomized control trials (RCT) of patients who were treated with 5-ASA for induction or maintenance found this therapy was superior to placebo whether remission was defined clinically or endoscopically.35 In general, mesalamine products are preferred over sulfasalazine. The dose of mesalamine for patients with extensive disease is 2-3 grams per day.1 If patients do not respond, the dose can be increased above 3 grams.1 Patients with distal disease benefit from topical mesalamine (either enema or suppository), while those with extensive disease do best with a combination of topical and oral therapies.1
The ACG guidelines do not recommend switching to an alternative 5- ASA formulation if a patient with mildly active UC fails to achieve remission with a different preparation.1,36 Patients who don’t respond to mesalamine can consider controlled-release budesonide if they have mildly active extensive UC.1,10 Oral budesonide is given at a dose of 9 mg/day.37 Additionally, oral corticosteroids can be considered at doses of 40-60 mg/day (prednisone equivalent) as an alternative to budesonide for those with moderately active extensive disease who are either refractory to oral ASAs or need better symptom control.1,10 Clinical response should be seen within one week.1 Steroid tapers can be tailored to the patient and guided by clinical symptoms and the use of alternative therapies.1
Medication adherence is a critical component in the treatment of mildly active UC.1 An estimated 68% of patients on more than four prescription medications are likely to be nonadherent.38 Patients who do not remain adherent are likely to have their disease relapse, and pharmacists can play an important role in assisting patients with strategies to optimize adherence to their regimens.
Induction Therapy for Moderate to Severe Active Disease
Mesalamine products are preferred for moderate to severe UC, with an alternative of oral budesonide.1 Systemic corticosteroids can be considered in patients as an alternative induction treatment for those with severe active UC or for patients unresponsive to the maximum doses of mesalamine products.1 The recommended dose of corticosteroids in these cases is 40-60 mg/day (prednisone equivalent).1 ASA therapy may be considered as monotherapy for induction of moderately but not severely active UC.1 Results of a meta- analysis found patients benefited from doses of 2.4 grams per day.1,35,39 The use of a 5–ASA monotherapy has not shown the same effects for severe disease.1 Due to their slow-acting nature, thiopurines are not useful in causing remission in moderate to severe active UC.1
TNF-alpha inhibitors can be considered for patients with moderate to severe disease who do not respond to ASAs or corticosteroids or who are corticosteroid dependent.1 Treatment-naïve patients can consider infliximab or vedolizumab in combination with an immunomodulator like azathioprine.1 The guidelines state a combination of infliximab or vedolizumab with a thiopurine is preferred, as this has shown greater efficacy in inducing corticosteroid-free remission in moderate to severe UC.1 Importantly, vedolizumab should not be combined with TNF-alpha inhibitors.1 Ustekinumab, tofacitinib, or upadacitinib are options for patients with previous exposure to infliximab.1 Finally, ozanimod is an option for induction and maintenance therapy in moderate to severe active UC.1
Therapy for Active Severe or Fulminant Disease
Patients with active severe or fulminant UC cannot be managed outpatient and must be hospitalized.9 Medications are given parenterally, and patients do not eat by mouth (“NPO”) to promote bowel rest.9 Adjacent therapies include VTE prophylaxis and testing and treating for C. Difficile infections if present.9
Methylprednisolone IV 40-60 mg daily is a first-line treatment for this subset of patients.1 Corticosteroids can be trialed for 3-5 days to see if patients can avoid a colectomy.1 If patients are unresponsive, cyclosporine or infliximab can be trialed. Infliximab is dosed as 5 mg/kg IV (single dose), and cyclosporine is given as a continuous infusion at a rate of 2-4 mg/kg/day to achieve a serum level of 200-400 mcg/L.1 If infliximab therapy is successful, patients can continue its use.1 If cyclosporine is successful, patients can switch to azathioprine.1
Maintenance Therapy
The goal of maintenance treatment is to keep patients in remission.1 The choice of medication will depend on the patient’s previous response to treatment, patient-specific considerations, and safety considerations.10
A meta-analysis of 11 trials demonstrated the efficacy of oral 5-ASA agents (mesalamine, olsalazine, and sulfasalazine) compared with a placebo in patients with quiescent UC (distal, left-sided, or extensive colitis) in the maintenance of remission.1 Oral ASA medications are preferred for patients who have mildly active extensive or left-sided disease and can be given at doses of 2+ grams per day.1 In the meta-analysis, higher doses of 5-ASA were associated with lower relapse rates.1 Mesalamine suppositories can be used at doses of 1 gram per day to treat proctitis.1 Over time, these agents can be given at less frequent intervals, with a goal of every third night.1 ASA products can be continued for patients with moderate to severe active disease; however, this is not likely to maintain remission for long periods. Patients who achieved remission of moderate to severe disease with corticosteroids can be given thiopurine for maintenance.1 Overall, corticosteroids should not be used as maintenance therapy, given their host of serious side effects combined with a lack of efficacy to maintain remission.1 In patients who achieve remission, steroids should be tapered over the course of 4-12 weeks.1
Other remission maintenance options with moderate to severe UC include vedolizumab, usteokinumab, tofacitinib, and upadacitinib. They are also options for patients whose azathioprine treatment has failed or who are steroid-dependent.1 If patients achieve remission with either biologic agents or azathioprine, ASA products should be stopped.1
Drug Monitoring
Therapeutic drug monitoring is an important tool for evaluating a drug treatment plan in patients with IBD.40 It is also complex. Pharmacy teams should remember that the association of a side effect in a patient does not always mean the medication caused the side effect. Medical societies and IBD experts support monitoring and its use. The challenge for clinicians is to close the knowledge gaps that have limited its use. 40 Drug therapy monitoring is reviewed in Table 10.
