MATH MATTERS: A REVIEW OF CALCULATIONS FOR STERILE COMPOUNDING
Liz Fredrickson, PharmD, BCPS
Liz Fredrickson is an Associate Professor of Pharmacy Practice and Pharmaceutical Sciences at the Northeast Ohio Medical University College of Pharmacy. She serves as Director of Instructional Labs and is course director for the Basic Pharmaceutics Lab and Parenteral Products and Parenteral Products Lab courses.
Topic Overview
A strong understanding of pharmaceutical calculations provides pharmacy technicians and pharmacists the ability to prepare compounded sterile preparations (CSPs) accurately and safely. When performing calculations, it is extremely important that error is avoided. This course provides an overview of fundamental pharmaceutical calculations and related concepts, including units of measure, conversions, the ratio and proportion method, dimensional analysis, and expressions of concentrations. Displacement volume and flow rate calculations will be reviewed, as well as calculations related to drug dosing and reconstitution. Step-by-step example problems will be provided to aid the learner in understanding these concepts.
Accreditation Statement

RxCe.com LLC is accredited by the Accreditation Council for Pharmacy Education (ACPE) as a provider of continuing pharmacy education.
Universal Activity Number (UAN): The ACPE Universal Activity Number assigned to this activity is
Pharmacist 0669-0000-23-218-H07-P
Pharmacy Technician 0669-0000-23-219-H07-T
Credits: 2 contact hours of continuing education credit
Type of Activity: Application
Media: Internet/Home study Fee Information: $6.99
Estimated time to complete activity: 2 contact hours, including Course Test and course evaluation
Release Date: December 16, 2023 Expiration Date: December 16, 2026
Target Audience: This educational activity is for pharmacists.
How to Earn Credit: From December 16, 2023, through December 16, 2026, participants must:
Read the “learning objectives” and “author and planning team disclosures;”
Study the section entitled “educational activity;” and
Complete the Course Test and Evaluation form. The Course Test will be graded automatically. Following successful completion of the Course Test with a score of 70% or higher, a statement of participation will be made available immediately. (No partial credit will be given.)
Credit for this course will be uploaded to CPE Monitor®.
Learning Objectives: Upon completion of this educational activity, participants should be able to:
Convert units of measurement
Recognize methods used to complete sterile compounding calculations
Perform calculations necessary to prepare compounded sterile preparations
Disclosures
The following individuals were involved in developing this activity: Liz Fredrickson, PharmD, BCPS, and Pamela Sardo, PharmD, BS. Pamela Sardo, PharmD, BS, was an employee of Rhythm Pharmaceuticals until March 2022 and has no conflicts of interest or relationships regarding the subject matter discussed. There are no financial relationships relevant to this activity to report or disclose by any of the individuals involved in the development of this activity.
© RxCe.com LLC 2023: All rights reserved. No reproduction of all or part of any content herein is allowed without the prior, written permission of RxCe.com LLC.
Introduction
Preparing a compounded sterile preparation requires successfully completing many steps. This course will provide an overview of fundamental pharmaceutical calculations and related concepts, including units of measurement, conversions, the ratio and proportion method, dimensional analysis, and expressions of concentrations. Displacement volume and flow rate calculations will be reviewed, as well as calculations related to drug dosing and reconstitution. Step-by-step example problems will be provided to aid the learner in understanding these concepts.
Calculation Considerations
Successfully preparing a compounded sterile preparation (CSP) requires accurately weighing ingredients, compounding the preparation, labeling the preparation, and documenting the process.1 A key component to ensuring the quality and accuracy of preparations is performing pharmaceutical calculations. Pharmaceutical calculations encompass an area of study that applies the basic principles of mathematics to the preparation and efficacious use of pharmaceutical preparations.2 This allows compounding personnel to verify doses, calculate flow rates, and determine the correct quantities of ingredients to be measured. Within United States Pharmacopeia (USP) Chapter <797> Pharmaceutical Compounding - Sterile Preparations, the ability to accurately complete calculations is a requirement of compounding personnel and those who directly oversee compounding personnel.1
Errors in calculations can have potentially devastating consequences, including major patient harm or death.3 Thus, the goal in performing calculations should be one hundred percent accuracy.3 If not, patients may receive a subtherapeutic product and derive little or no benefit.3 Conversely, the product could be supratherapeutic, resulting in adverse events or toxicities.3
Before performing pharmaceutical calculations, compounding personnel should establish a standardized process. The following questions can be used to guide this process. The following questions can be asked to establish this process:2
What is the goal of this problem?
