THE HANDLING OF HAZARDOUS DRUGS
Steve Malen, PharmD, MBA
Dr. Steve Malen graduated with a dual degree: Doctor of Pharmacy (PharmD) and Master of Business Administration (MBA) from the University of Rhode Island. Over his career, he has worked as a clinical pharmacist in the retail, specialty, and compounding sectors. He specialized and taught on topics from vaccines to veterinary compounding. Dr. Malen has also written a science fiction novel and taught and co-founded the concept of Patient Empowered Blockchain (P.E.B.). Currently, Dr. Malen continues to write, teach, and consult various companies in the healthcare sector.
Topic Overview
Drugs used to treat cancer, some antiviral drugs, hormone agents, and bioengineered drugs, are dangerous to pharmacy staff and other healthcare workers because of their toxicity. When clinicians are exposed to these drugs in the course of their occupations, they are at risk for a range of toxic effects, including cancer, impairment of reproduction, and genetic mutations. These drugs are now described as “hazardous drugs,” which replaced their prior designation as cytotoxic or antineoplastic drugs. The United States Pharmacopeia (USP), Chapter 800, is the latest regulation regarding the handling of HDs in the healthcare setting in order to “promote patient safety, worker safety, and environmental protection.”
Accreditation Statement:

RxCe.com LLC is accredited by the Accreditation Council for Pharmacy Education (ACPE) as a provider of continuing pharmacy education.
Universal Activity Number (UAN): The ACPE Universal Activity Number assigned to this activity is
Pharmacist 0669-0000-22-076-H07-P
Pharmacy Technician 0669-0000-22-077-H07-T
Credits: 2 hours of continuing education credit
Type of Activity: Knowledge
Media: Internet Fee Information: $6.99
Estimated time to complete activity: 2 hours, including Course Test and course evaluation
Release Date: December 10, 2022 Expiration Date: December 10, 2025
Target Audience: This educational activity is for pharmacists/pharmacy technicians.
How to Earn Credit: From December 10, 2022, through December 10, 2025, participants must:
Read the “learning objectives” and “author and planning team disclosures;”
Study the section entitled “educational activity;” and
Complete the Course Test and Evaluation form. The Course Test will be graded automatically. Following successful completion of the Course Test with a score of 70% or higher, a statement of participation will be made available immediately. (No partial credit will be given.)
Learning Objectives: Upon completion of this educational activity, participants should be able to:
Review the latest regulations relating to the handling of hazardous drugs.
Identify the importance of regulations and standards in handling hazardous drugs for the safety of patients, pharmacy staff, and the environment.
Describe the complexity of the hazardous drug regulations for staff working in a hazardous compounding pharmacy or looking to start one.
Describe the procedures to follow in the event of exposure to hazardous drugs.
Disclosures
The following individuals were involved in the development of this activity: Susan DePasquale, MSN, PMHNP-BC, and Steve Malen, PharmD. There are no financial relationships relevant to this activity to report or disclose by any of the individuals involved in the development of this activity.
ⓒ RxCe.com LLC 2022: All rights reserved. No reproduction of all or part of any content herein is allowed without the prior, written permission of RxCe.com LLC.
Introduction
Drugs used to treat cancer, some antiviral drugs, hormone agents, and bioengineered drugs, are dangerous to pharmacists and other healthcare workers because of their toxicity. Exposure to these hazardous drugs in the course of professional practice can injure the healthcare worker. Proper precautions and care are necessary to prevent these occupational injuries. Pharmacists and pharmacy staff can appreciate the importance of handling hazardous drugs properly through a brief review of the history of hazardous drugs and the development of guidelines for their proper handling.
History of Hazardous Drugs
In the 1930s, nitrogen mustard was developed for the treatment of lymphoma.1 Other antineoplastic drugs followed in the 1940s, and their toxicity has been acknowledged since.2 When pharmacists and other healthcare workers were exposed to these drugs in the course of their occupations, they were at risk for a range of toxic effects, including cancer, impairment of reproduction, and genetic mutations.1,3,4
These hazards led to the U.S. National Institute of Occupational Safety and Health (NIOSH), conducting a survey, in the early 1980s, of commercial chemical products in a number of industries, including healthcare.5 The survey sought “to develop estimates of the number of U.S. workers potentially exposed to chemical, physical, and biological agents in selected industrial sectors, and, obtain data describing the nature and extent of these potential exposures and the degree to which businesses have implemented programs to reduce health problems.”5 The results of this survey were published around 1983 as the National Occupational Exposure Survey (NOES).5 In 1990, the American Society of Hospital Pharmacists, now the American Society of Health-System Pharmacists (ASHP), introduced the term “hazardous drugs.”4,6,7 The term “hazardous drugs” is now used to describe these dangerous drugs. This term replaced their prior designation as cytotoxic or antineoplastic drugs.6 The United States Pharmacopeia (USP) publishes the guidelines for the handling of HDs.