Table 10
Drug Therapy Monitoring10
| Drug(s) | Adverse Drug Reaction | Monitoring Parameters | Comments |
| Mesalamine | Nausea, vomiting, headache | GI disturbances | |
| Corticosteroids | Hyperglycemia, dyslipidemia | Blood pressure, fasting lipid panel | Avoid long-term use if possible or consider budesonide |
| Osteoporosis, hypertension, acne | Glucose, vitamin D, bone density | ||
| Edema, infection, myopathy, psychosis | |||
| Azathioprine/ mercaptopurine | Bone marrow suppression, pancreatitis, lymphoma | Complete blood count | Check TPMT activity or NUDT15 phenotype |
| Liver dysfunction, rash, arthralgia | serum creatinine (Scr), BUN, liver function tests, genotype/ phenotype | May monitor TGN | |
| Infliximab Adalimumab Golimumab | Infusion-related reactions (infliximab), infection Heart failure, optic neuritis, demyelination, injection site reaction, signs of infection | Blood pressure/ heart rate (infliximab) Neurologic exam, mental status Trough concentrations (infliximab) Antidrug antibodies (all agents) | Need negative PPD and viral serologies |
Vedolizumab | Infusion-related reactions | Brain MRI, mental status, progressive multifocal leukoencephalopathy | Vedolizumab not associated with PML |
| Ustekinumab | Infections, skin cancers | Signs/symptoms of infection, annual skin exam | Rare instances of reversible posterior leukoencephalopathy syndrome (RPLS) |
| Drug(s) | Adverse Drug Reaction | Monitoring Parameters | Comments |
| Avoid live vaccines |
Upadacitinib Risk of infection, higher
rate of MACE (CV death, stroke, MI), bone marrow suppression, thrombosis, increase lipids and LFTs, lymphoma, fetal toxicity
Tofacitinib Infection, thrombosis,
lymphoma, elevated cholesterol, CPK, LFTs, lymphopenia, neutropenia, anemia
Ozanimod Infection, heart rate, blood
pressure, LFTs, respiratory rate, fetal abnormalities, macular edema, headache
Symptoms of infection, thrombosis, chest pain, neurologic deficits
Symptoms of Infection or thrombosis
Symptoms of infection or respiratory dysfunction, changes in vision
Avoid live vaccines and use with strong CYP 3A4 inducers or inhibitors. Avoid with azathioprine or cyclosporine
Avoid live vaccines. Screen for baseline TB. Do not initiate in patients with lymphocytes < 500/mm3(0.5 ×
109/L), ANC
<1000/mm3(1 ×
109/L), or hemoglobin <9
g/dL (90 g/L; 5.59 mmol/L). Monitor lipids and LFTs every 4-8 weeks. Gastrointestinal perforation has been reported with use of the XR formulation. Drug interactions with CYP3A4 and 2C19
inhibitor
Need baseline ECG, WBC, LFTs,
ophthalmic assessment, and testing for varicella zoster antibodies. Contraindicated if patient has experienced MI, unstable angina, stroke, TIA, decompensated
| Drug(s) | Adverse Drug Reaction | Monitoring Parameters | Comments |
heart failure, or Mobitz type II second- or third- degree AV block, sick sinus syndrome in the last 6 months, presence of sleep apnea, and concomitant use of MAOI.
