THE ABCs OF IBS
L. Austin Fredrickson, MD
Dr. Fredrickson is an associate professor of internal medicine at Northeast Ohio Medical University, where he serves as core faculty and teaches diagnostics, therapeutics, clinical skills, and health humanities. He is board- certified in general internal medicine and practices rural primary care.
Topic Overview
Irritable bowel syndrome, or IBS, is a challenging gastrointestinal disorder that not only causes abdominal pain but often leads to decreased mental health, well-being, and quality of life for patients. The pathophysiology and treatment strategies associated with IBS are complex, and its management requires a multidisciplinary approach centered on the patient’s predominant symptoms. This continuing education program will discuss the pathological processes involved in the development of IBS, the approach to diagnosis, and treatment strategies. Nonpharmacologic and pharmacologic approaches will be explored with a focus on emerging treatment strategies.
Accreditation Statement

RxCe.com LLC is accredited by the Accreditation Council for Pharmacy Education (ACPE) as a provider of continuing pharmacy education.
Universal Activity Number (UAN): The ACPE Universal Activity Number assigned to this activity is
Pharmacist 0669-0000-24-040-H01-P
Pharmacy Technician 0669-0000-24-041-H01-T
Credits: 2 contact hour(s) (0.2 CEU(s)) of continuing education credit
Type of Activity: Knowledge
Media: Internet/Home study Fee Information: $6.99
Estimated time to complete activity: 2 contact hour(s) (0.2 CEU(s)), including Course Test and course evaluation
Release Date: March 24, 2024 Expiration Date: March 24, 2027
Target Audience: This educational activity is for pharmacists and pharmacy technicians.
How to Earn Credit: From March 24, 2024, through March 24, 2027, participants must:
Read the “learning objectives” and “author and planning team disclosures;”
Study the section entitled “Educational Activity;” and
Complete the Course Test and Evaluation form. The Course Test will be graded automatically. Following successful completion of the Course Test with a score of 70% or higher, a statement of participation will be made available immediately. (No partial credit will be given.)
Credit for this course will be uploaded to CPE Monitor®.
Learning Objectives: Upon completion of this educational activity, participants should be able to:
Describe the pathophysiology of irritable bowel syndrome (IBS)
Differentiate the types of IBS based on presenting symptoms
Identify nonpharmacologic strategies for the management of IBS
Compare and contrast pharmacologic agents used in the management of IBS
Disclosures
The following individuals were involved in developing this activity: L. Austin Fredrickson and Pamela Sardo, PharmD, BS. Pamela Sardo and L. Austin Fredrickson have no conflicts of interest or financial relationships regarding the subject matter discussed. There are no financial relationships or commercial or financial support relevant to this activity to report or disclose by RxCe.com or any of the individuals involved in the development of this activity.
© RxCe.com LLC 2024: All rights reserved. No reproduction of all or part of any content herein is allowed without the prior, written permission of RxCe.com LLC.
Educational Activity
The ABCs of IBS Introduction
Irritable bowel disorder is a gastrointestinal disorder that causes abdominal pain, decreased mental health, and a lower quality of life for patients. This condition is very common in the United States. It is difficult to confirm irritable bowel disorder in a patient, but a thorough patient history makes a diagnosis more certain. Its management is also complex, requiring a multidisciplinary approach centered on the patient’s predominant symptoms. This continuing education program will discuss the pathological processes involved in developing irritable bowel disorder, the approach to diagnosis, and treatment strategies. Nonpharmacologic and pharmacologic approaches will be explored, focusing on emerging treatment strategies.
Disorders of Gut-Brain Interaction
Disorders of gut-brain interaction (DGBI), previously known as functional bowel disorders, encompass a group of more than 30 gastrointestinal disorders with symptoms that result from interactions between altered gut signaling, altered mucosal immune function, microbial dysbiosis, and central nervous symptoms dysregulation of gut signaling and motor function modulation.1 These disorders can be further classified into eight smaller categories. (See Table 1).1
Table 1
Functional Gastrointestinal Disorders
| Category | Example |
| Esophageal disorders | Globus |
| Gastroduodenal disorders | Functional dyspepsia |
| Bowel disorders | Irritable bowel syndrome |
| Centrally mediated disorders of gastrointestinal pain | Narcotic bowel syndrome |
| Gallbladder and Sphincter of Oddi (SO Disorders | Biliary pain |
| Anorectal disorders | Functional anorectal pain |
| Childhood Functional GI Disorders: Child/Adolescent | Postprandial distress syndrome |
| Childhood Functional GI Disorders: Neonate/Toddler | Infant regurgitation |
Among these bowel disorders, irritable bowel disorder (IBS) is a highly prevalent, chronic, and debilitating condition most notable for symptoms of recurrent abdominal pain and abnormal bowel habits.2 This challenging gastrointestinal disorder not only causes abdominal pain but often leads to decreased mental health, well-being, and quality of life for patients.2 On a broader scale, IBS contributes significantly to healthcare system costs, with direct costs estimated between 1.5-10 billion dollars per year in the United States.3
Epidemiology
A recent large-scale, nationwide, cross-sectional study estimated the prevalence of IBS within the United States at 6.1%.4 This is higher than previous estimates of between 4.7 and 5.3%.4 Among study participants, women were more likely to have IBS compared to men, and non-Hispanic whites were more likely than racial and ethnic minorities.4 Further, individuals of younger age, current smokers, those with fibromyalgia, and those with prior gastroenteritis were also more likely to have IBS.4
Risk factors for developing IBS can be categorized as either peripheral or central factors. (See Table 2).5 Peripheral factors account for a higher proportion of patients with IBS and are more likely to cause IBS of mild-to- moderate severity.5 Central factors, while accounting for a lower percentage of cases, tend to cause more severe IBS.5 Enteric infection is the most notable risk factor for IBS, present in up to 10% of all IBS patients.5
Table 2
Risk Factors for IBS5
| Peripheral Risk Factors | Central Risk Factors |
| Food | Life stress |
| Acute gastrointestinal infection | Somatization |
| Mucosal inflammation | Anxiety or depression |
| Abdominal or pelvic surgery | Poor coping skills |
| Menses | Poor social support |
| Maladaptive cognitions | |
| Abuse |
Pathophysiology
The pathophysiology of IBS is complex and only partially understood. Research into this disorder continues to uncover new revelations. Numerous factors may contribute to this disorder, including motility abnormalities, brain- gut interactions, leaky gut dysbiosis, visceral hypersensitivity, bile acid malabsorption, genetics, and psychological factors, such as anxiety and depression.6 Table 3 summarizes contributors to the pathophysiology of IBS.