The Occupational Safety and Health Administration (OSHA) published guidelines in 1986. Thereafter, OSHA produced updates in 1996, expanding the recommendations to include all “hazardous” drugs, and in 1999, OSHA added recommendations that included testing, personal protective equipment, ventilated cabinets, closed system transfer devices (CSTDs), and needleless devices.4 OSHA currently provides guidance to employers in the healthcare industry.4,8,9
The NIOSH studied the literature published on the dangers of hazardous drugs and provided recommendations for their proper handling in 2004.4,10 The NIOSH adopted the term “hazardous drugs” first used by the ASHP in this 2004 publication.7,10
The recommendations did not create mandatory standards. The NIOSH has updated the HD list periodically between 2010 and today.4 In 2008, USP 797, applicable to sterile compounding, included recommendations for HD preparation due to conflicts with NIOSH; however, this only pertained to HDs used in the context of sterile compounding.11
The ASHP released its most recent revision to its guidelines in 2006.4,12 The Oncology Nursing Society began publishing its guidelines in 1984, with periodic updates thereafter.4
Despite these regulations and recommendations from various organizations, harm to healthcare workers in the preparation of HDs continued to occur systematically, which led to multiple investigations.4 This evidence of harm to pharmacy staff came to a climax in 2010 with the release of two studies showing continued harm to pharmacy staff, including cancer.4 At this same time, an investigative report on the story of Sue Crump, who was a pharmacist in Seattle, was published. Sue Crump died of pancreatic cancer at the age of 55.4 Sue Crump, had been diagnosed with cancer after working as a pharmacist for many years. She used some protection when handling HDs, but it was minimal and inadequate. Even after Sue Crump’s story came out, nurses continued to handle HDs while disregarding national guidelines. As a
result of these events, Washington State passed first-of-its-kind legislation, requiring HD standards.4 Soon thereafter, USP 800 was created.13
The USP 800 took the previous recommendations and developed from them a federal “minimum standard” for handling HDs.14 USP 800 was officially published in 2016 and created guidance on the handling of HDs in the healthcare setting, including sterile and nonsterile compounding.14
The NIOSH published an updated list of HDs in 2020.15 This updated list is not final but is a draft awaiting comment from various experts and stakeholders. This new list seeks to incorporate more recent studies into the proper handling of HDs. This list added 16 drugs and removed five drugs from the original list. This update also added a carcinogen section and altered the organization of the information to be more intuitive.15
Importance of HDs to Pharmacy Staff
Some consider pharmacists and pharmacy staff to be “the clear choice to take on the challenge of evaluating hazardous risk of drugs and implementing appropriate procedures to manage those risks.”1 However, this challenge comes with a significant burden on pharmacy resources: “the task of every entity independently evaluating every dosage for each of the hundreds of drugs on the NIOSH list.”1 Pharmacists and pharmacy staff will likely play a major role in developing the best practices in this area.1
Healthcare workers who prepare or administer hazardous drugs (e.g., those used for cancer therapy, and some antiviral drugs, hormone agents, and bioengineered drugs), are at risk for a range of toxic effects, including, but not limited to, adverse reproductive outcomes.1,4
The handling of HD is a serious topic that uniquely affects the health of healthcare workers, and pharmacy staff, in particular. This training could be the difference between a safe work environment, or injury, including serious injury or death, for pharmacy staff. Pharmacy technicians are a particular risk since they are the ones working with hazardous substances the most. They
are the ones receiving products from distributors, preparing the compounds, and packaging them for the patient.16,17
USP 800
As stated above, USP 800 is the latest regulation regarding the handling of HDs in the healthcare setting. Its purpose is “promote patient safety, worker safety, and environmental protection.”18 This regulation is enforced by several agencies, including the FDA, OSHA, and the boards of pharmacies. Unfortunately, every state may regulate USP 800 in its preferred way, given the lack of specific guidance. This can range from not requiring it to requiring it for all pharmacies that handle HDs regardless of the compound. From a legal perspective, check with your board of pharmacy regarding their enforcement of USP 800; however, best practice protocols require pharmacy staff to be familiar with USP 800 and to implement whatever parts of it are applicable to their practice. As stated before, this will ensure patient safety, the protection of staff, and the environment. If a person works in a compounding pharmacy, that person should consult his or her compounding distributor, as the compounding distributor often has information and guidance to provide to staff.
The list of HDs is maintained by NIOSH, and the categorization is based on the type of drug, dosage form, risk of exposure, packaging, and manipulation. Exposure to HDs can be via the skin, mucosal, inhalation, injection, and ingestion. These exposures can happen during receipt of the drug by the pharmacy or patient, during dispensing by the pharmacy, during compounding or manipulation, during administration to a patient, via spill, transport, or waste. Even the outside of containers of HDs can be contaminated, which is why it is important for non-compounding pharmacies to receive HDs safely. It is required for each pharmacy to have someone responsible for creating and implementing procedures that adhere to USP 800.