Tips for Pharmacy Technicians
Pharmacy technicians are important members of the health care team. Oftentimes, patients may come to the pharmacy seeking treatment for GI- related symptoms, and pharmacy technicians can assist pharmacists in identifying patients who may have IBD or be candidates for referral. Pharmacy technicians can also recognize when patients may not adhere to IBD therapies and assist pharmacists in recognizing and helping these patients. Additionally, pharmacy technicians have served on clinical care teams, where they assist with submitting prior authorizations to insurance companies, following up on appeals, and determining if patients are eligible for financial assistance.41
Summary
Ulcerative colitis is one of two gastrointestinal disorders that encompass IBD. Treatment of the disease can involve various pharmacologic therapies. These agents should be selected based on disease activity and risk of disease progression, considering adverse effects and costs to patients. Pharmacists are vital to the care team and can use their medication expertise to guide treatment decisions and counsel patients.
Course Test
Which of the following factors has a protective effect related to the development of ulcerative colitis in Caucasians?
Smoking
Smoking cessation
Breastfeeding cessation
Low vitamin D levels
Which of the following pathologic features indicate ulcerative colitis more than Crohn’s disease?
Fistulas
Ileal involvement
Crypt abscesses
Cobblestone appearance
Which of the following is true regarding the pathophysiology of ulcerative colitis?
Ulcerative colitis can occur in any part of the gastrointestinal tract
Ulcerative colitis is typically confined to the colon and rectum, affecting mucosal and submucosal layers
Ulcerative colitis is often mild and not progressive in nature
Unlike Crohn’s disease, ulcerative colitis is not associated with extraintestinal complications
Which of the following medications are available in topical and oral formulations and are used for induction treatment of mild- moderate ulcerative colitis?
Mesalamine (5-ASA)
Methylprednisolone
Infliximab
Vedolizumab
Vedolizumab should not be used in combination with which of the following?
Mesalamine
TNF-alpha inhibitors
Sulfasalazine
Corticosteroids
A patient who may receive azathioprine therapy should have which of the following tests completed prior to initiating therapy?
PML test
PPD
TPMT
TGN
If a patient takes ketoconazole (a major inducer of CYP3A4), which ulcerative colitis medications should the patient avoid?
Upadacitinib
Vedolizumab
Sulfasalazine
Ustekinumab
Which of the following anti-TNF agents is a human monoclonal antibody that binds to human tumor necrosis factor-alpha, thereby interfering with endogenous TNFα activity?
Infliximab
Adalimumab
Certolizumab
Golimumab
Patients should be counseled to do the following before starting anti-TNF therapy.
Receive the live, attenuated influenza vaccine
Be treated for tuberculosis
Receive the pneumococcal pneumonia vaccine
Receive treatment for viral hepatitis
Which of the following is an appropriate counseling point pharmacists can provide to patients with Crohn’s disease?
The majority of medications used to treat ulcerative have an onset of action of 16-20 weeks
Numerous vaccinations are required prior to starting azathioprine therapy
Being nonadherent to medication therapies can increase the risk of relapse
Most cases of ulcerative colitis are limited in nature, and medications are often only needed for 2-4 weeks
References
Rubin DT, Ananthakrishnan AN, Siegel CA, Sauer BG, Long MD. ACG Clinical Guideline: Ulcerative Colitis in Adults. Am J Gastroenterol. 2019;114(3):384-413. doi:10.14309/ajg.0000000000000152
Ungaro R, Mehandru S, Allen PB, Peyrin-Biroulet L, Colombel JF. Ulcerative colitis. Lancet. 2017;389(10080):1756-1770. doi:10.1016/S0140-6736(16)32126-2
Fumery M, Singh S, Dulai PS, Gower-Rousseau C, Peyrin-Biroulet L, Sandborn WJ. Natural History of Adult Ulcerative Colitis in Population- based Cohorts: A Systematic Review. Clin Gastroenterol Hepatol. 2018;16(3):343-356.e3. doi:10.1016/j.cgh.2017.06.016
Lungaro L, Costanzini A, Manza F, et al. Impact of Female Gender in Inflammatory Bowel Diseases: A Narrative Review. J Pers Med. 2023;13(2):165. Published 2023 Jan 17. doi:10.3390/jpm13020165
Beaugerie L, Massot N, Carbonnel F, Cattan S, Gendre JP, Cosnes J. Impact of cessation of smoking on the course of ulcerative colitis. Am J Gastroenterol. 2001;96(7):2113-2116. doi:10.1111/j.1572- 0241.2001.03944.x
Takeuchi K, Smale S, Premchand P, et al. Prevalence and mechanism of nonsteroidal anti-inflammatory drug-induced clinical relapse in patients with inflammatory bowel disease. Clin Gastroenterol Hepatol. 2006;4(2):196-202. doi:10.1016/s1542-3565(05)00980-8
Evans JM, McMahon AD, Murray FE, McDevitt DG, MacDonald TM. Non- steroidal anti-inflammatory drugs are associated with emergency admission to hospital for colitis due to inflammatory bowel disease. Gut. 1997;40(5):619-622. doi:10.1136/gut.40.5.619
Singh S, Graff LA, Bernstein CN. Do NSAIDs, antibiotics, infections, or stress trigger flares in IBD?. Am J Gastroenterol. 2009;104(5):1298- 1314. doi:10.1038/ajg.2009.15
Hemstreet BA. Inflammatory Bowel Disease. In: DiPiro JT, Yee GC, Haines ST, Nolin TD, Ellingrod VL, Posey L. eds. DiPiro’s Pharmacotherapy: A Pathophysiologic Approach, 12th Edition. McGraw Hill; 2023.