What mathematical process(es) is/are needed to solve this problem?
How can I correctly complete those processes?
Performing calculations involves thoroughly reviewing both the medication order and formula used to compound the preparation. If there are errors within the medication order, these will need to be addressed.2 Additionally, the formula may need to be adjusted to meet the specific needs of the patient. Once these documents have been reviewed, the next step is to organize all the information necessary to perform calculations.2 This could include the patient’s weight or the quantity of medication requested by the medication order.2
There are often many methods by which to perform pharmaceutical calculations. While it is beneficial to be familiar with the various methods by which to solve a problem, it is best to use a method that is comfortable and familiar.2 It is also very important to double-check your work or have a colleague check your work prior to compounding the preparation.2 When performing calculations, it is best practice to use as few steps as possible.2 The more steps involved, the more likely a mathematical error is to occur.2 It is also helpful to approximate the correct answer prior to completing the calculation.2 This provides you with an estimate with which to compare your actual answer. For example, you can determine if your answer is higher or lower than anticipated and recognize a mistake prior to compounding the preparation. It is also useful to think critically and determine how reasonable your answer seems in terms of the units of measurement and the numerical value of the answer.2 Finally, the best way to become skillful at calculations is to practice! The more you review and study these concepts, the more competent you will become.
Systems of Measurement
Various measurement systems are utilized within pharmacy practice, and quantities of ingredients (volumes or weights) used to create sterile compounds can be expressed using various systems of measurement. For compounding personnel, it is important to understand these systems and how to convert between them. There are four main systems of measurement. These include:
The metric system
The apothecary system
The avoirdupois system
The household system
The Metric System
The metric system is the most utilized measurement system within pharmaceutical compounding, and it is also the preferred system of measurement within the practice of pharmacy.2 Metric units of weight and volume are commonly utilized to determine dosages and drug strengths within sterile compounding.2 The metric system is based on the decimal system, in which measurements are made in multiples (or fractions) of 10. The metric system includes three base units: gram for weight, liter for volume, and meter for length.2
Prefixes are used to denote ingredient quantities that are either larger or smaller than these base units.2,3 These prefixes are defined in Table 1.2 Compounding personnel commonly need to adjust between units of the metric system. Understanding these prefixes allows compounding personnel to adjust from one denomination to another easily.
Table 1
Metric System Prefixes2,3
| Prefix | Meaning |
| mega- | one million times the base unit (106) |
| kilo- | one thousand times the base unit (103) |
| deci- | one-tenth the base unit (10-1) |
| centi- | one-hundredth the base unit (10-2) |
| milli- | one-thousandth the base unit (10-3) |
| micro- | one-millionth the base unit (10-6) |
| nano- | one-billionth the base unit (10-9) |
Converting from one denomination to another is a simple process that involves moving the decimal point either to the left or right. For example, to convert from liters to milliliters, you would move the decimal point three places to the right.2 Doing so signifies multiplying by 1000.2 Also important is that when adding, subtracting, multiplying, or dividing, you must make sure all values have the same units.2 A value in grams cannot be added to a value in milligrams without first converting to the same unit.2 For example, you cannot add 100 mg to 1 g. Instead, you could convert 1 gram to 1000 mg and then add 100 mg + 1000 mg to get a total of 1100 mg.