Facilities and Engineering Controls
Wherever HDs are handled, it is important to put up signs at the entrance and restrict access to the area. It is important for the HDs to be located away “from breakrooms and refreshment areas for personnel, patients, or visitors.”19 This designated area is for the purpose of receiving, unpacking, and storing compounding products. In certain cases, especially with compounding, it is important to have negative pressure to prevent hazardous vapors from traveling to a non-hazardous area. It is recommended to have a generator connected to the ventilation system to make sure ventilation is maintained in case of power loss.
Receipt
Must be neutral or negative pressure to surrounding areas but not in sterile compounding areas.
Storage
Hazardous drugs must be stored to prevent spillage or breakage if the container falls and not on the floor. If a pharmacy is in a part of the country with a higher chance of natural disasters, extra precautions must be taken. Refrigerated antineoplastic HDs must be stored in an HD refrigerator in a negative pressure area such as a storage room, buffer room, or a containment-segregated compounding area (C-SCA).19,20 In the case of a buffer room then, the exhaust from the refrigerator needs to be added.
Compounding
When compounding HDs, the first thing to consider is a containment primary engineering control (C-PEC) which is basically a hood with ventilation.
The second thing is a containment secondary engineering control (C- SEC) which is a room where the hood is placed. Finally, a closed-system drug- transfer device (CSTD), in some cases, must be used.20 The Centers for
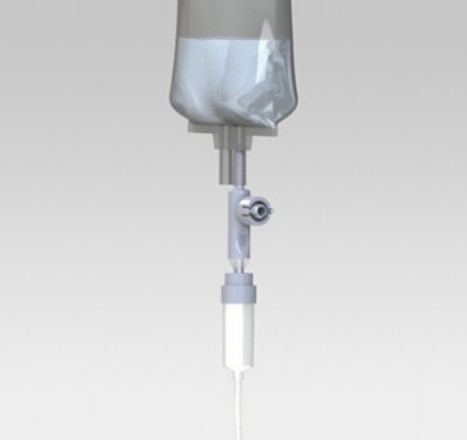
Disease Control and Prevention (CDC) displays the following CSTD on its website:21
7
The C-SEC or compounding area must be “externally vented, be physically separated, have an appropriate air exchange, have a negative pressure between 0.01 and 0.03 inches of water column relative to all adjacent areas.”18 Negative pressure gauges are used to measure negative pressure in the compounding area.
The C-PEC or hood must constantly ventilate while working. If it stops for any reason, all work must stop, it must be covered, and after it is back to working condition, all surfaces must be cleaned, and work can only start per the hood “recovery time.” A sink for hand washing and an eyewash station must be available.18
For sterile compounding, the C-PEC or hood needs to be in a separate room; however, for non-sterile compounding, this can be avoided if the room “can continuously maintain ISO 7 classification,” which means the room uses HEPA filtration.18 This filtration “maintains air cleanliness levels of a maximum of 10,000 particles per cubic foot.”18 If the non-sterile compounding hood and sterile compounding hood are placed in the same room, they need to be placed at least 1 meter apart, and particle-generating activity in the non-sterile hood cannot be performed at the same time as sterile compounding.
Nonsterile Compounding
USP 795 must be followed in addition to USP 800. A hood or C-PEC is required if, during compounding, “particles, aerosols or gasses” are produced, which is most compounding. Nonsterile compounding hoods are different from sterile compounding hoods, although nonsterile compounding may be performed in a sterile compounding hood; however, it must be decontaminated after each use. The surfaces of the room surrounding the nonsterile compounding hood must be “smooth, impervious, free from cracks and crevices, and non-shedding.”18
Sterile Compounding
In addition to USP 800, USP 797 must be followed. The hood must be externally vented and maintained at ISO Class 5 or better. Different types of sterile compounding hoods depend on the volatility of the compounds. It also gets complicated with the use of a laminar airflow workbench (LAFW) or a compounding aseptic isolator (CAI) which depends on the type of HD being compounded. It is very important to evaluate the compounds you create and make sure they are being used with all the necessary devices and under the right conditions.
ISO Class 7 buffer room with ISO class 7 ante-room
The sterile compounding hood needs to be inside the buffer room. The ante-room must have a hand-washing sink. It is not recommended for the
negative-pressure buffer room to be connected to the positive-pressure non- HD buffer room. Beyond-use dates (BUDs) are calculated based on specifications in USP 797. Practically, compounds are prepared using formulas that have BUDs assuming certain compounding conditions, and the recipes are followed perfectly. C-SCA or containment segregated compounding area is allowed only for low & medium-risk HDs and must have a hand-washing sink at least 1 meter from the hood.
Environmental Quality and Control
At least every 6 months, it is required to collect wipe samples to test for surface HDs. There are HD wipe test kits available for purchase, and if any of these tests show a positive result, they must be documented and investigated to figure out the cause. After mitigating the problem, the wipe sampling needs to be repeated. This is why it is important to have strict and specific procedures for decontaminating every surface where HDs are compounded, including walls, ceilings, and floors.