Gros B, Kaplan GG. Ulcerative Colitis in Adults: A Review. JAMA. 2023;330(10):951-965. doi:10.1001/jama.2023.15389
Kaplan GG, Ng SC. Understanding and Preventing the Global Increase of Inflammatory Bowel Disease [published correction appears in Gastroenterology. 2017 Jun;152(8):2084]. Gastroenterology. 2017;152(2):313-321.e2. doi:10.1053/j.gastro.2016.10.020
Vedamurthy A, Ananthakrishnan AN. Influence of Environmental Factors in the Development and Outcomes of Inflammatory Bowel
Disease. Gastroenterol Hepatol (N Y). 2019;15(2):72-82.
Desai J, Elnaggar M, Hanfy AA, Doshi R. Toxic Megacolon: Background, Pathophysiology, Management Challenges and Solutions [published correction appears in Clin Exp Gastroenterol. 2021 Jul 19;14:309- 310]. Clin Exp Gastroenterol. 2020;13:203-210. Published 2020 May
19. doi:10.2147/CEG.S200760
Limdi JK, Farraye FA. An Update on Surveillance in Ulcerative
Colitis. Curr Gastroenterol Rep. 2018;20(2):7. Published 2018 Mar 7. doi:10.1007/s11894-018-0612-2
Satsangi J, Silverberg MS, Vermeire S, Colombel JF. The Montreal classification of inflammatory bowel disease: controversies, consensus, and implications. Gut. 2006;55(6):749-753. doi:10.1136/gut.2005.082909
Roda G, Narula N, Pinotti R, et al. Systematic review with meta- analysis: proximal disease extension in limited ulcerative colitis. Aliment Pharmacol Ther. 2017;45(12):1481-1492. doi:10.1111/apt.14063
Sturm A, Maaser C, Calabrese E, et al. ECCO-ESGAR Guideline for Diagnostic Assessment in IBD Part 2: IBD scores and general principles and technical aspects. J Crohns Colitis. 2019;13(3):273-284. doi:10.1093/ecco-jcc/jjy114
Ko CW, Singh S, Feuerstein JD, et al. AGA Clinical Practice Guidelines on the Management of Mild-to-Moderate Ulcerative
Colitis. Gastroenterology. 2019;156(3):748-764. doi:10.1053/j.gastro.2018.12.009
Colombel JF, Narula N, Peyrin-Biroulet L. Management Strategies to Improve Outcomes of Patients With Inflammatory Bowel
Diseases. Gastroenterology. 2017;152(2):351-361.e5. doi:10.1053/j.gastro.2016.09.046
Derwa Y, Gracie DJ, Hamlin PJ, Ford AC. Systematic review with meta- analysis: the efficacy of probiotics in inflammatory bowel
disease. Aliment Pharmacol Ther. 2017;46(4):389-400. doi:10.1111/apt.14203
Goodhand JR, Wahed M, Mawdsley JE, Farmer AD, Aziz Q, Rampton DS. Mood disorders in inflammatory bowel disease: relation to diagnosis, disease activity, perceived stress, and other factors. Inflamm Bowel Dis. 2012;18(12):2301-2309. doi:10.1002/ibd.22916
Ananthakrishnan AN, Gainer VS, Perez RG, et al. Psychiatric co- morbidity is associated with increased risk of surgery in Crohn's disease. Aliment Pharmacol Ther. 2013;37(4):445-454. doi:10.1111/apt.12195
Lialda (Mesalamine). Package Insert. Shire US, Inc. 2007.