Table 3
Components of IBS Pathophysiology5,6
| Component | Details |
| Motility abnormalities | Increased rectosigmoid motor activity up to 3 hours after eating Rapid colonic transit and abdominal pain |
Visceral hypersensitivity (the threshold for stomach pain is lower than normal) | Exaggerated sensory response to visceral stimulation Pain post-eating |
| Central nervous system dysregulation | Stress and emotional disorders are associated with symptom exacerbation |
| Psychologic factors | Anxiety and depression |
| Diet | FODMAPSa can cause increased GI water secretion and fermentation in the colon, which can result in the production of short-chain fatty acids, causing luminal distention and subsequent IBS symptoms |
| Postinfectious IBS | GI infections can predispose patients to IBS |
| Immune activation/mucosal inflammation | Low-grade inflammation (activated lymphocytes, mast cells) |
| Altered gut flora | Overgrowth of small intestinal bacteria Gut dysbiosis + genetic and environmental factors can alter the permeability of the intestines |
| Abnormal serotonin pathways | In some IBS-D patients, serotonin- containing enterochromaffin cells in the colon are increased Increased serotonin can cause postprandial symptoms |
| Genetics | Variants on chromosome 9 are linked to the functions of diverse ion channels and autonomic dysfunction. Up to 2% of IBS patients have missense mutations in SCN5A, which can also cause affected smooth muscle function and mechanical sensitivity |
aFODMAPS: fermentable oligosaccharides, disaccharides, monosaccharides, and polyols
Clinical Presentation and Diagnosis
The diagnosis of IBS may challenge clinicians for numerous reasons, including the ability of symptoms to change over time and mimic other disorders, a lack of awareness of the current guidelines being used to diagnose IBS, and the lack of a biomarker or test to confirm the diagnosis.7 While clinicians may feel more comfortable using a diagnosis of exclusion, the 2021 American College of Gastroenterology 2021 Guidelines recommend using a positive diagnostic strategy or symptom-based diagnosis to diagnose IBS.2,7 With this approach, a careful history, physical examination, and use of a standard definition are employed.2 It has been suggested that this approach improves patient education and reassurance and improves time to appropriate treatment and cost-effectiveness.2
As the diagnosis of IBS has evolved, some clinicians may diagnose IBS in patients who do not meet the current diagnostic criteria. However, some of the treatments for IBS are generally good advice for patients whose abdominal pain comes from other disorders, so being aware of best practices for abdominal pain, in general, helps diagnose and educate patients about IBS.
Patients presenting with IBS most notably exhibit abdominal pain, which is necessary for the diagnosis.2 However, because abdominal pain is a symptom present in many conditions, other conditions must be ruled out.5 Irritable bowel disorder is notable for pain in the lower abdomen that is intermittent, not continuous.5 To confirm the diagnosis, the pain should be associated with defecation and an alteration in stool frequency or consistency.5 The Rome IV diagnostic criteria for IBS are used to diagnose this disorder. (See Table 4).2
Table 4
Rome IV Diagnostic Criteria2
| Recurrent abdominal pain on average at least 1 day per week in the last three months associated with 2 or more of the following criteria: |
| Related to defecation |
| Associated with a change in frequency of stool |
| Associated with a change in the form or appearance of stool |
| These criteria should be fulfilled for the last 3 months, with symptom onset at least 6 months before the diagnosis |
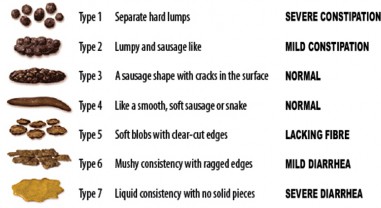
Irritable bowel disorder can be classified into four different subtypes based on the predominant bowel habit.7 The ACG Guidelines suggest categorizing patients to improve therapeutic management strategies by enabling clinicians to best treat the most troublesome symptoms.2 Irritable bowel disorder subtypes are based on the patient’s main bowel habit on any day with abnormal bowel habits rather than an average of all days.2 The Bristol Stool Form Scale (BSFS) is utilized to classify abnormal bowel habits. (See Table 5 and Bristol Stool Form Scale Figure below).2,8
Table 5
Bristol Stool Form Scale2,7
| Type | Description | Association |
| Type 1 | Separate hard lumps (hard to pass) | Constipation |
| Type 2 | Sausage-shaped but lumpy | Constipation |
| Type 3 | Like a sausage but with cracks on its surface | Normal |
| Type 4 | Like a sausage or snake, smooth and soft | Normal |
| Type 5 | Soft blobs with clear-cut edges (passed easily) | Diarrhea |
| Type 6 | Fluffy pieces with ragged edges, a mushy stool | Diarrhea |
| Type 7 | Watery, no solid pieces, entirely liquid | Diarrhea |
Figure
Bristol Stool Form Scale8
A thorough history is needed to make the diagnosis of IBS.2 An important component of this is ruling out the presence of any warning signs, including patients 50 years or older who have not been screened for colon cancer, patients with GI bleeding, nocturnal passage of stools, unintentional weight loss, family history of inflammatory bowel disease or colorectal cancer, recent changes in bowel habits, presence of palpable abdominal mass or lymphadenopathy.2 If no warning signs are present, clinicians can then proceed to determine the frequency of symptoms and if the patient meets
Rome IV criteria.2 Finally, associated bowel habits should be reviewed to determine the IBS subtype.2 Often, patients will feel relief in having a diagnosis for their symptoms and complaints, as the unknown cause of their condition can worsen anxiety and even the IBS symptoms themselves. Empowering patients with a diagnosis they can research and find support groups for is important even when controlling the resultant symptoms may remain challenging. Irritable bowel disorder subtypes are described in Table 6.