Personal Protective Equipment
Personal Protective Equipment (PPE) is used to protect the worker, patient, and caregiver. Disposable PPE should never be reused, and reusable PPE must be decontaminated after each use. When compounding HDs, it is required to use gowns, head/hair/shoe covers, and two pairs of chemotherapy gloves. When administering antineoplastic HDs, it is required to wear two pairs of chemotherapy gloves, and if they are injectable, it is also required to wear a gown. It is required to have a detailed SOP specific to PPE for every activity involving HDs, from receipt to waste disposal.18,22
Gloves
Chemotherapy gloves must be American Society for Testing and Materials (ASTM) standard D6978 or its successor and should be worn for handling all HDs. For sterile compounding, the outer glove must be sterile and should be changed every 30 minutes or when torn, punctured, or
contaminated. After using gloves, it is required to wash your hands with soap and water for your protection.
Gowns
Gowns must be disposable and resist permeability to specific HDs, as there is a variety of gowns available. Each gown has specific instructions on how often to change; however, if they do not change every 2-3 hours or after contamination. Gowns must only be worn in HD handling areas to prevent transmission.
Head, Hair, Shoe, and Sleeve Covers
If the worker has a beard or mustache, it is important to have extra protection. When entering and exiting the C-SEC, i.e., the compounding area, it is required to put on a second pair of shoe covers.
Eye and Face Protection
The most comprehensive protection comes from a full-facepiece respirator. For eye protection, goggles may be worn as eyeglasses or safety glasses, even with side shields that do not offer enough protection. Face shields may be combined with goggles to offer extra protection; however, face shields cannot be worn without the goggles.
Respiratory Protection
When unpacking HDs without a plastic container, the person must wear “an elastomeric half-mask with a multi-gas cartridge and P100-filter.”18 A more drug-specific cartridge can be used based on the HDs being unpacked. If respiratory protection is needed, do not use surgical masks. A fit-tested N95 or better respirator can be used as long as there is no risk from gases, vapors, or “direct liquid splashes.” For more specific instructions, follow the OSHA respiratory protection standard. It is important to have a “full-facepiece, chemical cartridge-type respirator or powered air-purifying respirator (PAPR) for a spill that is not contained with spill kit, for any cleaning underneath the
“work surface of a C-PEC” or if there is potential airborne exposure to powders or vapors.
Disposal of Used PPE
If any PPE is worn while handling HDs, assume it has been contaminated and placed in a waste container, and if worn during compounding must be disposed of prior to leaving the C-SEC. Chemotherapy gloves must be discarded inside the C-PEC or placed in a bag for discarding outside the C- PEC.
Hazard Communication Program
Every pharmacy must train its staff on its Standard Operating Procedures (SOPs) for handling HDs. These SOPs are usually available from the compounding distributors; however, a pharmacy must make sure they are specific to the pharmacy’s compounding activity. It is also important to have the following:
An implementation plan
Proper labeling of all HD chemicals
Safety Data Sheets (SDS) are available and accessible for each hazardous chemical
Training for all workers handing HDs before any work with HDs and whenever hazard changes
Written confirmation from workers of reproductive capability regarding understanding risks
Personnel Training
All training must be specific to work performed by the worker in regard to the handling of HDs and before any work has been done. This must be repeated every 12 months, whenever a new HD or equipment is added or if there is a change to the SOP. It is required to document all training and competency assessments. Training must include the following:
Overview of HDs and risks
Review of SOPs relating to HDs
PPE, equipment, and device use
HD exposure response protocol, including spill management
Disposal of HDs
Receiving
Receipt of HDs must be based on the SOP for the pharmacy. Immediately after unpacking, HDs should be brought to the HD storage area. Chemotherapy gloves, in addition to other appropriate PPE, should be worn, and a spill kit must be accessible during the unpacking of HDs. If the HD package is damaged, contact the supplier immediately.
Labeling, Packaging, Transport, and Disposal
SOPs must be created and address prevention/training for accidental exposure and the use of a spill kit. In order to prevent exposure, there are various controls such as a Luer Lock syringe, CSTDs, “impervious plastic bags” and cautionary labeling.
Dispensing Final Dosage Forms
If an HD does not require “any further manipulation,” then HD precautions do not need to be taken. For instance, if a pharmacy prepares enclosed capsules or injectable syringes, they can be counted or repackaged in the same location as non-HD products unless the product looks damaged. Counting or repacking of final HDs must be done carefully, and all equipment used should be decontaminated immediately. Tablets and capsules HDs should be counted with any “automated counting or packaging machines.”
Compounding
“A plastic-backed preparation mat” should always be used during compounding of HDs and should be changed immediately after a spill,
regularly during use, and at the end of the day. Any equipment used with HDs must be exclusive to HDs.