Example: Convert 700 milligrams (mg) to grams (g)
To convert from milligrams to grams, recall that 1 gram is equal to 1000 mg. This calculation then involves moving the decimal point to the left (or dividing by 1000). Knowing this, the following calculation can be set up:
700 mg x 1 g/1000 mg = x
Milligrams cancel, leaving the desired units of grams in the final answer: 700 mg x 1 g/1000 mg = x
x = 0.70 grams
The Apothecary System
Historically, the apothecary system was more widely utilized than it is today. However, its use persists among some healthcare professionals, so being able to convert between the apothecary system and metric system is helpful for compounding personnel.3 Within this system, the base unit of measure for weight is the grain.2,3 One grain is equivalent to 65 milligrams.3 Common units of measure for volume include the fluid ounce, gallon, quart, and pint.2,3
The Avoirdupois System
The avoirdupois system is used for measurements of weight, most commonly in the context of bulk or stock packages of drugs.2,3 Within the avoirdupois system, the grain is the basic unit of measurement.2,3 Other common units of measurement include the pound and ounce.2,3
The Household System
A final system of measurement is known as the household system. This is commonly utilized when measuring liquid dosage forms, such as suspensions and solutions.3 Prescribers may refer to units of measurement such as tablespoon and teaspoon when prescribing liquid medications. Because of this, the use of the household system may lead to errors in drug dosing, as patients will sometimes use measuring devices (such as soup spoons) that are not calibrated.3 It is best practice to dispense a calibrated measuring device (such as a syringe) with liquid compounded preparations to avoid dosing errors.3
Common Units of Measure
Table 2 details common units of weight, and Table 3 details common units of volume.
Table 2 Units of Weight
| Unit | Abbreviation | System |
| gram | g | metric |
| milligram | mg | metric |
| kilogram | kg | metric |
| microgram | mcg | metric |
| ounce | oz | avoirdupois |
| pound | lb | avoirdupois |
| grain | gr | avoirdupois/apothecary |
Table 3 Units of Volume
| Unit | Abbreviation | System |
| teaspoon | tsp or t | household |
| tablespoon | tbsp or T | household |
| fluid ounce | fl oz | apothecary |
| gallon | gal | apothecary |
| quart | qt | apothecary |
| pint | pt | apothecary |
| milliliter | mL | metric |
| liter | L | metric |
Converting Between Units
While the metric system is most utilized, there may be times when compounding personnel need to convert from one system to another, establishing equivalencies between systems.3 Below are common conversions for weight- and volume-based measurements.
Common Conversions: Weight
The following are common conversions for weight.
1 gram = 1000 mg
1 milligram = 1000 mcg
1 kg = 1000 g
1 kilogram = 2.2 lbs
1 pound = 454 g
1 pound = 16 oz
1 ounce = 28.35 g
1 ounce = 437.5 gr
1 grain = 64.8 mg
Common Conversions: Volume
The following are common conversions for volume measurements.
1 tsp = 5 mL
1 tbsp = 15 mL
1 fl oz = 29.57 mL
1 gal = 3,785 mL
1 gal = 4 qt
1 qt = 2 pt
1 pt = 473 mL
1 gal = 128 fl oz
1 liter = 1000 mL
Example: How many fluid ounces are in 1 gallon?
Reference the above information to recognize that 1 gallon is equivalent to 128 fluid ounces.
Choose Your Own Adventure: Ratios and Proportion Method and Dimensional Analysis
Ratios and Proportions
The ratio and proportion method is a common strategy to solve for an unknown variable when three other variables are known.2,3 Ratios and proportions offer useful ways to organize calculations.3 To understand this method, the concepts of ratios and proportions will be discussed first. A ratio is defined as a comparison of two or more numbers that indicate their size in relation to each other.2,3 In a basic sense, a ratio is a relationship between two values.2 Ratios can be written in several ways, including as a quotient, fraction.3 For example, when discussing the ratio of 3 to 5, write it as:
3:5 or 3/5
The fraction form is commonly used when drug concentrations are expressed (for example, 10 mg/mL).2,3 However, it is important to recognize that if the ratio is written as a fraction, this should not be read as a fraction. Using our example above, 3/5 does not mean “three-fifths” but rather “3 to 5”.