| Subtype | Abbreviation | Definition |
| IBS with predominant constipation | IBS-C | >25% of bowel movements associated with BSFS 1 or 2 and <25% associated with BSFS 6 or 7 |
| IBS with predominant diarrhea | IBS-D | >25% of bowel movements are associated with BSFS 6 or 7, and <25% are associated with BSFS 1 or 2 |
| IBS with mixed bowel habits | IBS-M | >25% of bowel movements associated with BSFS 1 or 2 and >25% associated with BSFS 6 or 7 |
| IBS un-subtyped | IBS-U | Indeterminable |
Table 6 IBS Subtypes
A physical examination should be completed, with benign findings notable for supporting an IBS diagnosis.2 Atypical or concerning findings should be worked up before labeling it IBS. The ACG Guidelines also recommend serologic testing to rule out celiac disease in patients with IBS and diarrhea symptoms due to evidence that finds patients with IBS symptoms have an increased chance of also having celiac disease.2,9,10
As mentioned above, there is no definitive biomarker for diagnosing IBS. This can frustrate clinicians who cannot rely upon laboratory values for either a diagnosis or a way to monitor and assess response to treatment over time. History and physical examinations remain the crucible for diagnosis. However, to assist clinicians in excluding a diagnosis of inflammatory bowel disease (which can present similarly to IBS-D), erythrocyte sedimentation rate (ESR)
and C-reactive protein (CRP) can be used.2 In a comprehensive meta-analysis a CRP less than 0.5 mg/dL yielded a 1% probability of IBD.11 CRP is also favorable due to its quick turnaround time and availability.2 Fecal lactoferrin (FL) and fecal calprotectin (fCal) are fecal-derived markers of intestinal inflammation that have been found to be superior to CRP and ESR in their ability to distinguish between IBD and IBS accurately.2 Specifically, fCal has a better negative predictive value than CRP and ESR.12 Both tests are safe and noninvasive. Given this information, the ACG Guidelines suggest either fCal or FL and CRP be checked in patients with no alarm symptoms and suspected IBS-D to rule out IBD.2
In terms of imaging, the ACG guidelines recommended against colonoscopy for patients with IBS who meet the following criteria: are younger than 45 years of age, and do not have alarming symptoms.2 Patients older than 60 years of age, female, and with intense diarrhea are at higher risk of microscopic colitis and may benefit from a colonoscopy.2 Recall microscopic colitis will not be determined by visualization alone, so biopsy during colonoscopy is key to that diagnosis. Those patients who meet the criteria for colorectal screening through their age and/or risk factors should undergo routine screening for primary prevention of colorectal cancer.
Approaches to IBS Management
The management of IBS is multifactorial, focusing on both lifestyle interventions and pharmacologic treatments based on the subtype of IBS present. The overarching goal of treatment is symptomatic relief via reducing both the severity and frequency of symptoms and improving the patient’s quality of life.2 The ACG recommends a multimodal treatment approach that includes dietary, behavioral, and pharmacological strategies. Decisions should be patient-centric, and shared decision-making should always be employed.2
Lifestyle Interventions
Changes in a patient’s diet may lead to improved symptoms; however, it is important for clinicians to explain to patients the functional nature of the
disease.6 In some cases, a dietary history may identify foods that trigger symptoms, such as coffee or legumes.6 Patients should work closely with their care team to avoid diets that are overly restrictive or nutritionally void.6 Patients can be empowered by explaining much of the treatment is in their hands. They can control variables themselves without clinician-prescribed pharmacotherapy.
To improve global symptoms, the ACG guidelines recommended a limited trial of low FODMAP in patients with IBS.2 A list of low and high FODMAP foods is presented in Table 7.13 In a meta-analysis of seven randomized controlled trials, a low FODMAP diet was compared to seven different comparators.14 With a risk ratio of 0.69, the low FODMAP diet was found to reduce global IBS symptoms compared with the other comparators significantly, but the study noted all included trials had a high risk of bias.14 In general, patients who respond to low FODMAP will do so within 2-6 weeks, though ongoing improvements may continue through longer periods of time.2 Low FODMAP is not without risks, including nutritional deficiencies, so the need for patient resources and counseling under a highly skilled dietician is important.2 More research is needed to support the recommendation for low FODMAP as well as to evaluate the risks and unknown consequences better.