Deactivating, Decontaminating, Cleaning and Disinfecting
By the end of every day, these activities must be performed, and sterile compounding areas and devices “must be subsequently disinfected.”18 Every pharmacy must have an SOP for this process which complies with USP 795 (if nonsterile) and USP 797 (if sterile). The SOP must have “procedures, agents used, dilutions (if used), frequency and documentation requirements.”18 The staff who perform these duties must be trained to protect themselves and the environment. PPE must be worn to protect the worker against the cleaning agents, including “two pairs of chemotherapy gloves and impermeable disposable gowns.”18 If splashing is possible, then “eye protection and face shields” are also required. Finally, if there is a chance of gas or powder exposure, then “respiratory protection” must also be used.18 Not all cleaning agents can be used with all surface materials, so make sure all the cleaning agents that are used are compatible with all the surface areas. For instance, your walls/ceilings/floors will most likely be cleaned with one product, while the hood and counters could be used with a different, most likely stronger product. It is also important not to directly spray cleaning agents but rather use wipes with a solution to prevent HD residue from spreading. When finished cleaning, all materials being thrown away must be done in a manner meeting EPA regulations as well as the SOP of the pharmacy.18
Deactivation
This process makes HDs inactive, and while there is “no proven method” there are “EPA-registered oxidizing agents” which are preferred. This process must be done carefully to prevent harm to workers but also to the surface being used, as even stainless-steel surfaces could be damaged. To prevent corrosion due to sodium hypochlorite, sodium thiosulfate or other agents can be used. Grade 304 stainless steel is popular as it is corrosion resistant.12
Decontamination
This process includes moving HD residue from surfaces to “wipes, pads, or towels” for disposal.18 It is imperative to make sure all the products are compatible with the surfaces being cleaned,23,24 which can be found in the manufacturer’s instructions. Recently, there have been many more products available for testing surfaces so that this protocol may be performed more often, and in line with the SOP of the pharmacy. HD containers often have heavy residue, so make sure to wipe that down as often as possible to prevent spread to other surfaces while not damaging the product label. In between compounding different HDs, make sure to clean the work surface, and the entire C-PEC must be “decontaminated at least daily (when used), any time a spill occurs, before and after certification, any time voluntary interruption occurs, and if the ventilation tool is moved.” Once monthly, it is also required to clean the area under the work tray in the C-PEC. While doing this, it is important to wear respiratory protection as the ventilation must be turned off.
Cleaning
This is the process of removing “contaminants” from “objects and surfaces” using various liquid cleaning agents.
Disinfection
This process serves to inhibit or destroy microorganisms and is vital for sterile compounding.
Spill Control
Training is required for all personnel, usually all employees, responsible for spill control. Spill kits need to have everything necessary, including signs to restrict access. Once a spill occurs, it is imperative to clean it immediately and document all details. If it is possible for any exposure to a human
(employee or non-employee) to occur, they must seek medical evaluation immediately, and everything must be documented.
Documentation and Standard Operating Procedures (SOPs): SOPs are mandatory for every situation where there is a possibility of exposure to HDs and must be reviewed/documented every 12 months. The following SOPs are mandatory (directly from USP 800):
Hazard communication program
Occupational safety program
Designation of HD areas
Receipt
Storage
Compounding
Use and maintenance of proper engineering controls (e.g., C-PECs, C- SECs, and CSTDs)
Hand hygiene and use of PPE based on activity (e.g., receipt, transport, compounding, administration, spill, and disposal)
Deactivation, decontamination, cleaning, and disinfection
Dispensing
Transport
Administering
Environmental monitoring (e.g., wipe sampling)
Disposal
Spill control
Medical surveillance
Medical Surveillance
In addition to SOPs, which minimize exposure to HDs, medical surveillance is an attempt to double-check that all the processes are continuing to work. Unlike SOPs, which are required, medical surveillance is recommended; however, they should not be treated lightly. For many organizations, this may be very difficult and expensive to do, so it is possible to contract out, and it is even one of the recommendations in USP 800. In
short, personnel who handle HDs are tracked medically, and their health outcomes and laboratory values are compared to the regular population. That way, if there was a problem with the current activities, then it could be caught. This is similar to using the wipe kits occasionally to test surfaces in the HD lab areas, except this is with the personnel. The goal of these continuous tests of personnel is for them to show no anomalies in their tests which would mean that the pharmacy is running a “tight ship” and personnel are not getting exposed to HDs. It is important from the beginning to get the baseline values of personnel to make clear that if anomalies do occur, they are from work. Medical surveillance programs must align with Human Resource policies and must include a standard process for figuring out which workers can be exposed to HDs.
It is important to get an “initial baseline assessment” of every worker’s health to get a better idea of the causal relationship between the hazards of the work environment and the health of the worker. If this is not taken into account, this could either mean false positives or false negatives in the surveillance program since your worker pool could be healthier or sicker than the average population. Then it is important to get a total hazard potential exposure calculation by the number of records and hours for each employee. Finally, based on these calculations, each employee should get a medical workup that is targeted toward the organs that could be affected. For instance, if an employee works with HD that causes harm to the liver, then that employee should get regular checks for liver damage. This may seem like a daunting and time-consuming task which is why it is not required, and you can use an outside company that has all the systems in place. Fortunately, blood or urine testing for HDs is not recommended for medical surveillance; however, it can be part of the SOP for spills.
It is crucial to make sure the medical records of all the employees in the surveillance program are protected and accessible only to those who need them to run the program. This is another reason to use an outside company, as this would allow for all the data to be assessed by non-employees of the company; that being said, an outside company also needs to keep that data confidential and per OSHA standards.