A proportion is defined as two ratios that are set equal.2,3 Proportions are the practical application of ratios and are useful when two properties are directly related to each other.2,3 Like ratios, you may see proportions written in several different ways, including the following:
a/b = c/d a:b = c:d a:b :: c:d
Using our example of 3 to 5 above, write it as follows:
3/5 = 6/10
3:5 = 6:10
3:5::6:10
This is read as 3 is to 5 as 6 is to 10.
When setting up a proportion, it is very important that the numbers in the numerator (numbers on top) and the numbers in the denominator (numbers on bottom) match. In other words, expressions on opposite sides of the equal sign should have the same units.2 If these are switched, mathematical errors will result. To help avoid errors, you should always write the corresponding units of all values within a calculation.2
For example, consider the following problem. If 4 mg of drug are contained in every 5 mL of solution, how many milligrams of drug are contained in 10 mL of solution? To avoid confusion, instead of setting up the proportion like this:
4/5 = x/10
It should be set up like this:
4 mg/5 mL = x mg/10 mL
Example: If 80 mg of gentamicin is contained within 2 mL gentamicin sulfate injection, how many milligrams of gentamicin are contained in 5 mL of the injection?4
Begin this problem by setting up a proportion:
80 mg gentamicin sulfate/2 mL injection = x mg gentamicin/5 mL injection You can then cross-multiply and divide to isolate and solve for “x”:
80 mg (5 mL) = x mg (2 mL) x mg = (80 mg x 5 mL)/2 mL x = 200 mg
Check your work! If 80 milligrams are contained within 2 mL and 5 mL is larger than 2 mL, then our answer should be larger than 80 mg. Five is more than double two, so our answer should be more than double 80, or greater than 160.
Dimensional Analysis
A second method to solve for an unknown variable is known as dimensional analysis. This involves creating relationships between quantities such that all units cancel except for the desired units.2,3 Dimensional analysis allows for the use of one single calculation, compared to the multiple steps involved in the ratio and proportion method.2,3 This is also a useful method when you must convert between different units of measurement.3 To set up a dimensional analysis equation, the following steps can be used.2
Write out the desired unit(s) you need to obtain, followed by an equal sign
Identify a relationship between the information in your problem that contains the units you need for your answer. Include a unit with every number.
Connect the relationships inverting dimensions as needed and add known conversions so units will cancel.
Solve the problem mathematically
At times, a direct conversion may not be possible, and an intermediate conversion may need to be included.2 For example, to go from feet to meters, you may first need to convert from feet to inches and then from inches to centimeters then centimeters to meters.
Example: A patient will take 5 mg/kg/day of a drug each day. How many milliliters of a 20 mL vial containing 100 mg/mL should be administered to
this patient if they weigh 150 pounds? To solve this problem, determine the intended units for the answer, which are in milliliters. Then proceed through the remaining steps. Part of this problem involves recognizing that 1 kg is equal to 2.2 lbs.
? milliliters = 1 mL/100 mg x 5 mg/kg/day x 1 kg/2.2 lbs x 150 lbs =3.4
mL
Compounding personnel should be familiar with the ratio and proportion method and dimensional analysis but can utilize whichever method is more familiar and comfortable.
Drug Dosing Calculations
A key part of determining the accuracy of a compounded sterile preparation is checking to make sure a reasonable and accurate dose is ordered for the patient. A dose is defined as a specific quantity of medication that is taken at a specific time for an intended medical effect.3 Doses can be determined in many ways, including based on a patient’s weight, body surface area, or age.3
There are three steps involved when calculating a dose based on a patient’s body weight.4
Convert the weight to kilograms
Set up a proportion equation using the prescribed dose of medication on one side of the equal sign and the unknown on the other side of the equal sign.
Solve for X.
Example: A dose of vancomycin 15 mg/kg is ordered for a patient who weighs 180 lbs. What is the dose that should be administered?
To solve, first convert the body weight from pounds to kilograms: 180 lbs x 1 kg/2.2 lbs = 81.2 kg
Next, utilize the ratio and proportion method:
15 mg/1 kg = x mg/81.2 kg Finally, solve for x:
x = 15 x 81.2 =1218 mg of vancomycin
A dose based on concentration can also be calculated using the following steps.4
Determine the concentration of the available medication (information may be found on the label of the drug or calculated by the compounding personnel)
Set up a proportion equation. Use the concentration of available medication on one side of the equal sign and the desired dose on the other side of the equal sign.