Table 7
Low FODMAP Diet13
| Foods to Include | Foods to Avoid |
| Banana | Asparagus |
| Blueberries | Eggplant |
| Sucrose/Glucose | Onion |
| Lettuces | Cow and goat milk |
| Rice | Yogurt |
| Spinach | Beans |
| Hard cheeses (brie) | Green bell peppers |
A second dietary recommendation from the ACG is the inclusion of soluble (not insoluble) fiber into the diet to treat global IBS symptoms.2 Dietary fiber is defined as carbohydrates that are not digested or absorbed in
the small intestine and have a degree of polymerization of 3 or more monomeric units.2 In general, individuals should eat a minimum of 25-35 grams of fiber per day.2 Fiber has numerous GI effects, affecting the gut microbiome, metabolism, transit time, stool consistency, and the absorption of bile acids.
In a systematic review and meta-analysis of the effects of fiber on IBS, fiber had a statistically significant benefit for IBS symptoms when compared to placebo. Notably, these study endpoints did not follow modern regulatory guidance.15 Like low FODMAP, more data is needed to determine the effects of fiber on IBS symptoms, specifically regarding the various IBS subtypes. However, fiber is associated with no significant adverse effects and thus has been recommended as a first line for symptomatic patients with IBS.2
Sources of fiber can include foods high in fiber and the use of bulking agents, including hydrophilic colloids.2 Examples of foods containing soluble fiber include psyllium, oat bran, barley, and beans.2 Psyllium (brand name Metamucil) is a bulk-producing fiber supplement available over the counter (OTC).16 The recommended dose for IBS is 3-4 grams per day in one or two divided doses.16 This dose can be titrated every 1-2 weeks to achieve a recommended intake of 25-35 grams of fiber per day.2 Fiber supplements generally do not cause major side effects, but patients can experience constipation, flatulence, and diarrhea, especially if the dose is titrated too quickly.2 This is another time where “start low and go slow" may alleviate patient frustration and side effect burden.
Various psychological treatments, including cognitive behavioral therapy and mindfulness-based therapies, have shown benefits in the treatment of IBS. CBT involves education regarding stress responses and how they relate to GI symptoms, developing insight into cognitive and behavioral responses to IBS symptoms and the fear of these symptoms, and modifying the response to decrease stress and physical reactivity related to IBS symptoms.17 Many stress relieving techniques benefit other conditions that IBS patients may have co-occurring, including migraines, anxiety, or fibromyalgia, so the benefits of these practices may go beyond improving their IBS symptoms alone.
Pharmacologic Treatment Strategies
Pharmacists play a vital role in the management of patients with IBS. Pharmacists can review patients’ medication lists to see if they are taking medications that may cause or worsen either constipation (such as opioids) or diarrhea (such as metformin). Pharmacists can also provide vital education regarding IBS treatments to ensure patients take the medications appropriately, are aware of serious side effects, and stay adherent to therapy. Finally, many IBS medications are cost-prohibitive, and pharmacists can assist patients in identifying patient assistance programs to lower drug costs.18 The pharmacologic treatment of IBS-C and IBS-D will be discussed below.
Treatment of IBS-C
The 2022 AGA Clinical Practice Guidelines on the Pharmacologic Management of Irritable Bowel Syndrome with Constipation guidelines conditionally recommend the use of four agents: tenapanor, plecanatide, tegaserod, and lubiprostone.19 Linaclotide is the only agent with a strong recommendation for use.19 These agents are discussed in detail below, and doses are summarized in Table 8. Additionally, several adjunct therapies are also recommended conditionally, including PEG laxatives, tricyclic antidepressants (TCAs), and antispasmodics.19
Table 8
Medications for the Treatment of IBS-C20-24
| Generic | Brand | Dose | Notes | Side Effects |
| Tenapanor | Ibsrela®; Xphozah® | 50 mg twice daily | Improvement should be seen by week 4 | Diarrhea, abdominal distention, dizziness |
| Plecanatide | Trulance | 3 mg once daily | Symptom improvement | Abdominal distention, |
| by day 7 | diarrhea, | |||
| flatulence, | ||||
| nausea | ||||
| Tegaserod | Zelnorm | 6 mg twice daily 30 | Discontinue if there is no | Headache, abdominal |
| minutes before meals | symptom improvement by 4-6 weeks | pain, nausea, diarrhea | ||
| Lubiprostone | Amitiza | 8 mcg twice daily | Symptom improvement may take 2+ months | Diarrhea, nausea, headache, dizziness |
| Linaclotide | Linzess | 290 mcg once daily; reduce to 145 mcg once daily if diarrhea develops | Symptom improvement by day 7 | Diarrhea, typically in the first 1-2 weeks |
Tenapanor
Tenapanor is a first-in-class small-molecule inhibitor of the GI sodium/hydroxide exchanger isoform 3.19 This isoform is responsible for sodium absorption on the apical surface of the small intestine and colon. Tenapanor decreases sodium and phosphate absorption and increases water secretion into the intestinal lumen.19 The safety and efficacy of tenapanor was demonstrated in 2, phase 3 RCTs in which patients received tenapanor 50 mg twice daily for 12 weeks.25-27 Patients in the tenapanor group had greater symptom relief compared to placebo (RR: 0.84; 95% CI).25-27 Tenapanor also did better with respect to abdominal pain and improvement in CSBM response.25-27 Diarrhea occurred in 14.8% of patients taking tenapanor compared to 2.3% in the placebo group. 25-27
Plecanatide
Plecanatide is a non-absorbed 16-amino acid peptide similar structurally to uroguanylin, which stimulates the guanylate cyclase C (GC-C) receptor on enterocytes via cyclic guanosine monophosphate.19 This ultimately leads to reductions in visceral hypersensitivity.19 Plecanatide is not sensitive to pH and has a greater affinity for the GC-C receptor than linaclotide.19 In two phase 3 trials, patients who received plecanatide had greater symptom relief compared