Continuous monitoring of the employee’s health as it relates to the potential exposure to HDs is crucial for the success of the program. The simultaneous continuous connection between the exposure to health data is vital to identify SOP prevention failure. Again, this is an involved process, so the use of an external company to do it would be advised. If any employee shows health changes, it is important to quickly assess whether it is possible the harm could have been caused by exposure to HDs they are working with, in particular, if there was an acute exposure or a spill to that particular HD. Additionally, it would be prudent to evaluate the SOPs and other controls in place in regard to applicable HDs. Finally, whenever an employee leaves the entity, it is important to conduct an “exit examination” for “medical, reproductive, and exposure histories.” This should be targeted based on the HDs potential exposed to each employee. Medical surveillance sounds like a particularly daunting task however it is important for proactive monitoring of exposure to HDs by the employees handling HDs. Given the potential risk, it is also good protection for the entity against frivolous claims. For instance, an employee of the entity may have a health problem after working that has nothing to do with exposure to HDs at work. A comprehensive surveillance program would be a way to show that it is not possible that an employee had a certain health problem from work given the medications they worked with. Regardless, surveillance programs are optional and most likely would only be possible in significantly large companies with enough budget for this kind of work. If you work at a small compounding pharmacy, it would be quite unaffordable to perform this work, whether in-house or contracting with an outside company.
Follow-Up Plan
Every time there is an exposure of HDs, there should be an immediate assessment of the SOPs and any controls in place. The assessment should be based on the exact type of exposure (i.e., spill, needle stick, etc.) and the exact HD involved. There also needs to be a “physical assessment” of the employee based on the organ involved. This would be a time to double-check that the controls in place are adequate and to “conduct environmental” samples. Something that could be missed that is very important is double
checking and documenting that all “engineering controls,” like the hood and ventilation from the room, are working properly. You may have noticed that there is a good amount of requirements for documentation, and that is because of the old adage in pharmacy. “if it isn’t documented, it didn’t happen.” Pharmacies should also document whether an employee was in compliance with existing policies, including PPE and other SOPs, as well as whether there is the availability of “appropriate PPE.” It is vital to create a plan to prevent future HD exposure to employees and document this plan. Confidential communication between employees and the medical surveillance team is important to discuss any changes in health relating to potential exposure to HDs. If it is determined that the employee was impacted by the exposure to HDs at the entity, it is vital to notify the employee and offer a change to their assignment quickly and privately. Also, make sure to use medical surveillance for the entire team that could be at risk of the same exposure to make sure the plan implemented is working.
Clinical setting variation
USP 800 was created as a baseline standard for handling hazardous drugs; however, it will affect entities differently based on the type of compounding, if at all, a pharmacy is doing. The more complicated compounding a pharmacy, especially with hazardous drugs, the more USP 800 will affect your entity. For instance, the average retail pharmacy, which does very limited compounding, like mixing some liquids, will have to adhere to USP 800 requirements having to do with receiving deliveries from a distributor. This is because it is possible for a hazardous drug to spill or arrive broken. Therefore, you need a dedicated area in your pharmacy with everything necessary just in case there is a spill, like a spill kit and proper PPE. Apart from that, there will be very little pharmacy needs to do as there is very little handling of HDs.
Once the pharmacy does hazardous compounding, that is when USP 800 will not only be something that will affect the employees every day but every minute of their job. Even non-sterile hazardous compounding requires a significant increase in controls in place, as mentioned above. Finally, there is
sterile compounding which is even more significant due to the high level of hygiene necessary. If you are working in a hospital setting or infusion, sterile compounding will be very common, and therefore, USP 800 will be enforced strictly.
Geography will also affect the employee as states have decided to enforce USP 800 differently. These differences make it important for pharmacies to be in communication with their board of pharmacy and state pharmacist associations to confirm the standards in their state.
Since there are many recommendations in USP 800 rather than requirements, there may also be large variations within a state between similar entities. Just like pharmacy practice not having to do with hazardous drugs, it is okay for companies to adopt different policies and for entities to change policies significantly. The main goal for each entity is to have the best policies in place with the information they have. As time goes on and more data is collected, such as exposure incidents and medical surveillance alerts, the entity must change its policies to mitigate these issues. If you are the pharmacy owner or pharmacy manager, you have a significant legal responsibility as an error can impact not only the health of your patients but your staff, so it would be advised to be on the more conservative side of USP 800. Furthermore, consult your compounding distributor or a qualified compounding pharmacist consultant to clarify what is necessary for your pharmacy. If a pharmacy is starting from scratch, it is advisable to hire a company with experience building compounding clean rooms, that are USP 800 compliant, to guide the construction of the room. They will know what is necessary, from materials for all surfaces to ventilation and spacing of various appliances like the hood and drainage.