Azulfadine. (Sulfasalazine). Package Insert. Pfizer. 2009.
Imuran (Azathioprine). Package Insert. Pharmaceuticals International.
Neoral (cyclosporine) [prescribing information]. East Hanover, NJ: Novartis; Jan 2024.
Remicade (Infliximab). Package Insert. Janssen Biotech. 2013.
Remicade (Infliximab). Package Insert. Janssen Biotech. 2013.
Simponi (golimumab) [prescribing information]. Horsham, PA: Janssen Biotech Inc; September 2019.
Beaugerie L, Rahier JF, Kirchgesner J. Predicting, Preventing, and Managing Treatment-Related Complications in Patients With Inflammatory Bowel Diseases. Clin Gastroenterol Hepatol. 2020;18(6):1324-1335.e2. doi:10.1016/j.cgh.2020.02.009
Entyvio (Vedolizumab). Package insert. 2022.
Xeljanz (Tofacitinib). Package insert. Pfizer.
RINVOQ (Upadacitinib). Package insert. 2023.
Zeposia (Ozanimod). Package insert. Bristol Myer Squibbs.
Ford AC, Achkar JP, Khan KJ, et al. Efficacy of 5-aminosalicylates in ulcerative colitis: systematic review and meta-analysis. Am J Gastroenterol. 2011;106(4):601-616. doi:10.1038/ajg.2011.67
Feagan BG, Chande N, MacDonald JK. Are there any differences in the efficacy and safety of different formulations of Oral 5-ASA used for induction and maintenance of remission in ulcerative colitis? evidence from cochrane reviews. Inflamm Bowel Dis. 2013;19(9):2031-2040. doi:10.1097/MIB.0b013e3182920108
Entocort (Budesonide). Package Insert. Perrigo. 2016.
Kane SV, Cohen RD, Aikens JE, Hanauer SB. Prevalence of nonadherence with maintenance mesalamine in quiescent ulcerative colitis. Am J Gastroenterol. 2001;96(10):2929-2933. doi:10.1111/j.1572-0241.2001.04683.x
Feagan BG, Macdonald JK. Oral 5-aminosalicylic acid for induction of remission in ulcerative colitis. Cochrane Database Syst Rev. 2012;10:CD000543. Published 2012 Oct 17. doi:10.1002/14651858.CD000543.pub3
Martins CA, Garcia KS, Queiroz NSF. Multi-utility of therapeutic drug monitoring in inflammatory bowel diseases. Front Med (Lausanne). 2022;9:864888. Published 2022 Jul 28. doi:10.3389/fmed.2022.864888
Choi DK, Rubin DT, Puangampai A, Lach M. Role and Impact of a Clinical Pharmacy Team at an Inflammatory Bowel Disease
Center. Crohns Colitis 360. 2023;5(2):otad018. Published 2023 Apr 15. doi:10.1093/crocol/otad018
DISCLAIMER
The information provided in this course is general in nature, and it is solely designed to provide participants with continuing education credit(s). This course and materials are not meant to substitute for the independent, professional judgment of any participant regarding that participant’s professional practice, including but not limited to patient assessment, diagnosis, treatment, and/or health management. Medical and pharmacy practices, rules, and laws vary
from state to state, and this course does not cover the laws of each state; therefore, participants must consult the laws of their state as they relate to their professional practice.
Healthcare professionals, including pharmacists and pharmacy technicians, must consult with their employer, healthcare facility, hospital, or other organization for guidelines, protocols, and procedures they are to follow. The information provided in this course does not replace those guidelines, protocols, and procedures but is for academic purposes only, and this course’s limited purpose is for the completion of continuing education credits.
Participants are advised and acknowledge that information related to medications, their administration, dosing, contraindications, adverse reactions, interactions, warnings, precautions, or accepted uses are constantly changing, and any person taking this course understands that such person must make an independent review of medication information prior to any patient assessment, diagnosis, treatment and/or health management. Any discussion of off-label use of any medication, device, or procedure is informational only, and such uses are not endorsed hereby.
Nothing contained in this course represents the opinions, views, judgments, or conclusions of RxCe.com LLC. RxCe.com LLC is not liable or responsible to any person for any inaccuracy, error, or omission with respect to this course or course material.
© RxCe.com LLC 2024: All rights reserved. No reproduction of all or part of any content herein is allowed without the prior, written permission of RxCe.com LLC.