to the placebo group (RR, 0.87; 95% CI).28 Patients also had greater improvements in abdominal pain.28 Diarrhea occurred in 4.3% of patients compared to 1% of patients taking the placebo.28
Linaclotide
Linaclotide is a non-absorbed 14-amino acid peptide that stimulates the GC-C receptor similar to plecanatide.19 This results in the secretion of chloride and bicarbonate, which inhibits colonic nociceptors.19 In a phase 3 trial, 60% of patients receiving linaclotide reported more than a 30% decrease in abdominal pain and discomfort compared to 48% of patients in the placebo group.29 Diarrhea occurred in 16.3% of patients compared to 2.3% taking placebo.29
Tegaserod
Tegaserod is a partial agonist of the 5-HT4 receptor, which stimulates gastrointestinal motility and increases fluid in the GI tract.19 This agent was initially approved in 2002 for short-term IBS-C treatment in women and removed from the market in 2007 due to a risk of cardiovascular ischemic events.19 After re-evaluating the data, the FDA re-approved tegaserod for women with IBS-C under 65 years of age and without a history of myocardial infarction, stroke, transient ischemic attack, or angina.19 Women who received tegaserod in trials had greater symptom relief compared to placebo (RR, 0.87; 95% CI), and its use was also associated with a 52.7% global relief compared to 44.8% in the placebo group.30-33 Tegaserod most commonly causes diarrhea and headache.199
Lubiprostone
Lubiprostone is a chloride channel type 2 activator that increases the influx of chloride into the lumen of the GI tract, which results in acceleration of the intestinal tract.19 In two phase 3 RCTs, lubiprostone was superior to placebo with regard to abdominal pain improvement and SBM response).34
Treatment of IBS-D
The 2022 AGA Clinical Practice Guidelines on the Pharmacologic Management of Irritable Bowel Syndrome with Diarrhea conditionally recommends the use of three agents: eluxadoline, rifaximin, and alosetron. (See Table 9).35 Additionally, several adjunct therapies are also recommended conditionally, including loperamide, TCAs, and antispasmodics.35 These agents are discussed in detail below.
| Generic | Brand | Dose | Notes | Side Effects |
| Eluxadoline | Vizberzi | 100 mg twice | Symptoms | Constipation, |
| daily; decrease | should improve | nausea, | ||
| to 75 mg twice | by one month. | abdominal | ||
| daily in patients | Use only in | pain, upper | ||
| with intolerance | patients with a | respiratory | ||
| gallbladder | tract infection | |||
| who do not | ||||
| consume >3 | ||||
| alcoholic | ||||
| beverages per | ||||
| day or have a | ||||
| history of | ||||
| alcohol use | ||||
| disorder | ||||
| Rifaximin | Xifaxan | 550 mg three | Reserve for | Edema, |
| times daily for | patients who | nausea, | ||
| 14 days | have failed | ascites, | ||
| other therapies | dizziness, | |||
| fatigue | ||||
| Alosetron | Lotronex | 0.5 mg twice | If there is no | Constipation |
| daily; | response after | (dose-related), | ||
| discontinue if | 4 weeks at 1 | abdominal | ||
| constipation | mg twice daily | pain, nausea | ||
| occurs; increase | dose, | |||
| to 1 mg twice daily if needed; | discontinue | |||
| max dose of 2 | ||||
| mg/day |
Table 9 Medications for IBS-D36-38
Eluxadoline
Eluxadoline is a mu and kappa-opioid receptor agonist and delta-opioid receptor antagonist that is minimally absorbed.35 In 2 phase 3 trials, more patients take eluxadoline (27.2%) were end-point responders compared to placebo (16.7%) (RR, 0.86; 95% CI).39 Other patients reported relief for more than 6 of the first 12 weeks compared to placebo.39 Eight percent of patients discontinued eluxadoline due to side effects compared to placebo (4%).39
Rifaximin
Rifaximin is a nonabsorbable oral antibiotic with activity against gram- positive and gram-negative anaerobic and aerobic bacteria.35 Three RCTs evaluated rifaximin against placebo for treatment of non-constipated Rome 2- positive IBS.40-42 Rifaximin had greater response based on endpoints compared to placebo and also did better with regard to relief of global symptoms and discomfort (RR, 0.87; 95% CI).40-42 In a retreatment trial, adverse events occurred in 3.3% of patients taking rifaximin.43
Alosetron
Alosetron is a selective 5-HT3 antagonist with works both centrally and peripherally.35 This agent was approved in 2000 to treat IBS-D in women and was removed due to the occurrence of serious side effects, including ischemic colitis and serious constipation complications.35 In 2002, it was reapproved by the FDA with use restricted to the treatment of severe IBS-D in women who are part of a risk management program.35 Since its reintroduction, the occurrence of complications due to constipation has declined, and ischemic colitis rates have remained stable.35
Pharmacologic Treatment Strategies: Adjunct Agents
Antispasmodic Agents
Antispasmodic agents assist in preventing cramps associated with intestinal spasms, and they are among the most frequently used medications in the treatment of IBS.6 Antispasmodics are a broad group encompassing direct smooth muscle relaxants, calcium antagonists, scopolamine derivatives, and combination medications.2 Recent ACG guidelines recommend the use of antispasmodic agents to treat global IBS symptoms.19,35 Because of their heterogeneity, it is difficult to ascertain the true efficacy of this group on IBS symptoms.2
Polyethylene Glycol (PEG)
Polyethylene glycol is an osmotic laxative approved for occasional constipation.2 Its use in the treatment of IBS has been evaluated in various studies.2 While some research suggests it improves stool frequency and consistency in patients with IBS-C, there is no data to show it improves overall symptoms or pain in this IBS cohort.2 Additionally, PEG use is associated with a number of side effects, including abdominal pain, diarrhea, nausea, and flatulence.2 With these factors in mind, the ACG does not currently recommend the use of PEG to relieve global IBS symptoms in patients with IBS-C.2