Patient Counseling on the Handling of Hazardous Drugs
The patient or the caregiver is at risk of exposure to HDs, so the following are good counseling points. Some HDs require specific PPE for the patient or the caregiver and must be disposed of in an HD waste container. Equipment involved in the administration of HDs must also be disposed of in
an HD waste container. It is important to use CSTDs, when possible, to decrease exposure. Crushing tablets or opening capsules should be avoided by changing to a liquid formulation. However, if this is not possible, then PPE should be used, and a plastic pouch can be used.
Summary
The proper handling of HDs could be the difference between safe handling and harm for pharmacists and pharmacy technicians. Since the 1970s in the US, there has been escalating evidence of harm to pharmacy staff due to various medications they were handling.
In 1990, the ASHP introduced the term “hazardous drugs.” The term “hazardous drugs” is now used to describe these dangerous drugs. This term replaced their prior designation as cytotoxic or antineoplastic drugs. The USP publishes the guidelines for the handling of HDs.
USP 800 is the latest regulation regarding the handling of HDs in the healthcare setting. Its purpose is to promote patient safety, worker safety, and environmental protection. The USP 800 took the previous recommendations and developed from them a federal “minimum standard” for handling HDs. USP 800 was officially published in 2016 and created guidance on the handling of HDs in the healthcare setting, including sterile and nonsterile compounding.
The NIOSH published an updated list of HDs in 2020. This updated list is not final but is a draft awaiting comment from various experts and stakeholders, including pharmacists and pharmacy staff.
Course Test
C-PEC is a
compounding room.
compounding hood.
compounding cabinet.
compounding drawer.
C-SEC is a
compounding room.
compounding hood.
compounding cabinet.
compounding instrument.
USP 795 provides guidelines for
non-compounding.
non-compounding hazardous.
non-sterile compounding.
sterile hazardous compounding.
USP 797 provides guidelines for
non-compounding.
non-compounding hazardous.
non-sterile compounding.
sterile compounding.
USP 800 provides guidelines for
non-compounding hazardous.
non-sterile compounding.
sterile-compounding.
handling hazardous drugs.
True or False: Only the U.S. Food and Drug Administration (FDA) enforces USP 800.
True
False
True or False: Every state enforces USP 800 the same.
True
False
USP 800 provides protection for
patients.
workers and caregivers.
environment.
All of the above
A pharmacy’s Standard Operating Procedures (SOPs) for handling HDs
involves voluntary medical surveillance of staff.
must be specific to a pharmacy’s compounding activity.
must be set by an outside, independent auditor.
are maintained by NIOSH.
At a minimum, training regarding HD SOPs for workers must be done
bi-annually.
weekly.
monthly.
yearly.
References
Andrews SR, Dill JL. USP <800>. Hosp Pharm. 2018;53(3):132-133. doi:10.1177/0018578718763850
Connor TH, McDiarmid MA. Preventing occupational exposures to antineoplastic drugs in health care settings. CA Cancer J Clin. 2006;56(6):354-365. doi:10.3322/canjclin.56.6.354
O'Connor S, Kerr KA, Chan A, et al. ISOPP Standards Committee. ISOPP Standards for the safe handling of cytotoxics. J Oncol Pharm Pract. 2021;28(3 Suppl): S1-S126. doi: doi.org/10.1177/10781552211070933
Walton AL, Eisenberg S, Friese CR. Hazardous Drugs: Legislative and Regulatory Efforts to Improve Safe Handling. Clin J Oncol Nurs. 2017;21(2):254-256. doi:10.1188/17.CJON.254-256
Centers for Disease Control and Prevention. National Occupational Exposure Survey (NOES). CDC. https://www.cdc.gov/noes/default.html. Accessed November 27, 2022.
American Society of Hospital Pharmacists. ASHP technical assistance bulletin on handling cytotoxic and hazardous drugs. Am J Hosp Pharm. 1990;47:1033-49.
Vuelta-Arce M, Chiapella-Micó C, Mestre-Prad MT, et al. Comprehensive tackling to the safe handling of hazardous drugs: a multidisciplinary approach to clinical practice. Int J Occup Med Environ Health. 2020;33(5):621-634. doi:10.13075/ijomeh.1896.01534
US Department of Labor. Occupational Safety and Health Administration. Hazardous Drugs. Overview. OSHA. Undated. https://www.osha.gov/hazardous-drugs. Accessed November 26, 2022.
US Department of Labor. Occupational Safety and Health Administration. Controlling Occupational Exposure to Hazardous Drugs. OSHA. August 1, 2016. https://www.osha.gov/hazardous-
drugs/controlling-occex. Accessed November 27, 2022.
Centers for Disease Control and Prevention. NIOSH alert; preventing occupational exposures to antineoplastic and other hazardous drugs in health care settings. CDC. 2004. https://stacks.cdc.gov/view/cdc/6030. Accessed November 28, 2022.
Beans BE. USP <800> Adds Significant Safety Standards: Facility Upgrades Needed to Protect Employees From Hazardous Drugs. P T. 2017;42(5):336-339.
American Society of Health-System Pharmacists. ASHP guidelines on handling hazardous drugs. Am J Health-Syst Pharm. 2006; 63:1172-93.