Solve for X
Example: An order is received for ampicillin 3 g IV Q6H. To prepare this order, use a vial of ampicillin for injection, which contains 250 mg/2 mL. How many milliliters of ampicillin are needed?
To solve this, start by determining the concentration of the medication. In this case, the concentration is 250 mg per 2 mL.
Next, set up a proportion equation. 250 mg/2 mL = 3000 mg/x mL Finally, solve for X:
250 mg (x mL) = 2 mL (3000 mg) x = 6000/250 = 24 mL
Drug Concentration Expressions
Concentration
Concentration is an expression of the ratio of the amount of an ingredient to the amount of total product.3 Depending on the formula, this could be expressed as a percentage strength or ratio strength.2 Concentration is a very useful concept when determining quantities of ingredients needed for a sterile preparation. The following two equations can be used to either solve for concentration or the quantity of solute.3
Concentration = quantity of solute (or drug)/quantity of preparation
Quantity of solute (or drug) = concentration x quantity of preparation
Percent
A percent is simply defined as the number of parts in 100.3 You may see percent written as either a decimal or a fraction.3 For example, fifty percent can be noted as follows:
50% = 0.50 = 50/100
Percentage strength is an important concept when preparing CSPs.2,3 Percentage strength is the number of parts of solute or drug in 100 units of solution or total mixture.2,3 You may see percentage strength expressed in three different ways, including the following:
Percent weight-in-weight: the number of grams of solute or drug in 100 grams of total weight (solute + diluent) of the preparation2
For example: 3% w/w = 2 grams/100 grams
Percent weight-in-volume: the number of grams of solute or drug in 100 mL of the total volume (solute and solvent) of the preparation.
For example: 3% w/v = 3 grams/100 mL
Percent volume-in-volume: the number of milliliters of solute or drug in 100 mL of the total volume (solute and solvent) of the preparation.
For example: 3% v/v = 3 mL/100 mL
Example: What quantity of active ingredient is present in 250 mL of a 5% w/v solution?
To solve this problem, first recognize that 5% w/v means 5 g in 100 mL. Now, use the concentration equation to solve for the quantity of the drug in grams:
Quantity of solute= concentration x quantity of preparation Quantity of solute = 5 g/100 mL x 250 mL
Quantity of solute = 12.5 g Check it Out!
If 5% w/v means 5 g in 100 mL, the quantity of the drug in 250 mL should be larger than 5 g, given that 250 mL is larger than 100 mL. If the answer were less than 5 g, check to see where an error was made.
Example: Calculate the number of grams of sodium chloride (NaCl) in 1 L of 1/2NS (0.45% NS).
To solve, first set up the equation:
0.45 g/100 mL = x g/1000 mL
And then cross-multiply on both sides of the equal sign:
100(x) =450
And solve for X:
x = 450/100 = 4.5 g
One liter of ½ NS contains 4.5 grams of sodium chloride.
Ratio Strength
Ratio strength is another way to express concentration. This is an alternative method to percentage strength in which drug concentrations are expressed in terms of a ratio.2,3 Ratio strength is written as 1 unit of solute (or drug) in the total amount of the solution (or mixture), and the numbers are read as parts.2,3 For example: 1:300 would be read as 1 part in 300 parts. Ratio strength uses the same units as percentage strength. The second number in the ratio is always the total quantity of the mixture.2,3
Ratio volume-in-volume: this represents a liquid in a liquid (units: mL in mL) Example: 1:100 solution means 1 mL in 10 mL
Ratio weight-in-volume: this represents a solid in a liquid (units: g in mL) Example: 1:100 preparation means 1 g in 100 mL
Ratio weight-in-weight: this represents a solid in a solid (units: g in g) Example: 1:100 preparation means 1 g in 100 g
Example: Express 0.07% as a ratio strength.