Probiotics
Due to the involvement of the gut microbiome in the development of IBS, the use of probiotics as a management strategy has increased in recent years.2 More research is needed to support the use of probiotics, given the current lack of strong, randomized controlled trials.2 Given the lack of data to support its efficacy, probiotics are not recommended by the ACG.2
Tricyclic Antidepressants and Selective Serotonin Inhibitors (SSRIs)
The peripheral and central effects of TCAs can lead to changes in motility, secretion, and sensation, and this class of medications has historically been used to manage IBS symptoms.2 The AGA currently maintains its recommendation to use TCAs in patients with IBS.19 When compared with placebo, these agents have demonstrated great relief of abdominal pain and provision of adequate relief of IBS symptoms.19 The effects of TCAs are mutually exclusive from their effects on depression, and the onset of effect can take up to several weeks.19 Selection of a TCA is dependent on the patient’s symptoms.19 Second-generation TCAs desipramine and nortriptyline are likely to be better tolerated in patients with IBS-C due to lesser anticholinergic effects compared to first-generation agents.19
Unlike TCAs, SSRIs are not recommended for patients with IBS.19 They do not appear to affect visceral sensations significantly, and studies have not shown a benefit regarding either global symptoms or abdominal pain in IBS.19
Loperamide
Loperamide is a synthetic peripheral opioid receptor agonist that inhibits peristalsis and prolongs intestinal transit time.35 Loperamide has shown efficacy in improving abdominal pain and stool consistency but not improvement in global symptoms of IBS.35
Tips and Considerations for Pharmacy Technicians
Pharmacy technicians are important members of the health care team. Oftentimes, patients may come to the pharmacy seeking treatment for GI- related symptoms, and pharmacy technicians can assist pharmacists in identifying patients who may have IBS or be candidates for referral. Pharmacy technicians can also recognize when patients may or may not be adherent to IBS therapies and assist pharmacists in recognizing and helping these patients.
Notably, PEG laxatives and loperamide received conditional adjunct recommendations, but two characteristics may make them attractive to many patients: cost and availability. These over-the-counter products are easily accessible and are also typically significantly more affordable than the other agents with higher recommendations. Patients are more likely to ask about these particular agents, so familiarity with both and the above patient education points can be particularly helpful for pharmacy technicians on the front lines of patient care.
Summary
Among bowel disorders, IBS is the most common. This chronic and debilitating condition is most notable for symptoms of recurrent abdominal pain and abnormal bowel habits. This challenging gastrointestinal disorder not only causes abdominal pain but often leads to decreased mental health, well- being, and quality of life for patients. Further, IBS contributes significantly to health care system costs, with direct costs in the United States estimated between 1.5-10 billion dollars per year. The pathophysiology and treatment strategies associated with IBS are complex, and its management requires a multidisciplinary approach centered on the patient’s predominant symptoms. Pharmacists should be familiar with both the nonpharmacologic and pharmacologic approaches to managing IBS-C and IBS-D.
Course Test
Which term is now utilized to refer to a group of more than 30 GI disorders formerly described as functional bowel disorders?
Gastrointestinal infections
Disorders of gut-brain interaction
Chronic abdominal pain syndromes
Microbial dysbiosis conditions
Which of the following is or are classic irritable bowel syndrome (IBS) symptom(s)?
Severe rectal bleeding
Recurrent episodes of vomiting
Recurrent abdominal pain with abnormal bowel habits
Persistent heartburn and acid reflux
Which of the following is a known peripheral risk factor for developing IBS?
Acute gastrointestinal infection
Normal aging
Cognitive behavioral therapy
Stroke
Which of the following components of IBS pathophysiology is an example of how dietary factors can cause or worsen symptoms?
Anxiety and depression worsen symptomatology
GI infections can predispose to IBS
High-grade inflammation and inflammatory markers trigger IBS
FODMAPS can cause increased GI water secretion and fermentation in the colon, causing luminal distension and subsequent IBS symptoms
When diagnosing IBS, which of the following is most important to avoid misdiagnosis?
Assess for ‘red flag’ criteria that require further workup
Check thyroid studies
Perform urinalysis
Do not check for celiac disease
Which subtype of IBS is associated with >25% of bowel movements associated with constipation (BSFS 1 or 2/0 and
<25% associated with diarrhea (BSFS 6 or 7)?
IBS-D
IBS-C
IBS-M
IBS-U
Which of the following lab findings are most appropriate for a patient newly diagnosed with IBS?
Elevated CRP, Elevated fecal lactoferrin
Elevated CRP, Elevated ESR
Normal CRP, normal fecal lactoferrin
Abnormal colonoscopy, abnormal celiac testing
Which lifestyle changes are recommended by the 2022 American College of Gastroenterology (ACG) guidelines for IBS patients?
Increase insoluble fiber to 10g/day
Increase FODMAP foods
Increase soluble fiber slowly to 25-35g/day
Increase stress
Which of the following medications has a strong recommendation from the ACG for patients with IBS-C?
Amitriptyline
Sertraline
Linaclotide
Polyethylene glycol
A 50-year-old patient without a gallbladder (status post cholecystectomy) drinks four alcoholic beverages daily. Which IBS-D treatment should be avoided in this patient?