Page MR, Protecting Health Care Workers from Chemotherapeutic Medication. Pharm. Times. 2015;6(3):30-34.
Mathias PI, MacKenzie BA, Toennis CA, Connor TH. Survey of guidelines and current practices for safe handling of antineoplastic and other
hazardous drugs used in 24 countries. J Oncol Pharm Pract.
2019;25(1):148-162. doi:10.1177/1078155217726160
Connor TH, MacKenzie BA, DeBord DG, Trout DB, O’Callaghan JP, Ovesen JL, Whittaker C.NIOSH List of Hazardous Drugs in Healthcare Settings, 2020. 2020. CDC. NIOSH.
https://www.cdc.gov/niosh/docket/review/docket233c/pdfs/DRAFT- NIOSH-Hazardous-Drugs-List-2020.pdf. Accessed December 19, 2022.
Davis J, Ayars C. Retention of Sterile Compounding Knowledge Among Pharmacy Technicians. J Pharm Technol. 2021;37(5):219-224. doi:10.1177/87551225211032395
Desselle SP, Hohmeier KC, McKeirnan KC. The Value and Potential Integration of Pharmacy Technician National Certification into Processes That Help Assure a Competent Workforce. Pharmacy (Basel). 2019;7(4):147. Published 2019 Nov 5. doi:10.3390/pharmacy7040147
United States Pharmacopeial Convention. USP 43-NF 38 as of June 1, 2020. United States Pharmacopeial Convention; Rockville, MD, USA: 2020. USP General Chapter <800> Hazardous Drugs-Handling in Healthcare Settings. https://www.usp.org/sites/default/files/usp/document/our- work/healthcare-quality-safety/general-chapter-800.pdf. Accessed December 19, 2022.
Centers for Disease Control and Prevention. Hazardous Drug Exposures in Healthcare.CDC. NIOSH. 2020. https://www.cdc.gov/niosh/topics/hazdrug/default.html. Accessed December 19, 2022.
Gurusamy KS, Best LM, Tanguay C, Lennan E, Korva M, Bussières JF. Closed-system drug-transfer devices plus safe handling of hazardous drugs versus safe handling alone for reducing exposure to infusional hazardous drugs in healthcare staff. Cochrane Database Syst Rev. 2018;3(3):CD012860. Published 2018 Mar 27. doi:10.1002/14651858.CD012860.pub2
Centers for Disease Control and Prevention. Hazardous Drug Exposures in Healthcare. Closed System Drug-Transfer Device (CSTD) Research. CDC. NIOSH. 2020.
https://www.cdc.gov/niosh/topics/hazdrug/CSTD.html. Accessed December 26, 2022.
Walton AL, Kneipp S, Linnan L, et al. Nursing Assistants' Use of Personal Protective Equipment Regarding Contact With Excreta Contaminated With Antineoplastic Drugs. Oncol Nurs Forum. 2019;46(6):689-700. doi:10.1188/19.ONF.689-700
Power LA, Sessink PJ, Gesy K, Charbonneau F. Hazardous drug residue on exterior vial surfaces: evaluation of a commercial manufacturing process. Hosp Pharm. 2014;49(4):355-362. doi:10.1310/hpj4904-355
Sessink PJ, Trahan J, Coyne JW. Reduction in Surface Contamination With Cyclophosphamide in 30 US Hospital Pharmacies Following Implementation of a Closed-System Drug Transfer Device. Hosp Pharm. 2013;48(3):204-212. doi:10.1310/hpj4803-204
DISCLAIMER
The information provided in this course is general in nature, and it is solely designed to provide participants with continuing education credit(s). This course and materials are not meant to substitute for the independent, professional judgment of any participant regarding that participant’s professional practice, including but not limited to patient assessment, diagnosis, treatment, and/or health management. Medical and pharmacy practices, rules, and laws vary from state to state, and this course does not cover the laws of each state; therefore, participants must consult the laws of their state as they relate to their professional practice.
Healthcare professionals, including pharmacists and pharmacy technicians, must consult with their employer, healthcare facility, hospital, or other organization, for guidelines, protocols, and procedures they are to follow. The information provided in this course does not replace those guidelines, protocols, and procedures but is for academic purposes only, and this course’s limited purpose is for the completion of continuing education credits.
Participants are advised and acknowledge that information related to medications, their administration, dosing, contraindications, adverse reactions, interactions, warnings, precautions, or accepted uses are constantly changing, and any person taking this course understands that such person must make an independent review of medication information prior to any patient assessment, diagnosis, treatment and/or health management. Any discussion of off-label use of any medication, device, or procedure is informational only, and such uses are not endorsed hereby.
Nothing contained in this course represents the opinions, views, judgments, or conclusions of RxCe.com LLC. RxCe.com LLC is not liable or responsible to any person for any inaccuracy, error, or omission with respect to this course, or course material.
ⓒ RxCe.com LLC 2022: All rights reserved. No reproduction of all or part of any content herein is allowed without the prior, written permission of RxCe.com LLC.