To solve, recognize that 0.07% is equal to 0.07/100. Then, use the ratio and proportion method:
Set up a proportion: 0.07/100 = 1/x parts
Cross-multiply: 0.07 x (x parts) = 100
Divide and solve for x: x = (100/0.07) x = 1428 so the ratio strength is 1:1428
Example: What is the percentage strength of a 1:3000 w/v solution? To solve, use the ratio and proportion method:
Set up a proportion: 1/3000 = x/100
Cross-multiply: 3000(x) = 100 (1)
Divide and solve for x: x = 100/3000
x = 0.033, so the percentage strength is 0.033% w/v
Displacement Volume
Many drugs lose potency when they are prepared as a liquid dosage form and need to be provided as dry powders to extend shelf lives.2 These products then need to be reconstituted with purified water to prepare them in a liquid dosage form.2,3 Typically, these products can be reconstituted by following instructions to add the correct amount of purified water or diluent to achieve the intended concentration. The final volume of the preparation is the sum of the diluent and volume occupied by the powder.2,3 Compounding personnel may need to adjust the concentration of the medication based on the prescriber’s recommendation. This is especially vital when compounding pediatric medications, as the doses are smaller, and error is more likely to occur with only small changes in volume.4
Compounding personnel should be familiar with the equation for powder volume, which is the volume occupied by dry powder in a container.4 This equation can also be rearranged to solve for the final volume of a preparation.4
Powder volume = final volume – diluent volume
Final volume = diluent volume + powder volume
The steps below are used to calculate displacement volumes.
Calculate the powder volume
Calculate the volume of diluent needed for the new concentration
Subtract the powder volume from the diluent volume needed to determine the final amount that should be added
It is important to note not all medications cause volume displacement after reconstitution.4 You can check the package insert of the medication or vial label to determine this.4
Example: You receive a medication order for ampicillin 250 mg IM Q6H. To prepare this order, you will be using ampicillin for injection with a concentration of 200 mg/mL and a quantity of 1 gram per vial. The label instructions state to add 3.5 mL of diluent to the vial to create a concentration of 250 mg/mL. Calculate the powder volume for this concentration.
To solve this, start by calculating the powder volume for the 250 mg/mL concentration.
250 mg/1 mL = 1000 mg/x mL 250(x) = 1000
x = 4 mL
4 mL is the volume of diluent that would need to be added to the vial to create a concentration of 250 mg/mL if no volume displacement occurred. For the next step, subtract the volume of the diluent from this volume to determine the displacement or powder volume.
4 mL – 3.5 mL = 0.5 mL
Now, use this information to calculate the volume of diluent that needs to be added to obtain a new concentration of 200 mg/mL.
200 mg/1 mL = 1000 mg/x mL 200x = 1000
x = 5 mL
The final step is to subtract the powder volume from this new volume to determine how much diluent to add.
5 mL - 0.5 mL = 4.5 mL
So, 4.5 mL of diluent will be added to the vial to create a final concentration of 200 mg/mL.
Alligation Method
The alligation method can be used to determine a new strength of a product that is between two strengths of products you have available in the pharmacy.2,3 This method determines how many parts of each preparation strength should be mixed. The new product strength will be between the two strengths used.2,3 This type of problem can be set up by first drawing a “tic tac toe” board, as shown below.
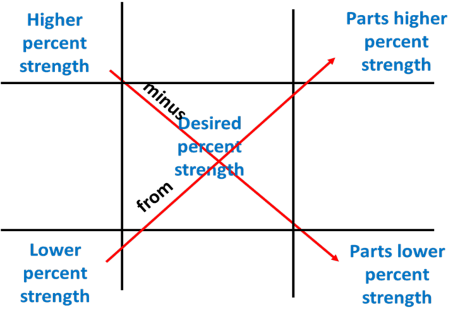
Next, values are placed as follows, using the “high goes high, low goes low” mnemonic. The higher percent strength value goes into the upper left corner of the board. The lower percent strength value goes into the lower left corner of the board. The desired strength goes into the middle of the board. Once these values are placed, the desired strength is subtracted from the higher percent strength, with the result placed into the lower right corner of the board. This value represents the parts of the lower percent strength product that are needed.4 Next, the lower percent strength is subtracted from the desired percent strength, with the resulting value placed in the upper right corner. This value represents the parts of the higher percent strength product that are needed.4 The two values on the right can be added to provide the total parts needed to compound the preparation.4
Example: What quantity of a 40% solution should be mixed with a 15% solution to create 1000 mL of a 20% solution?