Eluxadoline (Vizberzi)
Polyethylene glycol
Linaclotide
Rifaxamin
References
Drossman DA, Hasler WL. Rome IV-Functional GI Disorders: Disorders of Gut-Brain Interaction. Gastroenterology. 2016;150(6):1257-1261. doi:10.1053/j.gastro.2016.03.035
Lacy BE, Pimentel M, Brenner DM, et al. ACG Clinical Guideline: Management of Irritable Bowel Syndrome. Am J Gastroenterol. 2021;116(1):17-44. doi:10.14309/ajg.0000000000001036
Inadomi JM, Fennerty MB, Bjorkman D. Systematic review: the economic impact of irritable bowel syndrome. Aliment Pharmacol Ther. 2003;18(7):671-682. doi:10.1046/j.1365-2036.2003.t01-1-01736
Almario CV, Sharabi E, Chey WD, Lauzon M, Higgins CS, Spiegel BMR. Prevalence and Burden of Illness of Rome IV Irritable Bowel Syndrome in the United States: Results From a Nationwide Cross-Sectional Study. Gastroenterology. 2023;165(6):1475-1487. doi:10.1053/j.gastro.2023.08.010
Ford AC, Sperber AD, Corsetti M, Camilleri M. Irritable bowel syndrome.
Lancet. 2020;396(10263):1675-1688. doi:10.1016/S0140-
6736(20)31548-8
Owyang C. Irritable Bowel Syndrome. In: Loscalzo J, Fauci A, Kasper D, Hauser S, Longo D, Jameson J. eds. Harrison's Principles of Internal Medicine, 21e. McGraw Hill; 2022.
Lacy BE, Patel NK. Rome Criteria and a Diagnostic Approach to Irritable Bowel Syndrome. J Clin Med. 2017;6(11):99. Published 2017 Oct 26. doi:10.3390/jcm6110099
Bristol Stool Scale. WikiCommons. https://en.wikipedia.org/wiki/Bristol_stool_scale#/media/File:BristolSto olChart_(cropped).png. Accessed January 3, 2024.
Mein SM, Ladabaum U. Serological testing for coeliac disease in patients with symptoms of irritable bowel syndrome: a cost-effectiveness analysis. Aliment Pharmacol Ther. 2004;19(11):1199-1210. doi:10.1111/j.1365-2036.2004.01958.x
Spiegel BM, DeRosa VP, Gralnek IM, Wang V, Dulai GS. Testing for celiac sprue in irritable bowel syndrome with predominant diarrhea: a cost-effectiveness analysis. Gastroenterology. 2004;126(7):1721-1732. doi:10.1053/j.gastro.2004.03.012
Menees SB, Powell C, Kurlander J, Goel A, Chey WD. A meta-analysis of the utility of C-reactive protein, erythrocyte sedimentation rate, fecal calprotectin, and fecal lactoferrin to exclude inflammatory bowel disease in adults with IBS. Am J Gastroenterol. 2015;110(3):444-454. doi:10.1038/ajg.2015.6
Guerrant RL, Araujo V, Soares E, et al. Measurement of fecal lactoferrin as a marker of fecal leukocytes. J Clin Microbiol. 1992;30(5):1238- 1242. doi:10.1128/jcm.30.5.1238-1242.1992
Johns Hopkins Medicine. Low FODAMP diet: what you need to know. https://www.hopkinsmedicine.org/health/wellness-and- prevention/fodmap-diet-what-you-need-to-know. Accessed January 3, 2024.
Dionne J, Ford AC, Yuan Y, et al. A systematic review and meta-analysis evaluating the efficacy of a gluten free diet and a low FODMAP diet in treating symptoms of IBS. Am J Gastroenterol. 2018;113:1290–300.
Moayyedi P, Quigley EM, Lacy BE, et al. The effect of fiber supplementation on irritable bowel syndrome: a systematic review and meta-analysis. Am J Gastroenterol. 2014;109(9):1367-1374. doi:10.1038/ajg.2014.195
Metamucil Package Insert. Proctor and Gamble Company. Revised 12- 23.
Ballou S, Keefer L. Psychological Interventions for Irritable Bowel Syndrome and Inflammatory Bowel Diseases. Clin Transl Gastroenterol. 2017;8(1):e214. Published 2017 Jan 19. doi:10.1038/ctg.2016.69
Lacy BE, Pimentel M, Brenner DM, et al. ACG Clinical Guideline: Management of Irritable Bowel Syndrome. Am J Gastroenterol. 2021;116(1):17-44. doi:10.14309/ajg.0000000000001036
Chang L, Sultan S, Lembo A, Verne GN, Smalley W, Heidelbaugh JJ. AGA Clinical Practice Guideline on the Pharmacological Management of Irritable Bowel Syndrome With Constipation. Gastroenterology. 2022;163(1):118-136. doi:10.1053/j.gastro.2022.04.016
Ibsrela (Tenapanor) Package Insert. Ardelyx. 2019.
Trulance (Plecanatide) Package Insert. Salence Pharmaceuticals. 2021.
Zelnorm (Tegaserod) Package Insert. 2019.
Amitiza (lubiprostone) Package Insert. 2012.
Linzess (Linaclotide) Package Insert. 2017.
Chey WD, Lembo AJ, Rosenbaum DP. Tenapanor Treatment of Patients With Constipation-Predominant Irritable Bowel Syndrome: A Phase 2, Randomized, Placebo-Controlled Efficacy and Safety Trial. Am J Gastroenterol. 2017;112(5):763-774. doi:10.1038/ajg.2017.41.
Chey WD, Lembo AJ, Rosenbaum DP. Efficacy of Tenapanor in Treating Patients With Irritable Bowel Syndrome With Constipation: A 12-Week, Placebo-Controlled Phase 3 Trial (T3MPO-1). Am J Gastroenterol. 2020;115(2):281-293. doi:10.14309/ajg.0000000000000516
Chey WD, Lembo AJ, Yang Y, Rosenbaum DP. Efficacy of Tenapanor in Treating Patients With Irritable Bowel Syndrome With Constipation: A 26-Week, Placebo-Controlled Phase 3 Trial (T3MPO-2). Am J Gastroenterol. 2021;116(6):1294-1303. doi:10.14309/ajg.0000000000001056
Brenner DM, Fogel R, Dorn SD, et al. Efficacy, safety, and tolerability of plecanatide in patients with irritable bowel syndrome with constipation:
results of two phase 3 randomized clinical trials. Am J Gastroenterol.