To solve this, start by drawing a “tic tac toe” board.
Next, place your known and desired values. 40% is the higher percent strength, 15% is the lower percent strength, and 20% is the desired percent strength.

Now, subtract diagonally to determine parts of the 40% and 15% solutions needed.
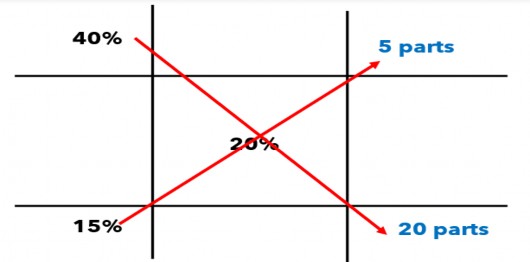
The total parts are equal to 20 + 5 or 25. The ratio and proportion method can be used to determine the quantity of the 40% solution as follows below.
5 parts/x mL = 25 parts/1000 mL x = 200 mL of the 40% solution
20 parts/x mL = 25 parts/1000 mL x = 800 mL of the 15% solution
Check it Out!
200 mL + 800 mL = 1000 mL, which is the total mentioned in the problem. This is a good initial indication that the problem has been solved correctly.
Flow Rate
Medications administered intravenously in hospitals and similar settings are given through electronic intravenous pumps.4 The rate at which each medication is infused is specific to that medication as well as the patient, and there can be adverse consequences when medications are infused too slowly or too quickly.4 The rate of infusion is also called the flow rate and is ultimately determined by recommendations from the manufacturer as well as standards set by the institution.4 Rates are programmed into infusion pumps using the units mL/hr, which allows these devices to regulate the flow to achieve the desired rate for the patient. There are two equations to solve for flow rate.4
Rate = Dose/Concentration Rate = Volume/Time
The steps below can be used to calculate flow rates using dimensional analysis.4
Set up dimensional analysis using the information you are provided. Make sure that all units in both the numerator and denominator cancel so your final units are in mL/hr or whatever the specified final units are.
Multiply all numerators
Multiply all denominators
Divide the answer in Step 2 by the answer in Step 3. Make sure all units in the numerator and denominator have been canceled appropriately to yield units of mL/hr.
Example: You receive an order for dopamine 400 mg in 250 mL D5W at 5 mcg/kg/min. The patient weighs 100 kg. Calculate the flow rate of dopamine in mL/hr. Round to the nearest whole number.
What equation should be used? Rate = dose/concentration
Start by converting the dose from 5 mcg/kg/min to mg/hr. Whenever doing weight-based dosing (as here), it is easiest to first multiply by the patient's weight to remove the units of "kg":
5 mcg/kg/min x 100 kg = 500 mcg/min
Now, change from mcg/min to mg/hr. Keep in mind 1 hr = 60 minutes and 1 mg = 1000 mcg: 500 mcg/min x 60 min/1 hr x 1 mg/1000 mcg = 30 mg/hr
The last step is to divide the dose by the concentration, which is the same as multiplying the dose by the inverse of the concentration: 30 mg/hr x 250 mL/400 mg = 18.75 mL/hr ⟶19 mL/hr (remember to round!)
Conversely, you could also complete this in one step using dimensional analysis: 5 mcg/kg/min x 100 kg x 60 min/1 hr x 1 mg/1000 mcg x 250 mL/400 mg = 19 mL/hr
Example: A 1500 mL IV bag of normal saline will be infused over 24 hours. Calculate the rate of infusion.
What equation should be used? Rate = volume/time
Rate = 1500 mL/24 hours Rate = 62.5 mL/hr
Summary
Understanding pharmaceutical calculations is important to ensure accuracy when compounding sterile preparations. Compounding personnel should be familiar with calculations involving ratios/proportions, dimensional analysis, expressions of concentration and displacement, and flow rate calculations. They should also be able to use the alligation method and complete calculations for dosing and reconstitution of powders.