2018;113(5):735-745. doi:10.1038/s41395-018-0026-7
Yang Y, Fang J, Guo X, et al. Linaclotide in irritable bowel syndrome with constipation: A Phase 3 randomized trial in China and other regions. J Gastroenterol Hepatol. 2018;33(5):980-989. doi:10.1111/jgh.14086
Müller-Lissner SA, Fumagalli I, Bardhan KD, et al. Tegaserod, a 5-HT(4) receptor partial agonist, relieves symptoms in irritable bowel syndrome patients with abdominal pain, bloating and constipation. Aliment Pharmacol Ther. 2001;15(10):1655-1666. doi:10.1046/j.1365- 2036.2001.01094.x
Lefkowitz M, Shi Y, Schmitt C, Krumholz S, Tanghe J: The 5-HT4 partial agonist, tegaserod, improves abdominal discomfort/pain and normalizes altered bowel function in irritable bowel syndrome (IBS). Am J Gastroenterol. 1999;94:266.
Food and Drug Administration. FDA Briefing Document: Joint Meeting of the Gastrointestinal Drugs Advisory Committee and Drug Safety and Risk Management Advisory Committees. Published October 17, 2018.https://www.fda.gov/media/119011/download. Accessed March 16, 2024.
Novick J, Miner P, Krause R, et al. A randomized, double-blind, placebo- controlled trial of tegaserod in female patients suffering from irritable bowel syndrome with constipation. Aliment Pharmacol Ther. 2002;16(11):1877-1888. doi:10.1046/j.1365-2036.2002.01372
Drossman DA, Chey WD, Johanson JF, et al. Clinical trial: lubiprostone in patients with constipation-associated irritable bowel syndrome-- results of two randomized, placebo-controlled studies. Aliment Pharmacol Ther. 2009;29(3):329-341. doi:10.1111/j.1365- 2036.2008.03881
Lembo A, Sultan S, Chang L, Heidelbaugh JJ, Smalley W, Verne GN. AGA Clinical Practice Guideline on the Pharmacological Management of Irritable Bowel Syndrome With Diarrhea. Gastroenterology. 2022;163(1):137-151. doi:10.1053/j.gastro.2022.04.017
Viberzi (Eluxadoline) Package Insert. 2017.
Xifaxan (Rifaximin) Package Insert. 2017.
Lotronex (Alosetron) Package Insert. 2002.
Dove LS, Lembo A, Randall CW, et al. Eluxadoline benefits patients with irritable bowel syndrome with diarrhea in a phase 2 study. Gastroenterology. 2013;145(2):329-38.e1. doi:10.1053/j.gastro.2013.04.006
Pimentel M, Lembo A, Chey WD, et al. Rifaximin therapy for patients with irritable bowel syndrome without constipation. N Engl J Med. 2011;364(1):22-32. doi:10.1056/NEJMoa1004409
Lembo A, Zakko SF, Ferreira NL, et al. T1390 Rifaximin for the Treatment of Diarrhea-Associated Irritable Bowel Syndrome: Short Term Treatment Leading to Long Term Sustained Response. Gastroenterology. 2008;134:A–545.
Longstreth GF, Thompson WG, Chey WD, Houghton LA, Mearin F, Spiller RC. Functional bowel disorders [published correction appears in Gastroenterology. 2006 Aug;131(2):688]. Gastroenterology. 2006;130(5):1480-1491. doi:10.1053/j.gastro.2005.11.061
Lembo A, Pimentel M, Rao SS, et al. Repeat Treatment With Rifaximin Is Safe and Effective in Patients With Diarrhea-Predominant Irritable Bowel Syndrome. Gastroenterology. 2016;151(6):1113-1121. doi:10.1053/j.gastro.2016.08.003
DISCLAIMER
The information provided in this course is general in nature, and it is solely designed to provide participants with continuing education credit(s). This course and materials are not meant to substitute for the independent, professional judgment of any participant regarding that participant’s professional practice, including but not limited to patient assessment, diagnosis, treatment, and/or health management. Medical and pharmacy practices, rules, and laws vary from state to state, and this course does not cover the laws of each state; therefore, participants must consult the laws of their state as they relate to their professional practice.
Healthcare professionals, including pharmacists and pharmacy technicians, must consult with their employer, healthcare facility, hospital, or other organization, for guidelines, protocols, and procedures they are to follow. The information provided in this course does not replace those guidelines, protocols, and procedures but is for academic purposes only, and this course’s limited purpose is for the completion of continuing education credits.
Participants are advised and acknowledge that information related to medications, their administration, dosing, contraindications, adverse reactions, interactions, warnings, precautions, or accepted uses are constantly changing, and any person taking this course understands that such person must make an independent review of medication information prior to any patient assessment, diagnosis, treatment and/or health management. Any discussion of off-label use of any medication, device, or procedure is informational only, and such uses are not endorsed hereby.
Nothing contained in this course represents the opinions, views, judgments, or conclusions of RxCe.com LLC. RxCe.com LLC is not liable or responsible to any person for any inaccuracy, error, or omission with respect to this course, or course material.
© RxCe.com LLC 2024: All rights reserved. No reproduction of all or part of any content herein is allowed without the prior, written permission of RxCe.com LLC.