Course Test
One gallon is equivalent to how many milliliters?
15 mL
29.57 mL
473 mL
3785 mL
Which of the following equations can be used to solve powder volume?
Powder volume = final volume – diluent volume
Powder volume = diluent volume – final volume
Powder volume = quantity of solute/quantity of preparation
Powder volume = volume of preparation – volume of solute
Which of the following equations can be used to calculate a flow rate?
Rate = time/volume
Rate = volume/concentration
Rate = dose/concentration
Rate = concentration/dose
Calculate the dose of vancomycin 17.5 mg/kg that will be administered to a patient who weighs 180 lbs.
10.2 mg
1010 mg
1430 mg
3150 mg
The label for a dry powder vial of an antibiotic has directions that state to add 1.5 mL of diluent to prepare 1.8 mL of injection. Calculate the powder volume occupied by the drug.
0.1 mL
0.3 mL
0.5 mL
0.6 mL
Convert 0.4% v/v to a ratio strength.
1:100
1:250
1:500
1:550
The label for a dry powder vial of an antibiotic has directions that state to add 1.5 mL of diluent to prepare 1.8 mL of injection. Calculate the concentration of the antibiotic in mg/mL. Round to the nearest whole number.
200 mg/mL
278 mg/mL
302 mg/mL
324 mg/mL
A 50% w/v dextrose solution and a 5% w/v dextrose solution are used to prepare 4500 mL of a 10% w/v solution. How many milliliters of the 5% solution are needed?
50 mL
400 mL
500 mL
4000 mL
A 50% w/v dextrose solution and a 5% w/v dextrose solution are used to prepare 4500 mL of a 10% w/v solution. How many milliliters of the 50% solution are needed?
50 mL
400 mL
500 mL
4000 mL
An order is placed for 250 mg of dobutamine in 250 mL of normal saline to be administered at a rate of 6.56 mcg/kg/min to a 190 lb patient. What is the flow rate in mL/hr?
13 mL/hr
23 mL/hr
34 mL/hr
43 mL/hr
References
General Chapter: USP. Pharmaceutical Compounding-Sterile Preparations <797>. In: USP-NF. Rockville, MD: USP; September 2023.
Teixeira M, Zatz J. Pharmaceutical Calculations. 5th ed. Wiley. 2017
Ansel H and Stockton S. Pharmaceutical Calculations. 15th edition. LWW. 2016.
Ochoa P and Vega J. Concepts in Sterile Preparations and Aseptic Technique. Jones and Bartlett Learning. 2015.
DISCLAIMER
The information provided in this course is general in nature, and it is solely designed to provide participants with continuing education credit(s). This course and materials are not meant to substitute for the independent, professional judgment of any participant regarding that participant’s professional practice, including but not limited to patient assessment, diagnosis, treatment, and/or health management. Medical and pharmacy practices, rules, and laws vary from state to state, and this course does not cover the laws of each state; therefore, participants must consult the laws of their state as they relate to their professional practice.
Healthcare professionals, including pharmacists and pharmacy technicians, must consult with their employer, healthcare facility, hospital, or other organization, for guidelines, protocols, and procedures they are to follow. The information provided in this course does not replace those guidelines, protocols, and procedures but is for academic purposes only, and this course’s limited purpose is for the completion of continuing education credits.
Participants are advised and acknowledge that information related to medications, their administration, dosing, contraindications, adverse reactions, interactions, warnings, precautions, or accepted uses are constantly changing, and any person taking this course understands that such person must make an independent review of medication information prior to any patient assessment, diagnosis, treatment and/or health management. Any discussion of off-label use of any medication, device, or procedure is informational only, and such uses are not endorsed hereby.
Nothing contained in this course represents the opinions, views, judgments, or conclusions of RxCe.com LLC. RxCe.com LLC is not liable or responsible to any person for any inaccuracy, error, or omission with respect to this course, or course material.
